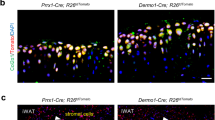
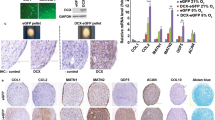
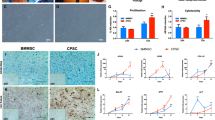
Abstract
Osteoarthritis (OA) is a degenerative disease resulting in irreversible, progressive destruction of articular cartilage1. The etiology of OA is complex and involves a variety of factors, including genetic predisposition, acute injury and chronic inflammation2,3,4. Here we investigate the ability of resident skeletal stem-cell (SSC) populations to regenerate cartilage in relation to age, a possible contributor to the development of osteoarthritis5,6,7. We demonstrate that aging is associated with progressive loss of SSCs and diminished chondrogenesis in the joints of both mice and humans. However, a local expansion of SSCs could still be triggered in the chondral surface of adult limb joints in mice by stimulating a regenerative response using microfracture (MF) surgery. Although MF-activated SSCs tended to form fibrous tissues, localized co-delivery of BMP2 and soluble VEGFR1 (sVEGFR1), a VEGF receptor antagonist, in a hydrogel skewed differentiation of MF-activated SSCs toward articular cartilage. These data indicate that following MF, a resident stem-cell population can be induced to generate cartilage for treatment of localized chondral disease in OA.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Microarray data described in this study was deposited with the gene expression omnibus with the accession number GSE151303. There are no restrictions on sharing the data. All data are available from the corresponding author upon reasonable request.
References
Robinson, W. H. et al. Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 12, 580–592 (2016).
Rahmati, M., Mobasheri, A. & Mozafari, M. Inflammatory mediators in osteoarthritis: a critical review of the state-of-the-art, current prospects, and future challenges. Bone 85, 81–90 (2016).
Marques-Rocha, J. L. et al. Noncoding RNAs, cytokines, and inflammation-related diseases. FASEB J. 29, 3595–3611 (2015).
Liu-Bryan, R. & Terkeltaub, R. Emerging regulators of the inflammatory process in osteoarthritis. Nat. Rev. Rheumatol. 11, 35–44 (2015).
Hoemann, C. D. et al. Chondroinduction is the main cartilage repair response to microfracture and microfracture with BST-CarGel: results as shown by ICRS-II histological scoring and a novel zonal collagen type scoring method of human clinical biopsy specimens. Am. J. Sports Med. 43, 2469–2480 (2015).
Mithoefer, K., McAdams, T., Williams, R. J., Kreuz, P. C. & Mandelbaum, B. R. Clinical efficacy of the microfracture technique for articular cartilage repair in the knee: an evidence-based systematic analysis. Am. J. Sports Med. 37, 2053–2063 (2009).
Goldberg, A., Mitchell, K., Soans, J., Kim, L. & Zaidi, R. The use of mesenchymal stem cells for cartilage repair and regeneration: a systematic review. J. Orthop. Surg. Res. 12, 39 (2017).
Vos, T. et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 380, 2163–2196 (2012).
Murray, C. J. et al. The state of US health, 1990–2010: burden of diseases, injuries, and risk factors. JAMA 310, 591–608 (2013).
Hootman, J. M., Helmick, C. G., Barbour, K. E., Theis, K. A. & Boring, M. A. Updated projected prevalence of self-reported doctor-diagnosed arthritis and arthritis-attributable activity limitation among US adults, 2015–2040. Arthritis Rheumatol. 68, 1582–1587 (2016).
Johnson, V. L. & Hunter, D. J. The epidemiology of osteoarthritis. Best. Pr. Res Clin. Rheumatol. 28, 5–15 (2014).
Marshall, M., Watt, F. E., Vincent, T. L. & Dziedzic, K. Hand osteoarthritis: clinical phenotypes, molecular mechanisms and disease management. Nat. Rev. Rheumatol. 14, 641–656 (2018).
Bijlsma, J. W., Berenbaum, F. & Lafeber, F. P. Osteoarthritis: an update with relevance for clinical practice. Lancet 377, 2115–2126 (2011).
