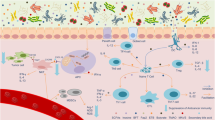
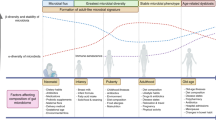
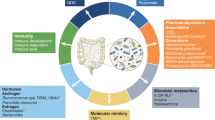
Abstract
Historically, cancer research and therapy have focused on malignant cells and their tumor microenvironment. However, the vascular, lymphatic and nervous systems establish long-range communication between the tumor and the host. This communication is mediated by metabolites generated by the host or the gut microbiota, as well by systemic neuroendocrine, pro-inflammatory and immune circuitries—all of which dictate the trajectory of malignant disease through molecularly defined biological mechanisms. Moreover, aging, co-morbidities and co-medications have a major impact on the development, progression and therapeutic response of patients with cancer. In this Perspective, we advocate for a whole-body ‘ecological’ exploration of malignant disease. We surmise that accumulating knowledge on the intricate relationship between the host and the tumor will shape rational strategies for systemic, bodywide interventions that will eventually improve tumor control, as well as quality of life, in patients with cancer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The paper does not contain any original or unpublished data.
References
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Nam, A. S., Chaligne, R. & Landau, D. A. Integrating genetic and non-genetic determinants of cancer evolution by single-cell multi-omics. Nat. Rev. Genet. 22, 3–18 (2021).
Vickovic, S. et al. SM-Omics is an automated platform for high-throughput spatial multi-omics. Nat. Commun. 13, 795 (2022).
Hanahan, D. Hallmarks of cancer: new dimensions. Cancer Discov. 12, 31–46 (2022).
Lopez-Otin, C. & Kroemer, G. Hallmarks of health. Cell 184, 33–63 (2021).
Martinez-Reyes, I. & Chandel, N. S. Cancer metabolism: looking forward. Nat. Rev. Cancer 21, 669–680 (2021).
Pavlova, N. N., Zhu, J. & Thompson, C. B. The hallmarks of cancer metabolism: still emerging. Cell Metab. 34, 355–377 (2022).
Stine, Z. E., Schug, Z. T., Salvino, J. M. & Dang, C. V. Targeting cancer metabolism in the era of precision oncology. Nat. Rev. Drug Discov. 21, 141–162 (2022).
Petrelli, F. et al. Association of obesity with survival outcomes in patients with cancer: a systematic review and meta-analysis. JAMA Netw. Open 4, e213520 (2021).
Yoo, S. K., Chowell, D., Valero, C., Morris, L. G. T. & Chan, T. A. Outcomes among patients with or without obesity and with cancer following treatment with immune checkpoint blockade. JAMA Netw. Open 5, e220448 (2022).
Lopez-Otin, C., Galluzzi, L., Freije, J. M. P., Madeo, F. & Kroemer, G. Metabolic control of longevity. Cell 166, 802–821 (2016).
Ringel, A. E. et al. Obesity shapes metabolism in the tumor microenvironment to suppress anti-tumor immunity. Cell 183, 1848–1866 (2020).
Brown, K. A. Metabolic pathways in obesity-related breast cancer. Nat. Rev. Endocrinol. 17, 350–363 (2021).
Kratofil, R. M. et al. A monocyte–leptin–angiogenesis pathway critical for repair post-infection. Nature 609, 166–173 (2022).
Boi, S. K. et al. Obesity diminishes response to PD-1-based immunotherapies in renal cancer. J. Immunother. Cancer 8, e000725 (2020).
McQuade, J. L. et al. Association of body-mass index and outcomes in patients with metastatic melanoma treated with targeted therapy, immunotherapy, or chemotherapy: a retrospective, multicohort analysis. Lancet Oncol. 19, 310–322 (2018).
Wang, Z. et al. Paradoxical effects of obesity on T cell function during tumor progression and PD-1 checkpoint blockade. Nat. Med. 25, 141–151 (2019).
Hahn, A. W. et al. Obesity is associated with altered tumor metabolism in metastatic melanoma. Clin. Cancer Res. CCR-22-2661 (2022).
Gurjao, C. et al. Discovery and features of an alkylating signature in colorectal cancer. Cancer Discov. 11, 2446–2455 (2021).
Llovet, J. M. et al. Hepatocellular carcinoma. Nat. Rev. Dis. Prim. 7, 6 (2021).
Papadimitriou, N. et al. An umbrella review of the evidence associating diet and cancer risk at 11 anatomical sites. Nat. Commun. 12, 4579 (2021).
Yonekura, S. et al. Cancer induces a stress ileopathy depending on beta-adrenergic receptors and promoting dysbiosis that contributes to carcinogenesis. Cancer Discov. 12, 1128–1151 (2022).
Baazim, H., Antonio-Herrera, L. & Bergthaler, A. The interplay of immunology and cachexia in infection and cancer. Nat. Rev. Immunol. 22, 309–321 (2022).
Larkin, J. R. et al. Metabolomic biomarkers in blood samples identify cancers in a mixed population of patients with nonspecific symptoms. Clin. Cancer Res. 28, 1651–1661 (2022).
