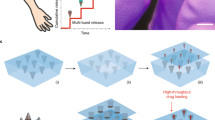
Abstract
Drug delivery to the skin is highly constrained by the stratum corneum barrier layer1. Here, we developed star-shaped particles, termed STAR particles, to dramatically increase skin permeability. STAR particles are millimeter-scale particles made of aluminum oxide or stainless steel with micron-scale projections designed to create microscopic pores across the stratum corneum. After gentle topical application for 10 s to porcine skin ex vivo, delivery of dermatological drugs and macromolecules, including those that cannot be given topically, was increased by 1 to 2 orders of magnitude. In mice treated with topical 5-fluorouracil, use of STAR particles increased the efficacy of the drug in suppressing the growth of subcutaneous melanoma tumors and prolonging survival. Moreover, topical delivery of tetanus toxoid vaccine to mice using STAR particles generated immune responses that were at least as strong as delivery of the vaccine by intramuscular injection, albeit at a higher dose for topical than intramuscular vaccine administration. STAR particles were well tolerated and effective at creating micropores when applied to the skin of human participants. Use of STAR particles provides a simple, low-cost and well-tolerated method for increasing drug and vaccine delivery to the skin and could widen the range of compounds that can be topically administered.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available from the corresponding author upon written request.
References
Bolognia, J. L., Schaffer, J. V. & Cerroni, L. (eds) Dermatology 4th edn (Elsevier, 2018).
Lim, H. W. et al. The burden of skin disease in the United States. J. Am. Acad. Dermatol. 76, 958–972.e2 (2017).
Hay, R. J. et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J. Invest. Dermatol. 134, 1527–1534 (2014).
Moustafa, F., Lewallen, R. S. & Feldman, S. R. The psychological impact of rosacea and the influence of current management options. J. Am. Acad. Dermatol. 71, 973–980 (2014).
Cazzaniga, S. et al. Medical, psychological and socio‐economic implications of chronic hand eczema: a cross‐sectional study. J. Eur. Acad. Dermatol. Venereol. 30, 628–637 (2016).
Watkinson, A. C., Kearney, M.-C., Quinn, H. L., Courtenay, A. J. & Donnelly, R. F. Future of the transdermal drug delivery market—have we barely touched the surface? Expert Opin. Drug Deliv. 13, 523–532 (2016).
Prausnitz, M. R. & Langer, R. Transdermal drug delivery. Nat. Biotechnol. 26, 1261–1268 (2008).
Pastore, M. N., Kalia, Y. N., Horstmann, M. & Roberts, M. S. Transdermal patches: history, development and pharmacology. Br. J. Pharmacol. 172, 2179–2209 (2015).
Choy, Y. B. & Prausnitz, M. R. The rule of five for non-oral routes of drug delivery: ophthalmic, inhalation and transdermal. Pharm. Res. 28, 943–948 (2011).
Karande, P., Jain, A., Ergun, K., Kispersky, V. & Mitragotri, S. Design principles of chemical penetration enhancers for transdermal drug delivery. Proc. Natl Acad. Sci. USA 102, 4688–4693 (2005).
Arora, A., Prausnitz, M. R. & Mitragotri, S. Micro-scale devices for transdermal drug delivery. Int. J. Pharm. 364, 227–236 (2008).
Prausnitz, M. R. Engineering microneedle patches for vaccination and drug delivery to skin. Annu. Rev. Chem. Biomol. Eng. 8, 177–200 (2017).
Ye, Y., Yu, J., Wen, D., Kahkoska, A. R. & Gu, Z. Polymeric microneedles for transdermal protein delivery. Adv. Drug Deliv. Rev. 127, 106–118 (2018).
Nguyen, T. T. & Park, J. H. Human studies with microneedles for evaluation of their efficacy and safety. Expert Opin. Drug Deliv. 15, 235–245 (2018).
Ripolin, A. et al. Successful application of large microneedle patches by human volunteers. Int. J. Pharm. 521, 92–101 (2017).
Rademaker, M. Adverse effects of isotretinoin: a retrospective review of 1743 patients started on isotretinoin. Australas. J. Dermatol. 51, 248–253 (2010).
Wollina, U., Ständer, K. & Barta, U. Toxicity of methotrexate treatment in psoriasis and psoriatic arthritis—short- and long-term toxicity in 104 patients. Clin. Rheumatol. 20, 406–410 (2001).
