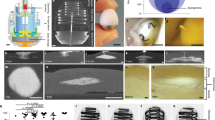
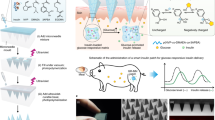
Abstract
Insulin and other injectable biologic drugs have transformed the treatment of patients suffering from diabetes1,2, yet patients and healthcare providers often prefer to use and prescribe less effective orally dosed medications3,4,5. Compared with subcutaneously administered drugs, oral formulations create less patient discomfort4, show greater chemical stability at high temperatures6, and do not generate biohazardous needle waste7. An oral dosage form for biologic medications is ideal; however, macromolecule drugs are not readily absorbed into the bloodstream through the gastrointestinal tract8. We developed an ingestible capsule, termed the luminal unfolding microneedle injector, which allows for the oral delivery of biologic drugs by rapidly propelling dissolvable drug-loaded microneedles into intestinal tissue using a set of unfolding arms. During ex vivo human and in vivo swine studies, the device consistently delivered the microneedles to the tissue without causing complete thickness perforations. Using insulin as a model drug, we showed that, when actuated, the luminal unfolding microneedle injector provided a faster pharmacokinetic uptake profile and a systemic uptake >10% of that of a subcutaneous injection over a 4-h sampling period. With the ability to load a multitude of microneedle formulations, the device can serve as a platform to orally deliver therapeutic doses of macromolecule drugs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information files.
References
Turner, R. C., Cull, C. A., Frighi, V. & Holman, R. R. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). J. Am. Med. Assoc. 281, 2005–2012 (1999).
Pratley, R. E. et al. Liraglutide versus sitagliptin for patients with type 2 diabetes who did not have adequate glycaemic control with metformin: a 26-week, randomised, parallel-group, open-label trial. Lancet 375, 1447–1456 (2010).
Korytkowski, M. When oral agents fail: practical barriers to starting insulin. Int. J. Obes. 26, S18–S24 (2002).
Boye, K. S. et al. Utilities and disutilities for attributes of injectable treatments for type 2 diabetes. Eur. J. Heal. Econ. 12, 219–230 (2011).
Calvert, M. J., McManus, R. J. & Freemantle, N. Management of type 2 diabetes with multiple oral hypoglycaemic agents or insulin in primary care: retrospective cohort study. Br. J. Gen. Pract. 57, 455–460 (2007).
Cleland, J. & Langer, R. in Formulation and Delivery of Proteins and Peptides (eds. Cleland, J. L. & Langer, R.) 1–19 (American Chemical Society, 1994).
Dallel, N., Kacem, M., Nabouli, R. M. & El May, M. Disposal of insulin syringes by diabetic patients. Report of 100 patients.Tunis. Med. 83, 390–392 (2005).
Anselmo, A. C., Gokarn, Y. & Mitragotri, S. Non-invasive delivery strategies for biologics. Nat. Rev. Drug Discov. 18, 19–40 (2018).
Harrison, G. A. Insulin in alcoholic solution by the mouth. Br. Med. J. 2, 1204–1205 (1923).
Abramson, A. et al. An ingestible self-orienting system for oral delivery of macromolecules. Science 363, 611–615 (2019).
Traverso, G. et al. Microneedles for drug delivery via the gastrointestinal tract. J. Pharm. Sci. 104, 362–367 (2015).
Scudellari, M. Shot to the Gut: “Robotic” Pill Sails Through Human Safety Study. https://spectrum.ieee.org/the-human-os/biomedical/devices/shot-to-the-gut-robotic-pill-sails-through-human-safety-study (accessed 25 September 2019).
Vllasaliu, D., Thanou, M., Stolnik, S. & Fowler, R. Recent advances in oral delivery of biologics: nanomedicine and physical modes of delivery. Expert Opin. Drug Deliv. 15, 759–770 (2018).
Davies, M. et al. Effect of oral semaglutide compared with placebo and subcutaneous semaglutide on glycemic control in patients with type 2 diabetes. J. Am. Med. Assoc. 318, 1460–1470 (2017).
Banerjee, A. et al. Ionic liquids for oral insulin delivery. Proc. Natl Acad. Sci. USA 115, 7296–7301 (2018).
Mathiowitz, E. et al. Biologically erodable microspheres as potential oral drug delivery systems. Nature 386, 410–414 (1997).