Steadman, J. R., Rodkey, W. G., Briggs, K. K. & Rodrigo, J. J. [The microfracture technic in the management of complete cartilage defects in the knee joint]. Orthopade 28, 26–32 (1999).
Makris, E. A., Gomoll, A. H., Malizos, K. N., Hu, J. C. & Athanasiou, K. A. Repair and tissue engineering techniques for articular cartilage. Nat. Rev. Rheumatol. 11, 21–34 (2015).
Knutsen, G. et al. Autologous chondrocyte implantation compared with microfracture in the knee. A randomized trial. J. Bone Jt. Surg. Am. 86-A, 455–464 (2004).
Devitt, B. M., Bell, S. W., Webster, K. E., Feller, J. A. & Whitehead, T. S. Surgical treatments of cartilage defects of the knee: systematic review of randomised controlled trials. Knee 24, 508–517 (2017).
Vanlauwe, J. et al. Five-year outcome of characterized chondrocyte implantation versus microfracture for symptomatic cartilage defects of the knee: early treatment matters. Am. J. Sports Med. 39, 2566–2574 (2011).
Piuzzi, N. S. et al. Accelerated growth of cellular therapy trials in musculoskeletal disorders: an analysis of the NIH Clinical Trials Data Bank. Orthopedics 42, e144–e150 (2019).
Sipp, D., Robey, P. G. & Turner, L. Clear up this stem-cell mess. Nature 561, 455–457 (2018).
Jo, C. H. et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells 32, 1254–1266 (2014).
Vega, A. et al. Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation 99, 1681–1690 (2015).
Pers, Y. M. et al. Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: a phase I dose–escalation trial. Stem Cells Transl. Med. 5, 847–856 (2016).
Worthley, D. L. et al. Gremlin 1 identifies a skeletal stem cell with bone, cartilage, and reticular stromal potential. Cell 160, 269–284 (2015).
Sacchetti, B. et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 131, 324–336 (2007).
Chan, C. K. et al. Identification and specification of the mouse skeletal stem cell. Cell 160, 285–298 (2015).
Chan, C. K. F. et al. Identification of the human skeletal stem cell. Cell 175, 43–56 e21 (2018).
Ambrosi, T. H., Longaker, M. T. & Chan, C. K. F. A revised perspective of skeletal stem cell biology. Front Cell Dev. Biol. 7, 189 (2019).
Luria, E. A., Owen, M. E., Friedenstein, A. J., Morris, J. F. & Kuznetsow, S. A. Bone formation in organ cultures of bone marrow. Cell Tissue Res. 248, 449–454 (1987).
Gronthos, S., Simmons, P. J., Graves, S. E. & Robey, P. G. Integrin-mediated interactions between human bone marrow stromal precursor cells and the extracellular matrix. Bone 28, 174–181 (2001).
Kratchmarova, I., Blagoev, B., Haack-Sorensen, M., Kassem, M. & Mann, M. Mechanism of divergent growth factor effects in mesenchymal stem cell differentiation. Science 308, 1472–1477 (2005).
Marecic, O. et al. Identification and characterization of an injury-induced skeletal progenitor. Proc. Natl Acad. Sci. USA 112, 9920–9925 (2015).
Jiang, Y. & Tuan, R. S. Origin and function of cartilage stem/progenitor cells in osteoarthritis. Nat. Rev. Rheumatol. 11, 206–212 (2015).
Ransom, R. C. et al. Genetic dissection of clonal lineage relationships with hydroxytamoxifen liposomes. Nat. Commun. 9, 2971 (2018).
Loeser, R. F., Collins, J. A. & Diekman, B. O. Ageing and the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 12, 412–420 (2016).
Gulati, G. S. et al. Isolation and functional assessment of mouse skeletal stem cell lineage. Nat. Protoc. 13, 1294–1309 (2018).
Eldracher, M., Orth, P., Cucchiarini, M., Pape, D. & Madry, H. Small subchondral drill holes improve marrow stimulation of articular cartilage defects. Am. J. Sports Med. 42, 2741–2750 (2014).
Rinkevich, Y., Lindau, P., Ueno, H., Longaker, M. T. & Weissman, I. L. Germ-layer and lineage-restricted stem/progenitors regenerate the mouse digit tip. Nature 476, 409–413 (2011).