Zhang, H. et al. Multiplexed nanomaterial-assisted laser desorption/ionization for pan-cancer diagnosis and classification. Nat. Commun. 13, 617 (2022).
Fujisaka, S. et al. Diet, genetics, and the gut microbiome drive dynamic changes in plasma metabolites. Cell Rep. 22, 3072–3086 (2018).
Gacesa, R. et al. Environmental factors shaping the gut microbiome in a Dutch population. Nature 604, 732–739 (2022).
Huang, S. et al. Identification and validation of plasma metabolomic signatures in precancerous gastric lesions that progress to cancer. JAMA Netw. Open 4, e2114186 (2021).
Ye, D., Guan, K. L. & Xiong, Y. Metabolism, activity, and targeting of d- and l-2-hydroxyglutarates. Trends Cancer 4, 151–165 (2018).
Malczewski, A. B., Navarro, S., Coward, J. I. & Ketheesan, N. Microbiome-derived metabolome as a potential predictor of response to cancer immunotherapy. J. Immunother. Cancer 8, e001383 (2020).
Wang, H. et al. The microbial metabolite trimethylamine N-oxide promotes antitumor immunity in triple-negative breast cancer. Cell Metab. 34, 581–594 (2022).
Taylor, S. R., Falcone, J. N., Cantley, L. C. & Goncalves, M. D. Developing dietary interventions as therapy for cancer. Nat. Rev. Cancer 22, 452–466 (2022).
Montegut, L., de Cabo, R., Zitvogel, L. & Kroemer, G. Science-driven nutritional interventions for the prevention and treatment of cancer. Cancer Discov. 12, 2258–2279 (2022).
Chen, A. C. et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N. Engl. J. Med. 373, 1618–1626 (2015).
Mitchell, S. J. et al. Nicotinamide improves aspects of healthspan, but not lifespan, in mice. Cell Metab. 27, 667–676 (2018).
Chandler, P. D. et al. Effect of vitamin D3 supplements on development of advanced cancer: a secondary analysis of the VITAL randomized clinical trial. JAMA Netw. Open 3, e2025850 (2020).
Peila, R. et al. A randomized trial of calcium plus vitamin d supplementation and risk of ductal carcinoma in situ of the breast. JNCI Cancer Spectr. 5, pkab072 (2021).
Ferrere, G. et al. Ketogenic diet and ketone bodies enhance the anticancer effects of PD-1 blockade. JCI Insight 6, e14207 (2021).
Dmitrieva-Posocco, O. et al. β-hydroxybutyrate suppresses colorectal cancer. Nature 605, 160–165 (2022).
Villarroel, A., Alvarez, O., Oberhauser, A. & Latorre, R. Probing a Ca2+-activated K+ channel with quaternary ammonium ions. Pflug. Arch. 413, 118–126 (1988).
Panebianco, C. et al. Butyrate, a postbiotic of intestinal bacteria, affects pancreatic cancer and gemcitabine response in in vitro and in vivo models. Biomed. Pharmacother. 151, 113163 (2022).
Hofer, S. J., Davinelli, S., Bergmann, M., Scapagnini, G. & Madeo, F. Caloric restriction mimetics in nutrition and clinical trials. Front Nutr. 8, 717343 (2021).
Allott, E. H. et al. Statin use is associated with lower risk of PTEN-null and lethal prostate cancer. Clin. Cancer Res. 26, 1086–1093 (2020).
Wang, Y. et al. A meta-analysis of statin use and risk of hepatocellular carcinoma. Can. J. Gastroenterol. Hepatol. 2022, 5389044 (2022).
Xu, W. H. & Zhou, Y. H. The relationship between post-diagnostic statin usage and breast cancer prognosis varies by hormone receptor phenotype: a systemic review and meta-analysis. Arch. Gynecol. Obstet. 304, 1315–1321 (2021).
Takada, K. et al. A propensity score-matched analysis of the impact of statin therapy on the outcomes of patients with non-small-cell lung cancer receiving anti-PD-1 monotherapy: a multicenter retrospective study. BMC Cancer 22, 503 (2022).
Santoni, M. et al. Statin use improves the efficacy of nivolumab in patients with advanced renal cell carcinoma. Eur. J. Cancer 172, 191–198 (2022).
Mao, W. et al. Statin shapes inflamed tumor microenvironment and enhances immune checkpoint blockade in non-small cell lung cancer. JCI Insight 7, e161940 (2022).
Nam, G. H. et al. Statin-mediated inhibition of RAS prenylation activates ER stress to enhance the immunogenicity of KRAS mutant cancer. J. Immunother. Cancer 9, e002474 (2021).
Khojandi, N. et al. Oxidized lipoproteins promote resistance to cancer immunotherapy independent of patient obesity. Cancer Immunol. Res 9, 214–226 (2021).
Harbeck, N. et al. Breast cancer. Nat. Rev. Dis. Prim. 5, 66 (2019).
Rebello, R. J. et al. Prostate cancer. Nat. Rev. Dis. Prim. 7, 9 (2021).
