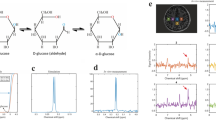
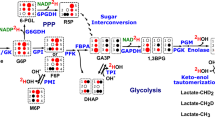
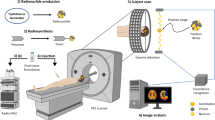
Abstract
Glucose is a major source of energy for most living organisms, and its aberrant uptake is linked to many pathological conditions. However, our understanding of disease-associated glucose flux is limited owing to the lack of robust tools. To date, positron-emission tomography imaging remains the gold standard for measuring glucose uptake, and no optical tools exist for non-invasive longitudinal imaging of this important metabolite in in vivo settings. Here, we report the development of a bioluminescent glucose-uptake probe for real-time, non-invasive longitudinal imaging of glucose absorption both in vitro and in vivo. In addition, we demonstrate that the sensitivity of our method is comparable with that of commonly used 18F-FDG-positron-emission-tomography tracers and validate the bioluminescent glucose-uptake probe as a tool for the identification of new glucose transport inhibitors. The new imaging reagent enables a wide range of applications in the fields of metabolism and drug development.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Hay, N. Reprogramming glucose metabolism in cancer: can it be exploited for cancer therapy? Nat. Rev. Cancer 16, 635–649 (2016).
Kaur, J. A comprehensive review on metabolic syndrome. Cardiol. Res. Pract. 2014, 21 (2014).
Chang, C.-H. & Pearce, E. L. Emerging concepts of T cell metabolism as a target of immunotherapy. Nat. Immunol. 17, 364–368 (2016).
Momcilovic, M. & Shackelford, D. B. Imaging cancer metabolism. Biomol. Ther. 26, 81–92 (2018).
Cox, B. L., Mackie, T. R. & Eliceiri, K. W. The sweet spot: FDG and other 2-carbon glucose analogs for multi-modal metabolic imaging of tumor metabolism. Am. J. Nucl. Med. Mol. Imaging 5, 1–13 (2015).
Hu, F. et al. Vibrational imaging of glucose uptake activity in live cells and tissues by stimulated raman scattering. Angew. Chem. 54, 9821–9825 (2015).
Kim, W. H., Lee, J., Jung, D. W. & Williams, D. R. Visualizing sweetness: increasingly diverse applications for fluorescent-tagged glucose bioprobes and their recent structural modifications. Sensors 12, 5005–5027 (2012).
Vallabhajosula, S., Solnes, L. & Vallabhajosula, B. A broad overview of positron emission tomography radiopharmaceuticals and clinical applications: what is new? Semin. Nucl. Med. 41, 246–264 (2011).
Tseng, J. C., Wang, Y., Banerjee, P. & Kung, A. L. Incongruity of imaging using fluorescent 2-DG conjugates compared to 18F-FDG in preclinical cancer models. Mol. Imaging Biol. 14, 553–560 (2012).
O’Neil, R. G., Wu, L. & Mullani, N. Uptake of a fluorescent deoxyglucose analog (2-NBDG) in tumor cells. Mol. Imaging Biol. 7, 388–392 (2005).
Speizer, L., Haugland, R. & Kutchai, H. Asymmetric transport of a fluorescent glucose analogue by human erythrocytes. Biochim. Biophys. Acta 815, 75–84 (1985).
Cheng, Z. et al. Near-infrared fluorescent deoxyglucose analogue for tumor optical imaging in cell culture and living mice. Bioconjug. Chem. 17, 662–669 (2006).
Kovar, J. L., Volcheck, W., Sevick-Muraca, E., Simpson, M. A. & Olive, D. M. Characterization and performance of a near-infrared 2-deoxyglucose optical imaging agent for mouse cancer models. Anal. Biochem. 384, 254–262 (2009).
Park, J., Lee, H. Y., Cho, M. H. & Park, S. B. Development of a cy3-labeled glucose bioprobe and its application in bioimaging and screening for anticancer agents. Angew. Chem. 46, 2018–2022 (2007).
Mezzanotte, L., van ‘t Root, M., Karatas, H., Goun, E. A. & Lowik, C. In vivo molecular bioluminescence imaging: new tools and applications. Trends Biotechnol. 35, 640–652 (2017).
Dothager, R. S. et al. Advances in bioluminescence imaging of live animal models. Curr. Opin. Biotechnol. 20, 45–53 (2009).