Fang, H. & Beier, F. Mouse models of osteoarthritis: modelling risk factors and assessing outcomes. Nat. Rev. Rheumatol. 10, 413–421 (2014).
Loeser, R. F. et al. Microarray analysis reveals age-related differences in gene expression during of harpagoside by upregulation of the BMP2 and Wnt signaling pathways in osteoblasts and suppression of differentiation in osteoclasts. J. Nat. Prod. 80, 434–442 (2017).
Ransom, R. C. et al. Mechanoresponsive stem cells acquire neural crest fate in jaw regeneration. Nature 563, 514–521 (2018).
Ray, A., Singh, P. N., Sohaskey, M. L., Harland, R. M. & Bandyopadhyay, A. Precise spatial restriction of BMP signaling is essential for articular cartilage differentiation. Development 142, 1169–1179 (2015).
Bragdon, B. et al. Earliest phases of chondrogenesis are dependent upon angiogenesis during ectopic bone formation in mice. Bone 101, 49–61 (2017).
Urist, M. R. Bone: formation by autoinduction. Science 150, 893–899 (1965).
Keeney, M. et al. Scaffold-mediated BMP-2 minicircle DNA delivery accelerated bone repair in a mouse critical-size calvarial defect model. J. Biomed. Mater. Res. A 104, 2099–2107 (2016).
Chung, H. J. et al. Anti-osteoporotic activity of harpagoside by upregulation of the BMP2 and Wnt signaling pathways in osteoblasts and suppression of differentiation in osteoclasts. J. Nat. Prod. 80, 434–442 (2017).
Gonzalez-Fernandez, T., Tierney, E. G., Cunniffe, G. M., O’Brien, F. J. & Kelly, D. J. Gene delivery of TGF-beta3 and BMP2 in an MSC-laden alginate hydrogel for articular cartilage and endochondral bone tissue engineering. Tissue Eng. Part A 22, 776–787 (2016).
Hamilton, J. L. et al. Targeting VEGF and its receptors for the treatment of osteoarthritis and associated pain. J. Bone Miner. Res. 31, 911–924 (2016).
Nagai, T. et al. Bevacizumab, an anti-vascular endothelial growth factor antibody, inhibits osteoarthritis. Arthritis Res Ther. 16, 427 (2014).
Tevlin, R. et al. Pharmacological rescue of diabetic skeletal stem cell niches. Sci. Transl. Med. 9, eaag2809 (2017).
Adouni, M. & Dhaher, Y. Y. A multi-scale elasto-plastic model of articular cartilage. J. Biomech. 49, 2891–2898 (2016).
Chandran, P. L., Dimitriadis, E. K., Mertz, E. L. & Horkay, F. Microscale mapping of extracellular matrix elasticity of mouse joint cartilage: an approach to extracting bulk elasticity of soft matter with surface roughness. Soft Matter 14, 2879–2892 (2018).
Kretzschmar, K. et al. Profiling proliferative cells and their progeny in damaged murine hearts. Proc. Natl Acad. Sci. USA 115, E12245–E12254 (2018).
Nagao, M. et al. Vascular endothelial growth factor in cartilage development and osteoarthritis. Sci. Rep. 7, 13027 (2017).
Steinberg, J. & Zeggini, E. Functional genomics in osteoarthritis: past, present, and future. J. Orthop. Res. 34, 1105–1110 (2016).
Prockop, D. J. et al. Defining the risks of mesenchymal stromal cell therapy. Cytotherapy 12, 576–578 (2010).
Jones, I. A., Togashi, R., Wilson, M. L., Heckmann, N. & Vangsness, C. T. Jr. Intra-articular treatment options for knee osteoarthritis. Nat. Rev. Rheumatol. 15, 77–90 (2019).
Conrad, B., Han, L. H. & Yang, F. Gelatin-based microribbon hydrogels accelerate cartilage formation by mesenchymal stem cells in three dimensions. Tissue Eng. Part A 24, 1631–1640 (2018).
Taylor, S. E. et al. Identification of human juvenile chondrocyte-specific factors that stimulate stem cell growth. Tissue Eng. Part A 22, 645–653 (2016).