Beyer, V. & Wolverton, S. E. Recent trends in systemic psoriasis treatment costs. Arch. Dermatol. 146, 46–54 (2010).
Good, L. M., Miller, M. D. & High, W. A. Intralesional agents in the management of cutaneous malignancy: a review. J. Am. Acad. Dermatol. 64, 413–422 (2011).
Horn, T. D., Johnson, S. M., Helm, R. M. & Roberson, P. K. Intralesional immunotherapy of warts with mumps, Candida, and Trichophyton skin test antigens: a single-blinded, randomized, and controlled trial. Arch. Dermatol. 141, 589–594 (2005).
Gill, H. S., Denson, D. D., Burris, B. A. & Prausnitz, M. R. Effect of microneedle design on pain in human subjects. Clin. J. Pain 24, 585–594 (2008).
Gomaa, Y. A. et al. Effects of microneedle length, density, insertion time and multiple applications on human skin barrier function: assessments by transepidermal water loss. Toxicol. In Vitro 24, 1971–1978 (2010).
Edens, C., Dybdahl-Sissoko, N. C., Weldon, W. C., Oberste, M. S. & Prausnitz, M. R. Inactivated polio vaccination using a microneedle patch is immunogenic in the rhesus macaque. Vaccine 33, 4683–4690 (2015).
Standard Specification for High-Purity Dense Aluminum Oxide for Medical Application (ASTM, 2012).
Inactive Ingredient Search for Approved Drug Products https://www.accessdata.fda.gov/scripts/cder/iig/index.cfm (US Food & Drug Administration, 2019).
Becker, L. C. et al. Safety assessment of alumina and aluminum hydroxide as used in cosmetics. Int. J. Toxicol. 35, 16S–33S (2016).
Gross, K., Kircik, L. & Kricorian, G. 5% 5‐fluorouracil cream for the treatment of small superficial basal cell carcinoma: efficacy, tolerability, cosmetic outcome, and patient satisfaction. Dermatol. Surg. 33, 433–439 (2007).
Taudorf, E. H. et al. Topically applied methotrexate is rapidly delivered into skin by fractional laser ablation. Expert Opin. Drug Deliv. 12, 1059–1069 (2015).
Lee, H. S., Ryu, H. R., Roh, J. Y. & Park, J.-H. Bleomycin-coated microneedles for treatment of warts. Pharm. Res. 34, 101–112 (2017).
Siddhapura, K., Harde, H. & Jain, S. Immunostimulatory effect of tetanus toxoid loaded chitosan nanoparticles following microneedles assisted immunization. Nanomedicine 12, 213–222 (2016).
Gupta, P. N. et al. Tetanus toxoid‐loaded transfersomes for topical immunization. J. Pharm. Pharmacol. 57, 295–301 (2005).
McKay, K. M. et al. Thickness of superficial basal cell carcinoma (sBCC) predicts imiquimod efficacy: a proposal for a thickness‐based definition of sBCC. Br. J. Dermatol. 169, 549–554 (2013).
Kirby, J. S. & Miller, C. J. Intralesional chemotherapy for nonmelanoma skin cancer: a practical review. J. Am. Acad. Dermatol. 63, 689–702 (2010).
Eskicirak, B., Zemheri, E. & Cerkezoglu, A. The treatment of psoriasis vulgaris: 1% topical methotrexate gel. Int. J. Dermatol. 45, 965–969 (2006).
Hutin, Y. J. F., Hauri, A. M. & Armstrong, G. L. Use of injections in healthcare settings worldwide, 2000: literature review and regional estimates. BMJ 327, 1075 (2003).
Alster, T. S. & Graham, P. M. Microneedling: a review and practical guide. Dermatol. Surg. 44, 397–404 (2018).
Cary, J. H., Li, B. S. & Maibach, H. I. Dermatotoxicology of microneedles (MNs) in man. Biomed. Microdevices 21, 66 (2019).
Riedel, R. & Chen, I.-W. Ceramics Science and Technology (WILEY-VCH Verlag, 2008).
Upadhyaya, A. & Upadhyaya, G. S. Powder Metallurgy: Science, Technology, and Materials (Universities Press, 2011).
Guidance Document for the Conduct of Skin Absorption Studies (OECD, 2004).