Pridgen, E. M., Alexis, F. & Farokhzad, O. C. Polymeric nanoparticle drug delivery technologies for oral delivery applications. Expert Opin. Drug Deliv. 12, 1459–1473 (2015).
Banerjee, A., Wong, J., Gogoi, R., Brown, T. & Mitragotri, S. Intestinal micropatches for oral insulin delivery. J. Drug Target. 25, 608–615 (2017).
Mitragotri, S., Burke, P. A. & Langer, R. Overcoming the challenges in administering biopharmaceuticals: formulation and delivery strategies. Nat. Rev. Drug Discov. 13, 655–672 (2014).
Bass, D. M., Prevo, M. & Waxman, D. S. Gastrointestinal safety of an extended-release, nondeformable, oral dosage form (OROS): a retrospective study. Drug Saf. 25, 1021–1033 (2002).
Coffey, J. W., Meliga, S. C., Corrie, S. R. & Kendall, M. A. F. Dynamic application of microprojection arrays to skin induces circulating protein extravasation for enhanced biomarker capture and detection. Biomaterials 84, 130–143 (2016).
Gatto, N. M. et al. Risk of perforation after colonoscopy and sigmoidoscopy: a population-based study. J. Natl Cancer Inst. 95, 230–236 (2003).
Podolsky, D. K. Healing the epithelium: solving the problem from two sides. J. Gastroenterol. 32, 122–126 (1997).
Classen, M., Tytgat, G. & Lightdale, C. Gastroenterological Endoscopy (Thieme, 2010).
Velitchkov, N. G., Grigorov, G. I., Losanoff, J. E. & Kjossev, K. T. Ingested foreign bodies of the gastrointestinal tract: retrospective analysis of 542 cases. World J. Surg. 20, 1001–1005 (1996).
Sandler, R. S., Stewart, W. F., Liberman, J. N., Ricci, J. A. & Zorich, N. L. Abdominal pain, bloating, and diarrhea in the United States. Dig. Dis. Sci. 45, 1166–1171 (2000).
Bass, D. M., Prevo, M. & Waxman, D. S. Gastrointestinal safety of an extended-release, nondeformable, oral dosage form (OROS: a retrospective study). Drug Saf. 25, 1021–1033 (2002).
Iddan, G., Meron, G., Glukhovsky, A. & Swain, P. Wireless capsule endoscopy. Nature 405, 417 (2000).
Hellmig, S. et al. Gastric emptying time of fluids and solids in healthy subjects determined by 13C breath tests: influence of age, sex and body mass index. J. Gastroenterol. Hepatol. 21, 1832–1838 (2006).
Wang, Y. R., Fisher, R. S. & Parkman, H. P. Gastroparesis-related hospitalizations in the United States: trends, characteristics, and outcomes, 1995–2004. Am. J. Gastroenterol. 103, 313–322 (2008).
Snoeck, V. et al. Gastrointestinal transit time of nondisintegrating radio-opaque pellets in suckling and recently weaned piglets. J. Control. Release 94, 143–153 (2004).
Cole, E. T. et al. Enteric coated HPMC capsules designed to achieve intestinal targeting. Int. J. Pharm. 231, 83–95 (2002).
Ye, Y., Yu, J., Wen, D. & Kahkoska, A. R. Polymeric microneedles for transdermal protein delivery. Adv. Drug Deliv. Rev. 127, 106–118 (2018).
Van Meer, L. et al. Injection site reactions after subcutaneous oligonucleotide therapy. Br. J. Clin. Pharmacol. 82, 340–351 (2016).
Fallingborg, J. Intraluminal pH of the human gastrointestinal tract. Dan. Med. Bull. 46, 183–196 (1999).