Caplin, M. E. & Ratnayake, G. M. Diagnostic and therapeutic advances in neuroendocrine tumours. Nat. Rev. Endocrinol. 17, 81–82 (2021).
Buque, A. et al. Immunoprophylactic and immunotherapeutic control of hormone receptor-positive breast cancer. Nat. Commun. 11, 3819 (2020).
Terrisse, S. et al. Immune system and intestinal microbiota determine efficacy of androgen deprivation therapy against prostate cancer. J. Immunother. Cancer 10, e004191 (2022).
Guan, X. et al. Androgen receptor activity in T cells limits checkpoint blockade efficacy. Nature 606, 791–796 (2022).
Pernigoni, N. et al. Commensal bacteria promote endocrine resistance in prostate cancer through androgen biosynthesis. Science 374, 216–224 (2021).
Vellano, C. P. et al. Androgen receptor blockade promotes response to BRAF/MEK-targeted therapy. Nature 606, 797–803 (2022).
Mauffrey, P. et al. Publisher Correction: Progenitors from the central nervous system drive neurogenesis in cancer. Nature 577, E10 (2020).
Silverman, D. A. et al. Cancer-associated neurogenesis and nerve-cancer cross-talk. Cancer Res. 81, 1431–1440 (2021).
Zhang, L. et al. Sympathetic and parasympathetic innervation in hepatocellular carcinoma. Neoplasma 64, 840–846 (2017).
Ferdoushi, A. et al. Tumor innervation and clinical outcome in pancreatic cancer. Sci. Rep. 11, 7390 (2021).
Ahmadi, N., Kelly, G., Low, T. H., Clark, J. & Gupta, R. Molecular factors governing perineural invasion in malignancy. Surg. Oncol. 42, 101770 (2022).
March, B. et al. Tumour innervation and neurosignalling in prostate cancer. Nat. Rev. Urol. 17, 119–130 (2020).
Gysler, S. M. & Drapkin, R. Tumor innervation: peripheral nerves take control of the tumor microenvironment. J. Clin. Invest. 131, e147276 (2021).
Wu, Y., Berisha, A. & Borniger, J. C. Neuropeptides in cancer: friend and foe? Adv Biol (Weinh) 6, e2200111 (2022).
Lu, C. et al. Hypoxia-activated neuropeptide Y/Y5 receptor/RhoA pathway triggers chromosomal instability and bone metastasis in Ewing sarcoma. Nat. Commun. 13, 2323 (2022).
Chakroborty, D. et al. Neuropeptide Y, a paracrine factor secreted by cancer cells, is an independent regulator of angiogenesis in colon cancer. Br. J. Cancer 127, 1440–1449 (2022).
Cheng, Y. et al. Depression-induced neuropeptide Y secretion promotes prostate cancer growth by recruiting myeloid cells. Clin. Cancer Res. 25, 2621–2632 (2019).
Gautam, J. et al. Tryptophan hydroxylase 1 and 5-HT7 receptor preferentially expressed in triple-negative breast cancer promote cancer progression through autocrine serotonin signaling. Mol. Cancer 15, 75 (2016).
Jiang, S. H. et al. Increased serotonin signaling contributes to the warburg effect in pancreatic tumor cells under metabolic stress and promotes growth of pancreatic tumors in mice. Gastroenterology 153, 277–291 (2017).
Li, T. et al. Overproduction of gastrointestinal 5-HT promotes colitis-associated colorectal cancer progression via enhancing NLRP3 inflammasome activation. Cancer Immunol. Res. 9, 1008–1023 (2021).
Gao, Y. et al. Glutamate decarboxylase 65 signals through the androgen receptor to promote castration resistance in prostate cancer. Cancer Res. 79, 4638–4649 (2019).
Tsuboi, M. et al. Prognostic significance of GAD1 overexpression in patients with resected lung adenocarcinoma. Cancer Med. 8, 4189–4199 (2019).
Huang et al. Cancer-cell-derived GABA promotes beta-catenin-mediated tumour growth and immunosuppression. Nat. Cell Biol. 24, 230–241 (2022).
Joseph, A. et al. Effects of acyl-coenzyme A binding protein (ACBP)/diazepam-binding inhibitor (DBI) on body mass index. Cell Death Dis. 12, 599 (2021).
Kim, H. B., Myung, S. K., Park, Y. C. & Park, B. Use of benzodiazepine and risk of cancer: a meta-analysis of observational studies. Int J. Cancer 140, 513–525 (2017).
Iqbal, U. et al. Is long-term use of benzodiazepine a risk for cancer? Medicine 94, e483 (2015).
Iqbal, U. et al. Benzodiazepines use and breast cancer risk: a population-based study and gene expression profiling evidence. J. Biomed. Inf. 74, 85–91 (2017).
Laforest, S. et al. Associations between markers of mammary adipose tissue dysfunction and breast cancer prognostic factors. Int J. Obes. 45, 195–205 (2021).
de Candia, P. et al. The pleiotropic roles of leptin in metabolism, immunity, and cancer. J. Exp. Med. 218, e20191593 (2021).