Genevois, C., Loiseau, H. & Couillaud, F. In vivo follow-up of brain tumor growth via bioluminescence imaging and fluorescence tomography. Int. J. Mol. Sci. 17, 1815 (2016).
Li, J., Chen, L., Du, L. & Li, M. Cage the firefly luciferin!—a strategy for developing bioluminescent probes. Chem. Soc. Rev. 42, 662–676 (2013).
Saxon, E. & Bertozzi, C. R. Cell surface engineering by a modified Staudinger reaction. Science 287, 2007–2010 (2000).
Sundhoro, M., Jeon, S., Park, J., Ramstrom, O. & Yan, M. Perfluoroaryl azide Staudinger reaction: a fast and bioorthogonal reaction. Angew. Chem. 56, 12117–12121 (2017).
Estensen, R. D. & Plagemann, P. G. W. Cytochalasin B: inhibition of glucose and glucosamine transport. Proc. Natl. Acad. Sci. USA 69, 1430–1434 (1972).
Park, J. et al. Impact of molecular charge on GLUT-specific cellular uptake of glucose bioprobes and in vivo application of the glucose bioprobe, GB2-Cy3. Chem. Commun. 50, 9251–9254 (2014).
Yun, J. et al. Glucose deprivation contributes to the development of KRAS pathway mutations in tumor cells. Science 325, 1555–1559 (2009).
Liu, Y. et al. A small-molecule inhibitor of glucose transporter 1 downregulates glycolysis, induces cell-cycle arrest, and inhibits cancer cell growth in vitro and in vivo. Mol. Cancer Ther. 11, 1672–1682 (2012).
Goodwin, J. et al. The distinct metabolic phenotype of lung squamous cell carcinoma defines selective vulnerability to glycolytic inhibition. Nat. Commun. 8, 15503 (2017).
Suzuki, S. et al. Involvement of GLUT1-mediated glucose transport and metabolism in gefitinib resistance of non-small-cell lung cancer cells. Oncotarget 9, 32667–32679 (2018).
Cushman, S. W. et al. Molecular mechanisms involved in GLUT4 translocation in muscle during insulin and contraction stimulation. Adv. Exp. Med. Biol. 441, 63–71 (1998).
Eguez, L. et al. Full intracellular retention of GLUT4 requires AS160 Rab GTPase activating protein. Cell Metab. 2, 263–272 (2005).
Capilla, E. et al. Functional characterization of an insulin-responsive glucose transporter (GLUT4) from fish adipose tissue. Am. J. Physiol. Endocrinol. Metab. 287, E348–E357 (2004).
Nedachi, T. & Kanzaki, M. Regulation of glucose transporters by insulin and extracellular glucose in C2C12 myotubes. Am. J. Physiol. Endocrinol. Metab. 291, E817–E828 (2006).
Maeda, N. et al. Diet-induced insulin resistance in mice lacking adiponectin/ACRP30. Nat. Med. 8, 731 (2002).
Zhang, J. H., Chung, T. D. & Oldenburg, K. R. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J. Biomol. Screen. 4, 67–73 (1999).
Blodgett, A. B. et al. A fluorescence method for measurement of glucose transport in kidney cells. Diabetes Technol. Ther. 13, 743–751 (2011).
Cao, Y. A. et al. Shifting foci of hematopoiesis during reconstitution from single stem cells. Proc. Natl. Acad.Sci. USA 101, 221–226 (2004).
Saltiel, A. R. & Kahn, C. R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414, 799–806 (2001).
Szablewski, L. Expression of glucose transporters in cancers. Biochim. Biophys. Acta 1835, 164–169 (2013).
Ojelabi, O. A., Lloyd, K. P., Simon, A. H., De Zutter, J. K. & Carruthers, A. WZB117 (2-fluoro-6-(m-hydroxybenzoyloxy) phenyl m-hydroxybenzoate) inhibits GLUT1-mediated sugar transport by binding reversibly at the exofacial sugar binding site. J. Biol. Chem. 291, 26762–26772 (2016).
Chavez, A. et al. Precise Cas9 targeting enables genomic mutation prevention. Proc. Natl. Acad. Sci. USA 115, 3669 (2018).
Shah, L., Laughlin, S. T. & Carrico, I. S. Light-activated Staudinger–Bertozzi ligation within living animals. J. Am. Chem. Soc. 138, 5186–5189 (2016).