Acknowledgements
We thank S. McDonnell, K. Ishida, J. Haupt and M. Jamiel for help with the in vivo porcine work. We thank C. Steiger and Y. L. Kong for helpful conversations on the capsule system. We thank J. Bales and the Edgerton Center at MIT for providing a high-speed camera setup. We thank A. Hupalowska for illustrations of the LUMI concept. We thank the Koch Institute for Integrative Cancer Research at MIT’s Histology Core, High Throughput Facility and Imaging Core. We thank the National Disease Research Interchange for help with the procurement of human cadaveric tissue. We thank U. Stilz, M. Bielecki, T. Kjeldsen, L. F. Iversen, J. Trosborg, P. Herskind, R. K. Kirk, F. Hubálek, J. J. Water, A. V. Friderichsen, S. Buckley, A. Vegge and T. B. Pedersen for helpful discussions throughout the development of the LUMI. We are grateful to R. Yona and all of the other members of the Langer and Traverso laboratories and Novo Nordisk for their expertise and discussions around biologic drug delivery. This work was funded in part by a grant from Novo Nordisk and NIH grant number EB000244. A.A. was supported in part by the NSF Graduate Research Fellowship Program fellowship. G.T. was supported in part by the Division of Gastroenterology at Brigham and Woman’s Hospital and the Department of Mechanical Engineering at MIT. D.D. was supported by the MIT Undergraduate Research Opportunities Program. N.R. was supported in part by the Viking Olof Björk scholarship trust. H.-C.L., T.Y. and J. Fujimoto were supported in part by NIH R01-CA075289-21.
Author information
Authors and Affiliations
Contributions
A.A. and E.C.-S. designed the study, performed the experiments, analyzed and interpreted the data, and wrote the manuscript. V.S., D.M., X.L., R.Y.T., D.D., Y.G., S.K., J.W., J. Fels, M.R.F. and N.R. designed, fabricated and evaluated the prototypes of the system. J.C., S.T. and A.H., designed, performed and helped to analyze the in vivo evaluation of the system. T.Y., H.-C.L. and J. Fujimoto designed, performed and interpreted the characterization of the prototype tissue interactions. U.R., R.L. and G.T. designed, supervised and reviewed the data, and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A.A., E.C.-S., D.D., N.R., M.R.F., Y.G., R.L. and G.T. are co-inventors on multiple patent applications describing oral biologic drug delivery. J. Fels, M.R.F. and U.R. work for Novo Nordisk. A.A., R.L. and G.T. report having received consulting fees from Novo Nordisk. A.A. reports having received consulting fees from Eli Lilly. Complete details of all relationships for profit and not for profit for G.T. can found at https://www.dropbox.com/sh/szi7vnr4a2ajb56/AABs5N5i0q9AfT1IqIJAE-T5a?dl=0. For a list of entities with which R.L. is involved (compensated or uncompensated), see https://www.dropbox.com/s/yc3xqb5s8s94v7x/Rev%20Langer%20COI.pdf?dl=0.
Additional information
Peer review information Brett Benedetti was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Luminal Unfolding Microneedle Injector (LUMI) schematic timeline.
LUMI devices were administered in enteric capsules. They actuated and unfolded in the small intestine, injecting drug loaded microneedles into the tissue wall. The microneedle patches and arms dissolved within several hours. The non-degradable parts of the device passed through the gastrointestinal tract and were excreted.
Extended Data Fig. 2 LUMI Fabrication Scheme and Generated Contact Forces.
a, Custom fabricated PDMS mold for creation of LUMI backbone. Metal cores were embedded in the elastomer during heating. b, Force applied to a steel compression platen by LUMI arm over time after actuation. (n = 1 representative device replicate). Scatter plots of the data are shown in Fig. 2f. Scale bars = 1 cm.
Extended Data Fig. 3 Prototype watertight capsule demonstration in water.
The LUMI is protected from external liquid until immediately before it unfolds. Before the actuation occurs, the PEG must dissolve over a period of 1–5 h. Scale bars = 1 cm.
Extended Data Fig. 4 In vitro and in vivo LUMI dissolution.
a, Bar (3.2 × 12.8 × 63.5 mm) and arm (3.6 mm equilateral triangle side length, 18.6 mm length) shapes used for flexural strength and dissolution testing respectively. Dissolution of arm shapes over time in vitro at 37 °C in simulated intestinal fluid is shown. Scale bars = 5 cm. b, A LUMI device is delivered to the small intestine in an enteric capsule and degrades over time. Stainless steel ball bearings 1 mm in diameter are placed on the arms to aid in visualization. The metal beads served as radio-opaque fiducials and were not part of the final design. Scale bars = 5 cm.
Extended Data Fig. 5 Penetration characterization for small intestine tissue.