Manieri, E. et al. Adiponectin accounts for gender differences in hepatocellular carcinoma incidence. J. Exp. Med. 216, 1108–1119 (2019).
Wu, Q. et al. IGF1 receptor inhibition amplifies the effects of cancer drugs by autophagy and immune-dependent mechanisms. J. Immunother. Cancer 9, e002722 (2021).
Matsushita, M. et al. Gut microbiota-derived short-chain fatty acids promote prostate cancer growth via IGF1 signaling. Cancer Res. 81, 4014–4026 (2021).
Zhang, H. et al. Impact of corticosteroid use on outcomes of non-small-cell lung cancer patients treated with immune checkpoint inhibitors: a systematic review and meta-analysis. J. Clin. Pharm. Ther. 46, 927–935 (2021).
Wang, Y. et al. Corticosteroid administration for cancer-related indications is an unfavorable prognostic factor in solid cancer patients receiving immune checkpoint inhibitor treatment. Int. Immunopharmacol. 99, 108031 (2021).
Yang, H. et al. Stress-glucocorticoid-TSC22D3 axis compromises therapy-induced antitumor immunity. Nat. Med. 25, 1428–1441 (2019).
Deng, Y. et al. Glucocorticoid receptor regulates PD-L1 and MHC-I in pancreatic cancer cells to promote immune evasion and immunotherapy resistance. Nat. Commun. 12, 7041 (2021).
Janowitz, T., Kleeman, S. & Vonderheide, R. H. Reconsidering dexamethasone for antiemesis when combining chemotherapy and immunotherapy. Oncologist 26, 269–273 (2021).
Zhong, S. et al. Beta-blocker use and mortality in cancer patients: systematic review and meta-analysis of observational studies. Eur. J. Cancer Prev. 25, 440–448 (2016).
Sivanesan, S., Tasken, K. A. & Grytli, H. H. Association of beta-blocker use at time of radical prostatectomy with rate of treatment for prostate cancer recurrence. JAMA Netw. Open 5, e2145230 (2022).
Kokolus, K. M. et al. Beta blocker use correlates with better overall survival in metastatic melanoma patients and improves the efficacy of immunotherapies in mice. Oncoimmunology 7, e1405205 (2018).
Magnon, C. et al. Autonomic nerve development contributes to prostate cancer progression. Science 341, 1236361 (2013).
Allen, J. K. et al. Sustained adrenergic signaling promotes intratumoral innervation through BDNF induction. Cancer Res. 78, 3233–3242 (2018).
Kamiya, A. et al. Genetic manipulation of autonomic nerve fiber innervation and activity and its effect on breast cancer progression. Nat. Neurosci. 22, 1289–1305 (2019).
Thaker, P. H. et al. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat. Med. 12, 939–944 (2006).
Zahalka, A. H. et al. Adrenergic nerves activate an angio-metabolic switch in prostate cancer. Science 358, 321–326 (2017).
Rutledge, A., Jobling, P., Walker, M. M., Denham, J. W. & Hondermarck, H. Spinal cord injuries and nerve dependence in prostate cancer. Trends Cancer 3, 812–815 (2017).
Daher, C. et al. Blockade of beta-adrenergic receptors improves CD8+ T-cell priming and cancer vaccine efficacy. Cancer Immunol. Res 7, 1849–1863 (2019).
Shaashua, L. et al. Perioperative COX-2 and beta-adrenergic blockade improves metastatic biomarkers in breast cancer patients in a phase-II randomized trial. Clin. Cancer Res. 23, 4651–4661 (2017).
Haldar, R. et al. Perioperative COX2 and beta-adrenergic blockade improves biomarkers of tumor metastasis, immunity, and inflammation in colorectal cancer: a randomized controlled trial. Cancer 126, 3991–4001 (2020).
Wang, X. et al. Prognostic value of depression and anxiety on breast cancer recurrence and mortality: a systematic review and meta-analysis of 282,203 patients. Mol. Psychiatry 25, 3186–3197 (2020).
Van der Elst, S., Bardash, Y., Wotman, M., Kraus, D. & Tham, T. The prognostic impact of depression or depressive symptoms on patients with head and neck cancer: a systematic review and meta-analysis. Head Neck 43, 3608–3617 (2021).
Dinesh, A. A., Helena Pagani Soares Pinto, S., Brunckhorst, O., Dasgupta, P. & Ahmed, K. Anxiety, depression and urological cancer outcomes: a systematic review. Urol. Oncol. 39, 816–828 (2021).
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Man, S. M. & Jenkins, B. J. Context-dependent functions of pattern recognition receptors in cancer. Nat. Rev. Cancer 22, 397–413 (2022).
Derynck, R., Turley, S. J. & Akhurst, R. J. TGFβ biology in cancer progression and immunotherapy. Nat. Rev. Clin. Oncol. 18, 9–34 (2021).
Hou, J., Karin, M. & Sun, B. Targeting cancer-promoting inflammation—have anti-inflammatory therapies come of age? Nat. Rev. Clin. Oncol. 18, 261–279 (2021).