Prausnitz, M. R., .Bose, V. G., Langer, R. & Weaver, J. C. Electroporation of mammalian skin: a mechanism to enhance transdermal drug delivery. Proc. Natl Acad. Sci. USA 90, 10504–10508 (1993).
Acknowledgements
The authors thank D. McAllister, S. Henry and W. Pewin for helpful discussions; R. Schafer for his expertise and assistance with laser micromachining; and D. Bondy for administrative support. This work was supported financially by the Georgia Research Alliance (no. GRA.VL17.B18, M.R.P.), the CDC Foundation with funding that originated from UNICEF (no. 969-17SC.GTRC, M.R.P.), the National Institutes of Health Director’s New Innovator Award (no. DP2HD091793, G.A.K.) and a Career Award at the Scientific Interface from the Burroughs Wellcome Fund (G.A.K.).
Author information
Authors and Affiliations
Contributions
A.R.T. and A.S. carried out the ex vivo transdermal diffusion studies. A.R.T., A.R., I.C.M., R.K.N. and L.O. carried out the in vivo studies. A.R.T. carried out the human studies and manufactured STAR particles. A.R.T., A.R., I.C.M., G.A.K. and M.R.P. designed the studies and conducted the data analysis. A.R.T. and M.R.P. wrote the manuscript. G.A.K. and M.R.P. supervised the project.
Corresponding author
Ethics declarations
Competing interests
A.R.T. and M.R.P. are inventors of patent applications (no. WO2017/151745Al) and have a financial interest in a company developing STAR particle-based products (that is, Microstar Biotech). This potential conflict of interest has been disclosed and is overseen by the Georgia Institute of Technology.
Additional information
Peer review information Michael Basson was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 mSTAR particles were engineered to have different geometries.
Representative brightfield microscopy images showing mSTAR particles with two arms (a), four arms (b) or six arms (c) imaged by brightfield microscopy. Images are representative of n = 4 replicates.
Extended Data Fig. 2 Skin abrasion results in tissue removal rather than puncture.
Images showing representative en face gentian violet stained porcine skin ex vivo following treatment with aloe vera gel (a), EKG abrasive gel (b), or P60 grit sandpaper (c) for 10 s. Representative brightfield microscopy image showing EKG abrasive crystals at magnification (d). Images are representative of n = 4 replicates.
Extended Data Fig. 3 mSTAR particles increased gentian violet staining of skin, which indicates disruption of the stratum corneum barrier on the surface of skin.
Representative en face images of gentian violet-stained skin following treatment with six-armed mSTAR particles at 0.5 wt% (a), 2.7 wt% (b), 5.4 wt% (c) or 10.8 wt% (d) mSTAR particles in 0.5 g gel applied to ~50 cm2 porcine skin. Results showing quantification of gentian violet skin staining as a percent of total surface area following treatment with mSTAR particles ex vivo (e). Data show mean ± S.E.M. for n = 4 replicates from distinct samples. Statistical significance of differences in gentian violet staining is shown in comparison to aloe vera gel treatment: ****p < 0.0001. Statistical comparisons based on two-way ANOVA. Effects of mSTAR particle application on skin permeability ex vivo (f). Transdermal flux of 1 mM FITC-labeled dextrans (4 kDa, 20 kDa) across porcine skin ex vivo following treatment with mSTAR particles (containing six arms at a concentration of 5.4 wt% mSTAR particles in 0.5 g gel applied to ~50 cm2 porcine skin). Data show mean ± standard deviation for n = 3 replicates from distinct samples. Transdermal flux of both dextrans was below the detection limit of 0.175 nmol/cm2 for skin without mSTAR particle treatment.
Extended Data Fig. 4 Additional characterization of mSTAR particles application to skin.
Effect of removing mSTAR particles and of microneedle patches on skin permeability. Quantification of steady-state transdermal flux (a) across porcine skin (~50 cm2) treated ex vivo with 5.4 wt% six-armed mSTAR particles in 0.5 g gel that were either left in place (‘leave on’) or removed (‘wipe away’) following their topical application and before flux measurements. Quantification of steady-state transdermal flux (b) across porcine skin (~50 cm2) treated ex vivo with 5.4 wt% six-armed mSTAR particles in 0.5 g gel or with a microneedle patch (0.36 cm2) containing 100 microneedles each measuring ~650 μm long, 300 μm in diameter at the base and tapering to a tip with <10 μm radius of curvature. After application, STAR particles and microneedle patches were removed before flux measurements. Data show mean ± standard deviation for n = 4 replicates from distinct samples. Representative image of mSTAR particles after application to porcine skin ex vivo for 10 s, which shows how mSTAR particles aggregate after use (c, d).