Sletten, E. M. & Bertozzi, C. R. Bioorthogonal chemistry: fishing for selectivity in a sea of functionality. Angew. Chem. 48, 6974–6998 (2009).
Young, C. D. et al. Modulation of glucose transporter 1 (GLUT1) expression levels alters mouse mammary tumor cell growth in vitro and in vivo. PLoS ONE 6, e23205–e23205 (2011).
Wellberg, E. A. et al. The glucose transporter GLUT1 is required for ErbB2-induced mammary tumorigenesis. Breast Cancer Res. 18, 131–131 (2016).
Hall, M. P. et al. Click beetle luciferase mutant and near infrared naphthyl-luciferins for improved bioluminescence imaging. Nat. Commun. 9, 132 (2018).
Loening, A. M. & Gambhir, S. S. AMIDE: a free software tool for multimodality medical image analysis. Mol. Imaging 2, 131–137 (2003).
Venneti, S. et al. Glutamine-based PET imaging facilitates enhanced metabolic evaluation of gliomas in vivo. Sci. Transl. Med. 7, 274ra217 (2015).
Momcilovic, M. & Shackelford, D. B. Targeting LKB1 in cancer—exposing and exploiting vulnerabilities. Br. J. Cancer 113, 574–584 (2015).
Acknowledgements
We thank the Leenaards Foundation, NCCR Chemical Biology and Swiss National Foundation (grant no. CR23I3_157023) for generous financial support. We thank the laboratory of P. Aebischer for the kind gift of C2C12-luc cells. We thank the laboratory of D. Felsher and A. Stahl for the kind gifts of human embryonic kidney 293 TN and 3T13-L1 cells, respectively. We thank J. Frigell for the synthesis of several GAz derivatives (data not shown). We thank A. Konovalova for performing initial in vitro experiments on GAz probes (data not shown) and help with the technical scale-up of some of the reagents. We also thank P. Gonschorek and B. Mangeat for help with analyzing sequences of Slc2a1 gene in CRISPR/Cas9-mediated GLUT1 knockout and in negative control. We also thank the laboratory of K. Sakamoto for the kind gift of GLUT4 antibodies. We thank EPFL core facilities for histology, mass spectrometry and nuclear magnetic resonance analysis (in particular, P. Mieville and E. Baudat for technical support).
Author information
Authors and Affiliations
Contributions
E.G. conceptualized the study. T.M., G.M., P.K., R.S., A.B. and A.Y. carried out the in vitro and in vivo experiments (except the PET study). N.B., P.K. and R.S. synthesized CLP and various GAz reagents. T.M. and A.B. carried out reaction kinetic studies. D.S., A.J., V.M. and G.A. designed and performed PET imaging experiments. E.G., T.M. and G.M. wrote the manuscript. A.Y., P.K., R.S., A.B., N.B. and D.S. edited the manuscript. E.G. acquired the funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Investigation of the mechanism of CLP cellular internalization.
(a) 4T1-luc cells were incubated with various concentrations of CLP (1, 5, 10, 20, 30 and 40 μM) in KRH buffer for different time intervals (5, 15, 30, 45, 60 and 120 min). After incubation, the plates were rinsed twice with PBS and immediately imaged with IVIS camera. Total photon flux was plotted vs. incubation time for each concentration to determine the slope, which represents the rate of CLP transport. The resulting values of transport rate were plotted vs concentration of CLP. We observed linear dependence of transport rate vs CLP concentration, which indicates that CLP is transported inside the cell via passive diffusion. Results are mean of 3 independent experiments. (b) Temperature dependence of CLP uptake. 4T1-luc cells were placed at 4 °C or 37 °C for 15 min followed by 60 min incubation with 10 μM CLP in KRH buffer at 4 °C or 37 °C. After incubation, the plates were rinsed twice with PBS and immediately imaged with IVIS camera. We observed significant decrease in total photon flux from cells incubated at 4 °C compared to control cells incubated at 37 °C, which confirms the internalization of CLP via a passive diffusion. Data are presented as mean ± SD (n = 4, biologically independent samples). P value was calculated using two tailed t-test. (c) Bioluminescent signal production by 4T1-luc cells incubated with CLP as a function of time. 4T1-luc cells were treated with 10 μM of CLP in KRH buffer and incubated for different time intervals (5, 15, 30, 45, 60 and 120 min). After incubation, the plates were rinsed twice with PBS and immediately imaged with IVIS camera. The best fit line was determined by linear regression analysis (r2 = 0.9204). The slope of this line represents the rate of transport. Data are represented as mean ± SD (n = 3, independent experiments). The results presented in experiments b and c further indicate that CLP is transported inside the cell via passive diffusion.