Hong, C. et al. cGAS–STING drives the IL-6-dependent survival of chromosomally instable cancers. Nature 607, 366–373 (2022).
Hua, X. et al. Association between post-treatment circulating biomarkers of inflammation and survival among stage II–III colorectal cancer patients. Br. J. Cancer 125, 806–815 (2021).
Shi, Y. et al. Circulating cytokines associated with clinical outcomes in advanced non-small cell lung cancer patients who received chemoimmunotherapy. Thorac. Cancer 13, 219–227 (2022).
Schalper, K. A. et al. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and reduced clinical benefit of immune-checkpoint inhibitors. Nat. Med. 26, 688–692 (2020).
Yuen, K. C. et al. High systemic and tumor-associated IL-8 correlates with reduced clinical benefit of PD-L1 blockade. Nat. Med. 26, 693–698 (2020).
Kroemer, G., Lopez-Otin, C., Madeo, F. & de Cabo, R. Carbotoxicity—noxious effects of carbohydrates. Cell 175, 605–614 (2018).
Chow, L. S. et al. Exerkines in health, resilience and disease. Nat. Rev. Endocrinol. 18, 273–289 (2022).
Font-Burgada, J., Sun, B. & Karin, M. Obesity and cancer: the oil that feeds the flame. Cell Metab. 23, 48–62 (2016).
Coombs, C. C. et al. Therapy-related clonal hematopoiesis in patients with non-hematologic cancers is common and associated with adverse clinical outcomes. Cell Stem Cell 21, 374–382 (2017).
Hong, W. et al. Clonal hematopoiesis mutations in patients with lung cancer are associated with lung cancer risk factors. Cancer Res. 82, 199–209 (2022).
Giles, A. J. et al. Activation of hematopoietic stem/progenitor cells promotes immunosuppression within the pre-metastatic niche. Cancer Res. 76, 1335–1347 (2016).
Routy, B. et al. The gut microbiota influences anticancer immunosurveillance and general health. Nat. Rev. Clin. Oncol. 15, 382–396 (2018).
Zhang, Z. J., Lehmann, C. J., Cole, C. G. & Pamer, E. G. Translating Microbiome Research From and To the Clinic. Annu. Rev. Microbiol. 76, 435–460 (2022).
Plovier, H. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 23, 107–113 (2017).
Bae, M. et al. Akkermansia muciniphila phospholipid induces homeostatic immune responses. Nature 608, 168–173 (2022).
Grajeda-Iglesias, C. et al. Oral administration of Akkermansia muciniphila elevates systemic antiaging and anticancer metabolites. Aging 13, 6375–6405 (2021).
Schneider, K. M. et al. Imbalanced gut microbiota fuels hepatocellular carcinoma development by shaping the hepatic inflammatory microenvironment. Nat. Commun. 13, 3964 (2022).
Depommier, C. et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat. Med. 25, 1096–1103 (2019).
Derosa, L. et al. Intestinal Akkermansia muciniphila predicts clinical response to PD-1 blockade in patients with advanced non-small-cell lung cancer. Nat. Med. 28, 315–324 (2022).
Lee, K. A. et al. Cross-cohort gut microbiome associations with immune checkpoint inhibitor response in advanced melanoma. Nat. Med. 28, 535–544 (2022).
Zeng, X. et al. Gut bacterial nutrient preferences quantified in vivo. Cell 185, 3441–3456 (2022).
Liu, Z. et al. Moderate-intensity exercise affects gut microbiome composition and influences cardiac function in myocardial infarction mice. Front. Microbiol. 8, 1687 (2017).
Munukka, E. et al. Six-week endurance exercise alters gut metagenome that is not reflected in systemic metabolism in over-weight women. Front. Microbiol. 9, 2323 (2018).
Simpson, R. C. et al. Diet-driven microbial ecology underpins associations between cancer immunotherapy outcomes and the gut microbiome. Nat. Med. 28, 2344–2352 (2022).
Kaplanov, I. et al. Blocking IL-1β reverses the immunosuppression in mouse breast cancer and synergizes with anti-PD-1 for tumor abrogation. Proc. Natl Acad. Sci. USA 116, 1361–1369 (2019).
Aggen, D. H. et al. Blocking IL1β promotes tumor regression and remodeling of the myeloid compartment in a renal cell carcinoma model: multidimensional analyses. Clin. Cancer Res. 27, 608–621 (2021).
Hailemichael, Y. et al. Interleukin-6 blockade abrogates immunotherapy toxicity and promotes tumor immunity. Cancer Cell 40, 509–523 (2022).
Bent, E. H. et al. Microenvironmental IL-6 inhibits anti-cancer immune responses generated by cytotoxic chemotherapy. Nat. Commun. 12, 6218 (2021).
Lopez-Bujanda, Z. A. et al. Castration-mediated IL-8 promotes myeloid infiltration and prostate cancer progression. Nat. Cancer 2, 803–818 (2021).
Perez-Ruiz, E. et al. Prophylactic TNF blockade uncouples efficacy and toxicity in dual CTLA-4 and PD-1 immunotherapy. Nature 569, 428–432 (2019).