Extended Data Fig. 5 cSTAR particles were fabricated with different numbers of arms.
Tip radius of curvature shown for cSTAR particles (a). Data show median ± 25% in boxes, and 95% confidence interval error bars for n = 63, n = 71, n = 120 and n = 144 replicates from distinct samples of three, four, six and nine-arm cSTARs, respectively. Representative scanning electron micrographs showing cSTAR particles with three (b), four (c), six (d) or nine (e) arms. Images are representative of n = 10 replicates.
Extended Data Fig. 6 Ceramic STAR particles are mechanically robust and do not break during skin application.
Representative images of aluminum oxide STAR particles before (a1, a2) and after (b1, b2) application to porcine skin ex vivo. Images are representative of n = 4 replicates. Quantification of mean fluorescence intensity of images in Fig. 3e–g presented in arbitrary units per square centimeter (A.U./cm2) of sulforhodamine B delivered into hairless rat skin for 3 h or 15 min in vivo following treatment with gel or mSTAR particles (c). In these studies, 5.4 wt% six-arm mSTAR particles in 0.5 g gel were rubbed onto ~50 cm2 skin on the dorsum for 10 s. Data show average ± S.E.M. (n = 4 replicates from distinct samples). Statistical comparison based on Student’s t-test.
Extended Data Fig. 7 Visual comparison of treatments used in assessment of mSTAR particles in human participants.
Brightfield microscopy images shown of a 26 gauge hypodermic needle (a), 0.5 mm stainless steel disks (b) or six-armed mSTAR particles (c). Images shown at the same scale. Images are representative of n = 4 replicates.
Extended Data Fig. 8 mSTAR particle skin application was well tolerated by human participants and generally induced a comfortable sensory feedback for study participants.
Results from human studies. Tolerability scores (see Table M4 in Online Materials) for skin of human participants (n = 11) (a) and sensations, and sensation comfort, reported by study participants (n = 11) (b) after application of a 26 gauge hypodermic needle inserted 0.5 cm into the skin, aloe vera gel, 2.5 wt% 0.5 mm diameter circular stainless steel disks in 0.5 g gel or 2.7 wt% six-armed mSTAR particles in 0.5 g gel applied to 25 cm2 skin.
Extended Data Fig. 9 mSTAR particle skin application perceived by study participants to be intuitive and similar to conventional skin products.
Evaluation of future self-administration of STAR particles by study participants (n = 11) provided at the end of the study. Subjects were asked the questions indicated and provided answers among five multiple-choice options indicated.
Extended Data Fig. 10 STAR particles must be intentionally rubbed on skin to function.
Representative images of pig skin ex vivo after treatment with STAR particles. Skin was stained with Gentian violet to identify sites of skin puncture after no treatment (a), firmly pressing cSTAR particles against the skin 10 times over the course of 10 s (b) or firmly rubbing cSTAR particles on the skin for 10 s (c). Images are representative of n = 4 distinct replicates.
Supplementary information
Supplementary Information
Supplementary Tables 1–4.
Rights and permissions
About this article
Cite this article
Tadros, A.R., Romanyuk, A., Miller, I.C. et al. STAR particles for enhanced topical drug and vaccine delivery. Nat Med 26, 341–347 (2020). https://doi.org/10.1038/s41591-020-0787-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-020-0787-6
This article is cited by
-
Non-invasive transdermal delivery of biomacromolecules with fluorocarbon-modified chitosan for melanoma immunotherapy and viral vaccines
Nature Communications (2024)
-
Rational design of a JAK1-selective siRNA inhibitor for the modulation of autoimmunity in the skin
Nature Communications (2023)
-
Improved pharmacodynamics of epidermal growth factor via microneedles-based self-powered transcutaneous electrical stimulation
Nature Communications (2022)
-
Robotics, microfluidics, nanotechnology and AI in the synthesis and evaluation of liposomes and polymeric drug delivery systems
Drug Delivery and Translational Research (2021)