Supplementary Figure 2 Determination of the optimal concentration of GAz4 for in vitro assays.
Concentration dependent bioluminescent light production from BiGluc probe in 3 different cell lines expressing luciferase (4T1-luc, HT1080-luc, C2C12-luc). The cells were treated with 10 µM of CLP in KRH buffer for one hour. The cells were rinsed twice with PBS followed by the addition of solutions with various concentrations of GAz4 reagent in KRH buffer ranging from 5 µM to 500 µM (x-axis). Each data point on the graph represents total bioluminescent signal integrated over 30 min and normalized to the background (CLP alone). Data are represented as mean ± SD (n = 4, biologically independent experiment).
Supplementary Figure 3 The in vitro cytotoxicity and BrdU proliferation assay of GAz4 and CLP reagents in 4T1-luc, C2C12-luc and HepG2-luc cells.
Three different types of cells were incubated for 1 or 24 h with 1 nM-5 mM concentrations of either GAz4 or CLP (upper graph in every panel). Cytotoxicity was determined using AlamarBlue assay as described in the instruction of Invitrogen commercial kit (Invitrogen, DAL1025). Resazurin, the component of this assay, oxidizes NADH to give a highly fluorescent compound and provides information on cellular redox state. Data are presented as mean ± SD (n = 3, biologically independent experiment). Cell viability was calculated as a percentage of viable cells, data were normalized to DMF concentration. Cell proliferation was measured with bromodeoxyuridine (BrdU) assay (bottom graph in every panel). Cells were incubated in growing medium supplemented with BrdU and either GAz4 (100 μM) or CLP (10 μM) for different time intervals (24, 48, and 72 h) at 37 °C and 5% CO2. BrdU incorporation into replicating DNA was measured by commercially available ELISA kit (Abcam, ab126572). According to manufacturer’s instruction. Data are presented as mean ± SD, n = 3, biologically independent experiments.
Supplementary Figure 4 Quantification of NAD+ levels in 4T1-luc cells treated with various concentration of GAz4 for 1 or 24 h.
4T1-luc cells were incubated with either 100 μM GAz4 for one hour (a) or various concentrations of GAz4 (50, 100 and 250 μM) for 24 h (b) at 37 °C and 5% CO2. NAD+ levels were determined using EnzyChrom NAD/NADH Assay kit according to manufacturer’s instructions (BioAssays Systems, Hayward, CA, USA). No significant difference in NAD+ levels was observed with different concentrations of GAz4 and incubation times. Results are shown as mean ± SD (n = 4 in a and n = 2 in b, biologically independent experiments). ns – Non-significant, p>0.05 by two-tailed t-test.
Supplementary Figure 5 HPLC-MS study of cross-reactivity of GAz4 with naturally occurring thiols.
The GAz4 was designed to have an enhanced reactivity toward triphenylphosphines by introducing multiple electron-withdrawing groups, which potentially makes it a strong oxidizer. Reactions with selected naturally abundant reducing agents (Cys, Glutathione (GSH)) were studied in order to check the biocompatibility of GAz4 probe. TCEP was used as a positive control to generate the reduced form of GAz4 as a reference. GAz4 solution (2.5 µL, 20 mM in DMSO) was added to a HPLC vial with solution of a reducing agent (50 µL, 5 mM in water) at room temperature. After onset, the reaction mixture was incubated at 37 °C and analyzed by HPLC-MS at 0, 10, 20, and 60 min. N/D – not detected.
Supplementary Figure 6 Comparison of BiGluc probe with radiolabeled and fluorescent probe upon inhibition of GLUT-mediated transport with endofacial and exofacial inhibitors.