Montfort, A. et al. Combining nivolumab and ipilimumab with infliximab or certolizumab in patients with advanced melanoma: first results of a phase Ib clinical trial. Clin. Cancer Res. 27, 1037–1047 (2021).
Demaria, M. et al. Cellular senescence promotes adverse effects of chemotherapy and cancer relapse. Cancer Discov. 7, 165–176 (2017).
Kohlhapp, F. J. et al. Venetoclax Increases intratumoral effector T cells and antitumor efficacy in combination with immune checkpoint blockade. Cancer Discov. 11, 68–79 (2021).
Triana-Martinez, F. et al. Identification and characterization of cardiac glycosides as senolytic compounds. Nat. Commun. 10, 4731 (2019).
Guerrero, A. et al. Cardiac glycosides are broad-spectrum senolytics. Nat. Metab. 1, 1074–1088 (2019).
Menger, L. et al. Cardiac glycosides exert anticancer effects by inducing immunogenic cell death. Sci. Transl. Med. 4, 143ra199 (2012).
Lin, T. P., Fan, Y. H., Chen, Y. C. & Huang, W. J. S. Digoxin lowers the incidence of prostate cancer: a nationwide population-based study. J. Chin. Med. Assoc. 83, 377–381 (2020).
Liao, X. et al. Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N. Engl. J. Med. 367, 1596–1606 (2012).
Simon, T. G. et al. Association of aspirin with hepatocellular carcinoma and liver-related mortality. N. Engl. J. Med. 382, 1018–1028 (2020).
Wang, Y. et al. Aspirin use and the risk of hepatocellular carcinoma: a meta-analysis. J. Clin. Gastroenterol. 56, e293–e302 (2022).
Liu, J., Zheng, F., Yang, M., Wu, X. & Liu, A. Effect of aspirin use on survival benefits of breast cancer patients: a meta-analysis. Medicine 100, e26870 (2021).
Xiao, S., Xie, W., Fan, Y. & Zhou, L. Timing of aspirin use among patients with colorectal cancer in relation to mortality: a systematic review and meta-analysis. JNCI Cancer Spectr. 5, pkab067 (2021).
McNeil, J. J. et al. Effect of aspirin on all-cause mortality in the healthy elderly. N. Engl. J. Med. 379, 1519–1528 (2018).
Zaman, F. Y., Orchard, S. G., Haydon, A. & Zalcberg, J. R. Non-aspirin non-steroidal anti-inflammatory drugs in colorectal cancer: a review of clinical studies. Br. J. Cancer 127, 1735–1743 (2022).
Arber, N. et al. Celecoxib for the prevention of colorectal adenomatous polyps. N. Engl. J. Med. 355, 885–895 (2006).
Burn, J. et al. Cancer prevention with aspirin in hereditary colorectal cancer (Lynch syndrome), 10-year follow-up and registry-based 20-year data in the CAPP2 study: a double-blind, randomised, placebo-controlled trial. Lancet 395, 1855–1863 (2020).
Pietrocola, F. et al. Aspirin recapitulates features of caloric restriction. Cell Rep. 22, 2395–2407 (2018).
Castoldi, F. et al. Autophagy-mediated metabolic effects of aspirin. Cell Death Discov. 6, 129 (2020).
Bessede, A., et al. Impact of acetaminophen on the efficacy of immunotherapy in cancer patients. Ann. Oncol. 33, 909–915 (2022).
Aiad, M. et al. Does the combined use of aspirin and immunotherapy result in better outcomes in non-small cell lung cancer than immunotherapy alone? Cureus 14, e25891 (2022).
Li, H. et al. The allergy mediator histamine confers resistance to immunotherapy in cancer patients via activation of the macrophage histamine receptor H1. Cancer Cell 40, 36–52 (2022).
Verdoodt, F. et al. Antihistamines and ovarian cancer survival: nationwide cohort study and in vitro cell viability assay. J. Natl Cancer Inst. 112, 964–967 (2020).
Chiang, C. H. et al. Efficacy of cationic amphiphilic antihistamines on outcomes of patients treated with immune checkpoint inhibitors. Eur. J. Cancer 174, 1–9 (2022).
Ridker, P. M. et al. Effect of interleukin-1beta inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet 390, 1833–1842 (2017).
Wong, C. C. et al. Inhibition of IL1β by canakinumab may be effective against diverse molecular subtypes of lung cancer: an exploratory analysis of the CANTOS Trial. Cancer Res. 80, 5597–5605 (2020).
Mullard, A. Novartis’s canakinumab stumbles in cancer, again. Nat. Rev. Drug Discov. 20, 888 (2021).
Fridman, W. H., Zitvogel, L., Sautes-Fridman, C. & Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 14, 717–734 (2017).
Fridman, W. H. et al. B cells and tertiary lymphoid structures as determinants of tumour immune contexture and clinical outcome. Nat. Rev. Clin. Oncol. 19, 441–457 (2022).
Yerly, L. et al. Integrated multi-omics reveals cellular and molecular interactions governing the invasive niche of basal cell carcinoma. Nat. Commun. 13, 4897 (2022).