Glyn-Jones, S. et al. Osteoarthritis. Lancet 386, 376–387 (2015).
Seita, J. et al. Gene Expression Commons: an open platform for absolute gene expression profiling. PLoS ONE 7, e40321 (2012).
Acknowledgements
The authors thank N. Williams, C. Queen, C. Crumpton, E. Kang, M. Butcher, L. Quinn, P. Perira and R. Mann for advice and assistance, and A. McCarty, M. Alvarez, and VSC Staff for animal model support. The authors also thank M. Ekhart, and the PAN facility at Stanford for help in gene-expression analysis. In addition, the authors thank S. Goodman and the Orthopedic Department of Stanford for donation of human adult specimens. The study was supported by NIH (R01 DE027323, R56 DE025597, R01 DE026730, R01 DE021683, R21 DE024230, U01HL099776, U24DE026914 and R21 DE019274), CIRMTR1-01249, Oak Foundation, Hagey Laboratory, Pitch Johnson Fund and Gunn/Olivier Research Fund to M.T. Longaker; Siebel Fellowship, PCF YI Award, Stinehart/Reed, and NIHNIAK99AG049958-01A1, American Federation of Aging Research and Arthritis National Research Foundation to C.K.F.C.; HHMI Fellowship to G.S.G.; the German Research Foundation (DFG) Fellowship 399915929 to T.H.A.; NIH, NIGMS R01GM123069, NIH1R01AR071379, ACS Clowes Award, Department of Defense CDMRP W81XWH-18-1-0653, OR170174 to B.L; PSRF National Endowment and Stanford TTE fellowship to M.P.M.; NIHS10 RR02933801 to Stanford Stem Cell FACS core. The project was supported, in part, by Award Number 1S10OD021514-01 from the National Center for Research Resources (NCRR). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NCRR or the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Conception and design was done by M.P.M., I.L.W., M.T. Longaker and C.K.F.C. Experiments, data analysis and data interpretation were carried out by M.P.M., L.S.K., M.T. Lopez, T.H.A., G.S.G., O.M., Y.W., R.C.R., M.Y.H., H.S., and L.Z. Synthesis of hydrogels was carried out by X.T. and F.Y. Atomic Force Microscopy training and assistance was provided by M.P.W. Drafting and revision of the manuscript was carried out by M.P.M., L.S.K., N.Q., B.L., D.C.W., S.B.G., M.T. Longaker and C.F.K.C. All authors received and approved the final document.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Michael Basson and Brett Benedetti were the primary editors on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Mouse microarray data analysis.
a, Pathways significantly enriched in Ad/MF and P3 mSSC populations. Top, (from left-right) Protein secretion, MYC targets, E2F targets. Bottom, (from left-right) G2M checkpoint, oxidative phosphorylation and TGFb signaling. b-c, mSSC expression (b) and bulk tissue expression (c) of key genes involved in cartilage development, cell cycle and inflammation. d, mSSC gene expression levels after in vitro treatment of PBS, BMP2, or sVEGFR1.
Extended Data Fig. 2 In vitro assays for groups (N, MF).
a, Alizarin red staining of uninjured/normal adult (N) and adult MF mSSCs (left) and mBCSPs (right), and quantification of the alizarin red stain after extraction and photometric measurement at OD450. (n = 3 mice per group). * 0.032; ** 0.0014. b, Alcian blue staining of N and MF mSSCs (left) and mBCSPs (right), and quantification of the Alcian blue stain after extraction and photometric measurement at OD595. (n = 3 mice per group). ** 0.0010; *** 0.0004. c, Oil red O staining of Ad and Ad/MF mSSCs (Left) and mBCSPs (Right). Scale bar 100μm. (n = 3 mice per group). Graphs show mean + /- s.e.m. Two-tailed Student’s t-test. Exact P values to 2 significant figures.