(a) Radioisotope-based ([3H]-2DG) measurement of glucose uptake in the presence or absence of GLUT inhibitor, cytochalasin B (10 µM), in 4T1-luc cells. (b) Light production from BiGluc probe in the presence or absence of cytochalasin B in two different cell lines (4T1-luc and HT1080-luc). The cells were first incubated for one hour with CLP (10 µM in 100 µL KRH) followed by 2 washes with PBS. Next, cells were treated for 10 min with cytochalasin B (10 µM in 100 µL KRH), washed, and incubated with GAz4 (100 µM in 100 µL KRH) reagent followed by immediate signal acquisition using CCD camera (IVIS, PerkinElmer, USA). The total photon flux from cells was adjusted to the appropriate D-luciferin control to account for the influence of cytochalasin B treatment on luciferase activity. (c) Measurement of glucose uptake by fluorescent probe GB2-Cy3 in the presence or absence of GLUT inhibitor 4,6-EDG (50 mM) in 4T1-luc cells. (d) Light production from BiGluc probe in the presence or absence of exofacial GLUT inhibitor, 4,6-O-ethylidene-α-D-glucose (4,6-EDG), in 4T1-luc cells. The cells were first incubated for one hour with CLP (10 µM in 100 µL KRH) followed by 2 washes with PBS. Next, cells were treated for 30 min with 4,6-EDG (50 mM in 100 µL KRH), washed, and incubated with GAz4 (100 µM in 100 µL KRH) reagent followed by immediate signal acquisition using CCD camera (IVIS, PerkinElmer, USA). The total photon flux from cells was normalized to the background (CLP alone) and then was adjusted to the appropriate D-luciferin control to account for the influence of 4,6-EDG treatment on luciferase activity. Data are presented as mean ± SD (n = 4 in a, c and d and n = 5 in b, biologically independent experiments). P values were calculated by two-tailed t-test
Supplementary Figure 7 Characterization of CRISPR/Cas9-mediated GLUT1 knockout and negative control 4T1-luc cells.
(a) The scheme depicts the part of the Slc2a1 gene between exons 3 and 6, surrounding the targeting regions (named, A, B and C) of CRISPR/Cas9 plasmids that are a mixture of three gRNAs (Supplementary Table 1). Targeting regions were amplified using two sets of primers: primers oCF and oCR for region C, and primers oABF and oABR for region AB. Representative sequencing results of TA cloned PCR fragments from a parental cell line (4T1-luc), two negative non-targeting gRNAs control clones (NC no. 1, NC no. 2) and two knockout clones (GLUT1-/- no. 1, GLUT1-/- no. 2) are shown. In total, 10 colonies per region per clone were sequenced. There was no change observed in the sequence of NC clones compared to the parental control line. GLUT1 knockout clones harbored knockout alleles with frameshift mutation resulting from NHEJ. In addition, one knockout allele in the GLUT-/- no. 1 clone resulted in a deletion of 1062 bp between the region C and regions AB. (b) Summary table of sequencing results showing mutations in the targeted regions. (c) Western blot analysis of GLUT1 expression in 4T1-luc cells, 4T1-luc NC (no. 1, no. 2) and 4T1-luc- GLUT1-/- (no. 1, no. 2). For gel source images, see Supplementary Fig. 15. All experiments were performed independently at least twice.
Supplementary Figure 8 Quantification of d-glucose uptake in CRISPR/Cas9-mediated GLUT1 knockout and shRNA-mediated GLUT1 knockdown in 4T1-luc cells using the BiGluc probe.
(a) Comparison of the glucose uptake in parental, non-coding gRNA control and GLUT1 knockout 4T1-luc cells (4T1-luc, 4T1-luc NC (no. 1, 2) and 4T1-luc- GLUT1-/- (no. 1, 2), respectively). Labels no. 1, no. 2 stand for two different clones. The cells were first incubated for one hour with CLP (10 µM), followed by 2 washes with PBS. Next, they were treated with GAz4 (100 µM) reagent that immediately followed by signal acquisition. Bars represent integrated area under the curve (total photon flux) over 30 minutes normalized to the background (CLP alone). (b) Effect of endofacial (cytochalasin B; nonspecific) and exofacial (WZB-117; GLUT1 specific) inhibitors of GLUT on glucose uptake by 4T1-luc cell line and its clones. The cells were first incubated for one hour with CLP (10 µM in 100 µL KRH) that followed by 2 washes with PBS. Next, cells were treated for 10 min with cytochalasin B (10 µM in 100 µL KRH) or WZB-117 (50 µM in 100 µL KRH), washed, and incubated with GAz4 (100 µM in 100 µL KRH) reagent that followed by immediate signal acquisition using CCD camera (IVIS, PerkinElmer, USA). The total photon flux from cells was integrated over 30 min, normalized to the background (CLP alone) and then adjusted to the appropriate D-luciferin control to account for the influence of Cytochalasin B and WZB-117 treatment on luciferase activity. (c) Glucose uptake by control and GLUT1 shRNA transduced 4T1 cells (shGLUT no. 1-5) measured with BiGluc probe. The cells were first incubated for one hour with CLP (10 µM) that followed by 2 washes with PBS. Next, they were treated with GAz4 (100 µM) reagent that followed by signal acquisition. The total photon flux from cells was integrated over 30 min, normalized to the background (CLP alone). (d) Western blot analysis of GLUT1 expression in a control cell line and five shGLUT1 clones. Data on panels (a)-(c) are represented as mean ± SD (n = 4, biologically independent experiment). P values were calculated using two-tailed t-test. ns – Non-significant, p>0.05. T-test was performed between control and shGLUT1 (no. 1-5) in c. All experiments were performed independently at least twice. For gel source images, see Supplementary Fig. 15.