Forces required for needle displacement in (a) ex vivo human and (b) in vivo swine small intestine tissue, respectively. (c) A comparison between human and swine forces in the small intestine using 32 G needles. (Ex vivo tissue: n = 4–5 technical replicates each for 2 animal/patient replicates totaling n = 9–10; In vivo tissue: n = 5 technical replicates each for 3 animal replicates totaling n = 15. Error bars = SD. Center dot = mean.)
Extended Data Fig. 6 LUMI deployment with hypodermic needle.
a, Colored MicroCT reconstruction. Scale bar = 10 mm. b, Needle is same height as microneedles. Scale bar = 1 mm. c, MicroCT cross section of LUMI deployment. Tissue is outlined. Scale bar = 5 mm.
Extended Data Fig. 7 Optical coherence tomography (OCT) images of microneedle penetration.
a, b, OCT imaging showing (a) solid microneedles (Scale Bars = 1 mm) and (b) dissolving microneedle patches in ex vivo swine tissue (Scale bars = 0.5 mm). c, Fixture used to deploy LUMI during OCT imaging. Tissue holder dimensions are: 65 mm L x 140 mm W x 106 mm H. LUMI actuator dimensions are 67 mm L x 155 mm W x 93 mm H. d, e, OCT images showing the microneedles mounted in the LUMI arm (d) prior to insertion and (e) inserted into small intestine after deploying the arm from a 30 degree angle (Scale bars = 0.5 mm). Arrows in e point to the holes observed in the tissue corresponding to microneedles being inserted. The differences in the holes’ size reflect the different penetration depths from tilted insertion. Animations to the right denote the angle of the imager compared to the microneedle patch on the LUMI arm.
Extended Data Fig. 8 LUMI devices made with various excipients and biomacromolecules.
LUMI arms with microneedle patches made with different formulations and active pharmaceutical ingredient are pictured. Aside from the sorbitol patch, all patches use polyvinylpyrrolidone (PVP) as a binding agent. Activity of lysozyme and alpha-glucosidase in microneedle formulations are shown. Scale bars = 1 mm. (n = 5 device replicates. Error Bars = SD. Center line = mean.)
Extended Data Fig. 9 In vitro and in vivo microneedle dissolution and macromolecule transfer.
a, Insulin microneedle patch dissolution in 0.3% agarose gel. Patches which were inserted into the gel for a set period of time were compared to patches which were laid on top of the gel for 30 s (control). The figure includes views of the gel from the side (left) and top (right) after the patch was removed. Scale bars = 1 cm. b, Insulin transferred from the patch to the gel quantified via HPLC. (n = 3 device replicates. Error Bars = SD. Center line = mean. *P < 0.05, **P < 0.01). c, Dissolution of insulin microneedle patches after application to in vivo swine small intestine. Control patches were laid upon the tissue and all other patches were penetrated into to the tissue. Scale bars = 1 mm. d, In vivo imaging system fluorescent image of swine tissue applied with Texas red loaded microneedle patches. Patches were applied in vivo. The control patches were left to sit on top of the tissue, but they were not pressed into the tissue. Scale bar = 1 cm.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2
Supplementary Video 1
Penetration of needles into swine small intestine tissue
Source data
Source Data Fig. 1
Statistical Source Data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 4
Statistical Source Data
Source Data Extended Data Fig. 2
Statistical Source Data
Source Data Extended Data Fig. 5
Statistical Source Data
Source Data Extended Data Fig. 8
Statistical Source Data
Source Data Extended Data Fig. 9
Statistical Source Data
Rights and permissions
About this article
Cite this article
Abramson, A., Caffarel-Salvador, E., Soares, V. et al. A luminal unfolding microneedle injector for oral delivery of macromolecules. Nat Med 25, 1512–1518 (2019). https://doi.org/10.1038/s41591-019-0598-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-019-0598-9
This article is cited by
-
Microneedle-mediated nanomedicine to enhance therapeutic and diagnostic efficacy
Nano Convergence (2024)
-
End-to-end design of ingestible electronics
Nature Electronics (2024)
-
A wireless battery-free eye modulation patch for high myopia therapy
Nature Communications (2024)
-
Design of chimeric GLP-1A using oligomeric bile acids to utilize transporter-mediated endocytosis for oral delivery
Biomaterials Research (2023)
-
Advances of medical nanorobots for future cancer treatments
Journal of Hematology & Oncology (2023)