Extended Data Fig. 3 Microdissection before and after MF surgery.
a, Schematic depicting the distal femur of a mouse and its major tissue layers before and after MF surgery. b, Representative flow cytometry analysis of the articular surface (top), the periosteum (middle), and bone (bottom) with relative populations of mOP, mCP, mBCSP, and mSSC. c, Quantification of mSSC/million events in bone, cartilage, and periosteum. Graph shows mean + /- s.e.m. Ordinary one-way ANOVA test with post-hoc analysis using Šídák method to compare between specific means. Exact P values to 2 significant figures. (n = 5 mice per group). d, Quantification of mSSC/million events comparing MF joints and joints with articular cartilage and periosteum removed prior to MF surgery (mMF). (n = 5 mice per group). e, Alcian blue staining of mSSC following chondrogenic differentiation with treatment of PBS or BMP2 + sVEGFR1. Scale bars 100μm. Representative of three independent experiments. f, Quantification of the Alcian blue stain after extraction and photometric measurement at OD595. Unless otherwise stated, graphs show mean + /- s.e.m. Two-tailed Student’s t-test. Exact P values to 2 significant figures.
Extended Data Fig. 4 OA progression.
a, Schematic of the experimental outline. Destabilization of the medial meniscus (DMM) was performed on adult mice, and the distal femurs were harvested after 8 weeks. Analysis was performed by IF, pentachrome, and Safranin-O Fast Green staining. b, Left, representative IF of indicated markers in sham and DMM joints. Scale bars 100μm. Right, Safranin-O Fast Green and pentachrome stains of sham and joints 8 weeks after DMM. Scale bars 500μm. (n = 3 per group). c, Quantification of IF stains in sham versus DMM. Graphs show mean + /- s.e.m. Two-tailed Student’s t-test. Exact P values to 2 significant figures. (n = 3 per group).
Extended Data Fig. 5 IHC and quantification of OA/MF with factors at 4 weeks.
a, Representative IF of (from left-right) ACAN, COL 1, COL 2, COL 10 and MMP 13 in 3 treatment conditions (top: PBS, middle: BMP2, bottom: BMP2 + sVEGFR1) at 4 weeks after MF. Scale bars 250μm. (n = 8 per group). b, Quantification of IF stains for indicated markers of MF + factors at 4 weeks. Graphs show mean + /- s.e.m. Ordinary one-way ANOVA test with post-hoc analysis using Šídák method to compare between specific means. Exact P values to 2 significant figures. c, Representative IF isotype and positive controls. (n = 3 per group). d, Repeat examples of pentachrome staining of BMP2 + sVEGFR1 sections at low (scale bars 500μm) and high (scale bars 100μm) magnification. Representatives of eight independent experiments.
Extended Data Figure 6 Histology of OA/MF at 2 weeks and 8 weeks.
a, Representative pentachrome stains and respective IF of (from left-right) ACAN, COL 1, COL 2, COL 10 and MMP 13 in 3 treatment conditions (top: PBS; middle: BMP2; bottom: BMP2 + sVEGFR1) at 2 weeks after MF. Scale bars 500μm. (n = 4 per group). b, Representative pentachrome stains and respective IF stains for the indicated markers in 3 treatment conditions (top row: PBS; middle: BMP2; bottom: BMP2 + sVEGFR1) at 8 weeks after MF. Scale bars 500μm. (n = 4 per group). c, Quantification of IF stains for the indicated markers at 2 weeks (top) and 8 weeks (bottom) after MF. (n = 3 per group per timepoint). Graphs show mean + /- s.e.m. Ordinary one-way ANOVA test with post-hoc analysis using Šídák method to compare between specific means. Exact P values to 2 significant figures.
Extended Data Fig. 7 Week 16 timepoint.
a, Representative pentachrome and respective IF of (from left-right) ACAN, COL 1, COL 2, COL 10 and MMP 13 in 3 treatment conditions (top: PBS, middle: BMP2, bottom: BMP2 + sVEGFR1). Scale bars 500μm. (n = 4 per group). b, Quantification of IF stains for indicated markers of MF + factors. Graphs show Mean + /- SEM. Ordinary one-way ANOVA test with post-hoc analysis using Šídák method to compare between specific means. Exact P values to 2 significant figures. (n = 3 per group). c, From left-right, gross images of distal femur (scale bars 1 mm); respective pentachrome (scale bars 500μm); 3D Peak Force Error; 3D Deformation; Force Volume in 4 conditions (PBS, BMP2, BMP2 + sVEGFR1, Uninjured). (n = 4 per group).