Supplementary Figure 9 Differentiation of C2C12-luc myotubes.
(a) Morphology of C2C12-luc cells before (left) and after (right) incubation in differentiation medium for 5 days. Phase-contrast microscopy was performed with EVOS FL Auto microscope, 10×/0.30 NA objective (Life Technologies, California, USA). Bars, 400 µm. (b) Western blot analysis of differentiation status. C2C12-luc myoblast were incubated in differentiation medium for 9 days. Samples for western blots analysis of Myosin, Glycogen synthase and GLUT4 expression were taken on day 0, 1, 4, 7 and 9. All experiments were performed independently at least twice. For gel source images, see Supplementary Fig. 15.
Supplementary Figure 10 Quantification of d-glucose uptake upon insulin stimulation in adipocytes using BiGluc probe.
(a) Morphology of 3T3-L1-luc cells before (up) and after (bottom) incubation in differentiation medium. Phase-contrast microscopy was performed with EVOS FL Auto microscope, 20×/0.40 NA objective (Life Technologies, California, USA). Bars, 200 µm. (b) The effect of insulin stimulation on glucose uptake by 3T3-L1-luc adipocytes measure using BiGluc probe. The cells were treated with 10 µM of CLP with or without 100 nM of insulin in 100 µl of KRH buffer for one hour. The plates were rinsed twice with PBS that followed by the addition of 100 µM GAz4 in KRH buffer. Bars represent area under the curve (total photon flux) over 30 min, normalized to the background level (CLP alone). Data are presented as mean ± SD (n = 4, biologically independent experiment). P values were calculated using two-tailed t-test. All experiments were performed independently at least twice.
Supplementary Figure 11 Investigation of sensitivity and reliability of bioluminescent BiGluc assay in comparison to radioactive ([3H]-2DG) and fluorescent (2-NBDG) methods for quantification of insulin-induced glucose influx to C2C12-luc myotubes.
The cells were incubated with [3H]-2DG, BiGluc or 2-NBDG probe with various concentrations of insulin (0–200 nM) in KRH buffer. Effect of insulin was measured in relative units (RU, y-axis). RU were calculated by normalization of the total signal after insulin stimulation to the signal corresponding to the basal level of glucose uptake. Each data point represents mean ± SD (n = 4, biologically independent experiments). Linear regression analysis was used to determine the best-fit line and its slope. Table shows slopes of best-fit lines and z’ factors for three assays. The slopes characterize sensitivity of an approach for insulin induced glucose uptake. Slopes are presented as best fit value ± SEM. Z’ factors indicate reliability of an assay for detection of insulin induced glucose uptake.
Supplementary Figure 12 Investigation of the CLP clearance from plasma.
(a) Evolution of the whole-body BL-signal in time after injection of CLP in FVB-luc+/+ mice. The mice (n = 5) received i.v. injection of CLP (1.5 mM in 100 µl) and immediately after were imaged using CCD camera (IVIS Spectrum, PerkinElmer, USA). Images were acquired at 0, 4, 8, 12 and 24 hours post injection. Representative animal images are shown below. (b) Reduction of CLP concentration in blood plasma over time. Mice were injected with 1.5 mM CLP and the blood was collected at different time points. An excess of GAz4 was then added to blood plasma and the mixture was immediately added on top of 4T1-luc cells. The resulting BL signal was normalized to 1 µM luciferin signal in order to account for well to well cell number variation. Bars represent integrated area under the curve (total photon flux) over 30 minutes. Data are presented as mean ± SD (n = 5 in a, and n = 3 in b, biologically independent samples). (c) Evolution in time of whole-body BL-signal from FVB-luc+/+ mice treated with BiGluc probe. The mice received i.v. injection of 100 µL solution of CLP (1.5 mM in 0.1% BSA in PBS) followed by i.p. injection of GAz4 solution 24 h later. Bioluminescent signal acquisition started immediately after injection and was followed for 3 hours. Each point represents mean value (n = 3, biologically independent samples), error bars represent SD. (d) Signal to background ratio from various organs excised from FVB-luc+/+ mice 15 and 45 min after injection of BiGluc probe. Data are presented as mean ± SEM, n = 2 biologically independent samples.