Tselikas, L. et al. Feasibility, safety and efficacy of human intra-tumoral immuno-therapy. Gustave Roussy’s initial experience with its first 100 patients. Eur. J. Cancer 172, 1–12 (2022).
Le Cornet, C. et al. Circulating immune cell composition and cancer risk: a prospective study using epigenetic cell count measures. Cancer Res. 80, 1885–1892 (2020).
Jacquelot, N. et al. Predictors of responses to immune checkpoint blockade in advanced melanoma. Nat. Commun. 8, 592 (2017).
Lucca, L. E. et al. Circulating clonally expanded T cells reflect functions of tumor-infiltrating T cells. J. Exp. Med. 218, e20200921 (2021).
Holm, J. S. et al. Neoantigen-specific CD8 T cell responses in the peripheral blood following PD-L1 blockade might predict therapy outcome in metastatic urothelial carcinoma. Nat. Commun. 13, 1935 (2022).
Goubet, A. G. et al. Escherichia coli-specific CXCL13-producing TFH are associated with clinical efficacy of neoadjuvant PD-1 blockade against muscle-invasive bladder cancer. Cancer Discov. 12, 2280–2307 (2022).
Luoma, A. M. et al. Tissue-resident memory and circulating T cells are early responders to pre-surgical cancer immunotherapy. Cell 185, 2918–2935 (2022).
Bochem, J. et al. Early disappearance of tumor antigen-reactive T cells from peripheral blood correlates with superior clinical outcomes in melanoma under anti-PD-1 therapy. J. Immunother. Cancer 9, e003439 (2021).
Zitvogel, L., Perreault, C., Finn, O. J. & Kroemer, G. Beneficial autoimmunity improves cancer prognosis. Nat. Rev. Clin. Oncol. 18, 591–602 (2021).
Paillet, J. et al. Autoimmunity affecting the biliary tract fuels the immunosurveillance of cholangiocarcinoma. J. Exp. Med. 218, e20200853 (2021).
Zitvogel, L. & Kroemer, G. Cross-reactivity between microbial and tumor antigens. Curr. Opin. Immunol. 75, 102171 (2022).
Fluckiger, A. et al. Cross-reactivity between tumor MHC class I-restricted antigens and an enterococcal bacteriophage. Science 369, 936–942 (2020).
Kalaora, S. et al. Identification of bacteria-derived HLA-bound peptides in melanoma. Nature 592, 138–143 (2021).
Rouanne, M. et al. BCG therapy downregulates HLA-I on malignant cells to subvert antitumor immune responses in bladder cancer. J. Clin. Invest. 132, e145666 (2022).
Biot, C. et al. Preexisting BCG-specific T cells improve intravesical immunotherapy for bladder cancer. Sci. Transl. Med. 4, 137ra172 (2012).
Meier, S. L., Satpathy, A. T. & Wells, D. K. Bystander T cells in cancer immunology and therapy. Nat. Cancer 3, 143–155 (2022).
Upadhyay, R. et al. A critical role for fas-mediated off-target tumor killing in T-cell immunotherapy. Cancer Disco. 11, 599–613 (2021).
Leem, G. et al. Tumour-infiltrating bystander CD8+ T cells activated by IL-15 contribute to tumour control in non-small cell lung cancer. Thorax 77, 769–780 (2022).
Kohlhapp, F. J. et al. Non-oncogenic acute viral infections disrupt anti-cancer responses and lead to accelerated cancer-specific host death. Cell Rep. 17, 957–965 (2016).
Newman, J. H. et al. Intratumoral injection of the seasonal flu shot converts immunologically cold tumors to hot and serves as an immunotherapy for cancer. Proc. Natl Acad. Sci. USA 117, 1119–1128 (2020).
Routy, B. et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359, 91–97 (2018).
Derosa, L. et al. Microbiota-centered interventions: the next breakthrough in immuno-oncology? Cancer Discov. 11, 2396–2412 (2021).
Smith, M. et al. Gut microbiome correlates of response and toxicity following anti-CD19 CAR T cell therapy. Nat. Med. 28, 713–723 (2022).
Hagan, T. et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell 178, 1313–1328 (2019).
Vetizou, M. et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350, 1079–1084 (2015).
Luu, M. et al. Microbial short-chain fatty acids modulate CD8+ T cell responses and improve adoptive immunotherapy for cancer. Nat. Commun. 12, 4077 (2021).
Park, E. M. et al. Targeting the gut and tumor microbiota in cancer. Nat. Med. 28, 690–703 (2022).
Lopez-Otin, C., Pietrocola, F., Roiz-Valle, D., Galluzzi, L. & Kroemer, G. Meta-hallmarks of aging and cancer. Cell Metab. (in the press).
Lopez-Otin, C., Blasco, M., Partridge, L., Serano, M. & Kroemer, G. Hallmarks of aging. An expanding universe. Cell (in the press).
Vernieri, C. et al. Fasting-mimicking diet is safe and reshapes metabolism and antitumor immunity in patients with cancer. Cancer Discov. 12, 90–107 (2022).