Extended Data Fig. 8 Effects of aged and irradiated niches.
a, Quantification of mSSC/million events in normal and MF joints of adult (9wks) and aged (1 yr) mice. Graph shows mean + /- s.e.m. Two-tailed Student’s t-test. Exact P values to 2 significant figures. (n = 10 mice per group). b, Representative pentachrome stains of 2-weeks post MF surgery in aged joints with (from left-right) PBS, BMP2, and BMP2 + sVEGFR1. Scale bar 500μm. (n = 3 per group). c, Schematic of the experimental outline. Microfracture surgery is performed on the joints of GFP + adult mice (Ad/MF). GFP + mSSCs are sorted by flow cytometry and cotransplanted with BMP2 + sVEGFR1 into MF defects of irradiated mice. Analysis is performed by brightfield microscopy (BF), immunofluorescence (IF), and pentachrome staining. d, Top, Representative BF (left) and fluorescence microscopy (right) of GFP + Ad/MF mSSC 4 weeks after they were transplanted into the MF defect of an irradiated joint. Scale bars 500μm. (n = 5 mice). Middle, Representative pentachrome stain (scale bar 500μm) shown with higher magnification (scale bar 100μm) and respective IF of GFP + Ad/MF mSSC 4 weeks after they were transplanted into the MF defect of an irradiated joint. Positive staining marked by white arrows. Representatives of three separate experiments. Bottom right, a negative control IF stain was performed for each experiment. Scale bars 500μm.
Extended Data Fig. 9 Atomic Force Microscopy at 8 weeks post MF.
a, Left and middle, schematic of Rheology experimental outline. Right, Quantification for the modulus of the regenerate (PBS, BMP2, BMP2 + sVEGFR1.) Graph shows mean + /- s.e.m. Ordinary one-way ANOVA test (p = <0.0001) with post-hoc analysis using Šídák method to compare between specific means. Exact P values to 2 significant figures. (n = 10 mice per group). b, From left-right, gross images of distal femur (scale bars 1 mm); respective pentachrome (scale bars 500μm); 3D Peak Force Error; 3D Deformation; Force Volume in 4 conditions (PBS, BMP2, BMP2 + sVEGFR1, Uninjured) (n = 4 per group). Scale bar 500μm. c, Force graphs for PBS, BMP2, BMP2 + sVEGFR1, and uninjured. (n = 4 per group).
Extended Data Fig. 10 Functional assessment.
a, Schematic of the gait analysis performed on Ad/MF mice. b, Grimace assessment scores for adult mice following MF surgery of the distal femur with 3 treatment conditions (PBS, BMP2, BMP2 + sVEGFR1). Graphs show mean + /- s.e.m. Ordinary one-way ANOVA test (p = <0.0001) with post-hoc analysis using Šídák method to compare between specific means. Exact P values to 2 significant figures. (n = 5 mice per group). c, Gait assessment scores for maximum contact mean intensity with 4 conditions (PBS, BMP2, BMP2 + sVEGFR1, and Uninjured) at 4, 8, and 16 weeks after surgery. (n = 3 mice per group per timepoint).
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Murphy, M.P., Koepke, L.S., Lopez, M.T. et al. Articular cartilage regeneration by activated skeletal stem cells. Nat Med 26, 1583–1592 (2020). https://doi.org/10.1038/s41591-020-1013-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-020-1013-2
This article is cited by
-
HWJMSC-EVs promote cartilage regeneration and repair via the ITGB1/TGF-β/Smad2/3 axis mediated by microfractures
Journal of Nanobiotechnology (2024)
-
Identification of therapeutic targets in osteoarthritis by combining heterogeneous transcriptional datasets, drug-induced expression profiles, and known drug-target interactions
Journal of Translational Medicine (2024)
-
Profiling joint tissues at single-cell resolution: advances and insights
Nature Reviews Rheumatology (2024)
-
Intra-articular injection of ascorbic acid enhances microfracture-mediated cartilage repair
Scientific Reports (2024)
-
2‐year results of middle‐aged patients with two‐compartment cartilage lesions in one knee treated with two patient specific metal implants
Journal of Experimental Orthopaedics (2023)