Supplementary Figure 13 Characterization of 4T1-luc tumor xenografts (parent and GLUT1-knockout lines).
(a) Western blot analysis of GLUT1 expression in excised tumors. As expected, GLUT1 was present in 4T1-luc but not in 4T1-luc-GLUT1-/- no. 1. (b) Immunohistochemical staining of excised 4T1-luc and 4T-luc-GLUT1-/- no. 1 tumors after in vivo non-invasive imaging with BiGluc probe. Tumors were stained with an antibody selectively recognizing GLUT1 protein. Scale bar = 100 µm. (c) Total photon flux from tumor-bearing mice injected with 100 µM of D-luciferin in the presence and absence of GLUT1 specific inhibitor (WZB-117). The data indicate that the presence of the inhibitor does not have any effect on the light output from tumors. Data are represented as mean ± SD (n = 4 per group, biologically independent experiment). (d) Average tumor volume before 18F-FDG PET/CT imaging. No significant difference between groups was observed. Data are represented as mean ± SD (n = 10 per group, biologically independent experiment). P values were calculated using two-tailed t-test, ns – p>0.05. All experiments were performed at least twice.
Supplementary Figure 14 Immunohistochemical staining of 18F-FDG PET/CT imaged 4T1-luc and 4T1-luc-GLUT1−/− no. 1 tumors and characterization of 18F-FDG positive 4T1-luc-GLUT1−/− no. 1 tumors.
(a) Tumors were stained with an antibody recognizing the GLUT1 protein. The blue and red squares indicate 4T1-luc and 4T1-luc-GLUT1-/- tumors that were stained, respectively. Scale bar = 50 µm. All experiments were performed independently four times. (b) Comparison of SUVmax values of 4T1-luc tumors and two 18F-FDG-positive 4T1-luc-GLUT1-/- no. 1 tumors that escaped CRISPR-mediated deletion of GLUT1. The pink arrow indicates 4T1-luc tumor and the green arrow indicates 4T1-luc-GLUT1-/- no. 1 tumor. P values were calculated using a two-tailed t-test, ns – p>0.05. The data are presented as the mean ± SEM (n = 10 in 4T1-luc tumors, and n = 2 in GLUT1-deficient tumors, biologically independent experiments). (c) Immunohistochemical staining of (b). Scale bar = 50 µM.
Supplementary Figure 15 Full unedited gels from figures in the main text and Supplementary information.
Area under the red square is represented in the final figure.
Supplementary information
Supplementary Information
Supplementary Figs. 1–15 and Supplementary Notes 1–4
Supplementary Video 1
Measurement of cellular glucose uptake using BiGluc probe: The video shows the working principle of the BiGluc-based assay and role of each component of BiGluc probe in the generation of bioluminescent signal.
Source Data
Rights and permissions
About this article
Cite this article
Maric, T., Mikhaylov, G., Khodakivskyi, P. et al. Bioluminescent-based imaging and quantification of glucose uptake in vivo. Nat Methods 16, 526–532 (2019). https://doi.org/10.1038/s41592-019-0421-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-019-0421-z
This article is cited by
-
The transcription factor ChREBP Orchestrates liver carcinogenesis by coordinating the PI3K/AKT signaling and cancer metabolism
Nature Communications (2024)
-
Post-click labeling enables highly accurate single cell analyses of glucose uptake ex vivo and in vivo
Communications Biology (2024)
-
In vivo bioluminescence imaging of natural bacteria within deep tissues via ATP-binding cassette sugar transporter
Nature Communications (2023)
-
Recent Advances in Bioorthogonal Ligation and Bioconjugation
Topics in Current Chemistry (2023)
-
RuO2/rGO heterostructures as mimic peroxidases for colorimetric detection of glucose
Microchimica Acta (2022)