Khodabakhshi, A. et al. Effects of ketogenic metabolic therapy on patients with breast cancer: a randomized controlled clinical trial. Clin. Nutr. 40, 751–758 (2021).
Spencer, C. N. et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science 374, 1632–1640 (2021).
Ma, X. et al. Sodium butyrate modulates gut microbiota and immune response in colorectal cancer liver metastatic mice. Cell Biol. Toxicol. 36, 509–515 (2020).
Li, P. et al. 1α,25(OH)2D3 reverses exhaustion and enhances antitumor immunity of human cytotoxic T cells. J. Immunother. Cancer 10, e003477 (2022).
Hiller, J. G. et al. Preoperative beta-blockade with propranolol reduces biomarkers of metastasis in breast cancer: a phase II randomized trial. Clin. Cancer Res. 26, 1803–1811 (2020).
Sanchez-Paulete, A. R. et al. Cancer immunotherapy with immunomodulatory anti-CD137 and anti-PD-1 monoclonal antibodies requires BATF3-dependent dendritic cells. Cancer Disco. 6, 71–79 (2016).
Kalanxhi, E. et al. Systemic immune response induced by oxaliplatin-based neoadjuvant therapy favours survival without metastatic progression in high-risk rectal cancer. Br. J. Cancer 118, 1322–1328 (2018).
Acknowledgements
G.K. and L.Z. are supported by the Ligue contre le Cancer (équipe labellisée); Agence National de la Recherche (ANR) – Projets blancs; Cancéropôle Ile-de-France; Fondation pour la Recherche Médicale (FRM); a donation by Elior; Equipex Onco-Pheno-Screen; Gustave Roussy Odyssea, the European Union Horizon 2020 Projects Oncobiome and Crimson; Institut National du Cancer (INCa); Institut Universitaire de France; LabEx Immuno-Oncology (ANR-18-IDEX-0001); a Cancer Research ASPIRE Award from the Mark Foundation; the RHU Immunolife; Seerave Foundation; SIRIC Stratified Oncology Cell DNA Repair and Tumor Immune Elimination (SOCRATE); and SIRIC Cancer Research and Personalized Medicine (CARPEM). This study contributes to the IdEx Université de Paris ANR-18-IDEX-0001. J.L.M. acknowledges the Transdisciplinary Research in Energetics and Cancer Research Training Workshop R25CA203650 and the MD Anderson Cancer Center (MDA) Center for Energy Balance in Cancer Prevention and Survivorship and is supported by ASCO/CCF, the Melanoma Research Alliance, the Elkins Foundation, Seerave Foundation, Rising Tide Foundation, the Mark Foundation for Cancer Research, MDA Melanoma SPORE Developmental Research Program Award, MDA Physician Scientist Program and MDA Moonshot Program. M.M. was supported by NIH grants U24 AI118644-05S1, R01CA257195 and R01CA254104 (Tumor macs), as well as by the Samuel Waxman Cancer Research Foundation. A.F. received ANR funding for IHU-B PRISM.
Author information
Authors and Affiliations
Contributions
G.K. wrote the first draft and then received major input from J.L.M., M.M., F.A. and L.Z. All authors have read, edited and approved the paper.
Corresponding author
Ethics declarations
Competing interests
G.K. holds research contracts with Daiichi Sankyo, Eleor, Kaleido, Lytix Pharma, PharmaMar, Osasuna Therapeutics, Samsara Therapeutics, Sanofi, Tollys and Vascage. G. K. consults for Reithera. G.K. is on the board of directors of the Bristol Myers Squibb Foundation France. G.K. is a scientific co-founder of everImmune, Osasuna Therapeutics, Samsara Therapeutics and Therafast Bio. G.K. is the inventor of patents covering therapeutic targeting of aging, cancer, cystic fibrosis and metabolic disorders. G.K.’s brother, R.omano Kroemer, was an employee of Sanofi and now consults for Boehringer-Ingelheim. J. M. has served in advisory roles for BMS, Merck and Roche. M.M. reports grants from Regeneron, Inc, outside the submitted work. F.A. has grants and advisory roles (compensated to the hospital) for Daiichi Sankyo, Pfizer, Novartis, Astra Zeneca, Lilly and Roche. L.Z. has held research contracts with 9 Meters Biopharma, Daiichi Sankyo, and Pilege, was on the on the Board of Directors of Transgene, is a co-founder of everImmune, and holds patents covering the treatment of cancer and the therapeutic manipulation of the microbiota. None of the funders had any role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Peer review
Peer review information
Nature Medicine thanks Marcel van den Brink, Matthew Spitzer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary handling editor: Karen O’Leary, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kroemer, G., McQuade, J.L., Merad, M. et al. Bodywide ecological interventions on cancer. Nat Med 29, 59–74 (2023). https://doi.org/10.1038/s41591-022-02193-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-022-02193-4
This article is cited by
-
Translational research on drug development and biomarker discovery for hepatocellular carcinoma
Journal of Biomedical Science (2024)
-
Targeting immunogenic cell stress and death for cancer therapy
Nature Reviews Drug Discovery (2024)