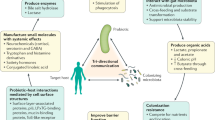
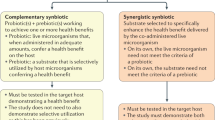
Abstract
Consumption of over-the-counter probiotics for promotion of health and well-being has increased worldwide in recent years. However, although probiotic use has been greatly popularized among the general public, there are conflicting clinical results for many probiotic strains and formulations. Emerging insights from microbiome research enable an assessment of gut colonization by probiotics, strain-level activity, interactions with the indigenous microbiome, safety and impacts on the host, and allow the association of probiotics with physiological effects and potentially useful medical indications. In this Perspective, we highlight key advances, challenges and limitations in striving toward an unbiased interpretation of the large amount of data regarding over-the-counter probiotics, and propose avenues to improve the quality of evidence, transparency, public awareness and regulation of their use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hill, C. et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 11, 506–514 (2014).
Global Probiotic Market Is Set For Rapid Growth and is Expected To Reach Value Around USD 65.87 Billion by 2024 (Zion Market Research, 2018).
Clarke, T. C., Black, L. I., Stussman, B. J., Barnes, P. M. & Nahin, R. L. Trends in the use of complementary health approaches among adults: United States, 2002–2012. Natl. Health Stat. Report. 79, 1–16 (2015).
Hoffmann, D. E. et al. Probiotics: achieving a better regulatory fit. Food Drug Law J. 69, 237–272 (2014).
Draper, K., Ley, C. & Parsonnet, J. Probiotic guidelines and physician practice: a cross-sectional survey and overview of the literature. Benef. Microbes 8, 507–519 (2017).
Williams, M. D., Ha, C. Y. & Ciorba, M. A. Probiotics as therapy in gastroenterology: a study of physician opinions and recommendations. J. Clin. Gastroenterol. 44, 631–636 (2010).
Rijkers, G. T. et al. Health benefits and health claims of probiotics: bridging science and marketing. Br. J. Nutr. 106, 1291–1296 (2011).
Saldanha, L. G. US Food and Drug Administration regulations governing label claims for food products, including probiotics. Clin. Infect. Dis. 46, S119–121 (2008).
Degnan, F. H. Clinical studies involving probiotics: when FDA’s investigational new drug rubric applies-and when it may not. Gut Microbes 3, 485–489 (2012).
Sniffen, J. C., McFarland, L. V., Evans, C. T. & Goldstein, E. J. C. Choosing an appropriate probiotic product for your patient: an evidence-based practical guide. PLoS One 13, e0209205 (2018).
El Hage, R., Hernandez-Sanabria, E. & Van de Wiele, T. Emerging trends in “smart probiotics”: functional consideration for the development of novel health and industrial applications. Front. Microbiol. 8, 1889 (2017).
Panigrahi, P. et al. A randomized synbiotic trial to prevent sepsis among infants in rural India. Nature 548, 407–412 (2017).
Kruis, W. et al. Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut 53, 1617–1623 (2004).
Canani, R. B. et al. Probiotics for treatment of acute diarrhoea in children: randomised clinical trial of five different preparations. Br. Med. J. 335, 340 (2007).
Ruszczyński, M., Radzikowski, A. & Szajewska, H. Clinical trial: effectiveness of Lactobacillus rhamnosus (strains E/N, Oxy and Pen) in the prevention of antibiotic-associated diarrhoea in children. Aliment. Pharmacol. Ther. 28, 154–161 (2008).
Gao, X. W., Mubasher, M., Fang, C. Y., Reifer, C. & Miller, L. E. Dose–response efficacy of a proprietary probiotic formula of Lactobacillus acidophilus CL1285 and Lactobacillus casei LBC80R for antibiotic-associated diarrhea and Clostridium difficile–associated diarrhea prophylaxis in adult patients. Am. J. Gastroenterol. 105, 1636–1641 (2010).
Fujimori, S. et al. A randomized controlled trial on the efficacy of synbiotic versus probiotic or prebiotic treatment to improve the quality of life in patients with ulcerative colitis. Nutrition 25, 520–525 (2009).
Benton, D., Williams, C. & Brown, A. Impact of consuming a milk drink containing a probiotic on mood and cognition. Eur. J. Clin. Nutr. 61, 355–361 (2007).
Kekkonen, R. A. et al. Probiotic intervention has strain-specific anti-inflammatory effects in healthy adults. World J. Gastroenterol. 14, 2029–2036 (2008).
Simon, M. C. et al. Intake of Lactobacillus reuteri improves incretin and insulin secretion in glucose-tolerant humans: a proof of concept. Diabetes Care 38, 1827–1834 (2015).
Pereg, D. et al. The effect of fermented yogurt on the prevention of diarrhea in a healthy adult population. Am. J. Infect. Control 33, 122–125 (2005).
Dietrich, C. G., Kottmann, T. & Alavi, M. Commercially available probiotic drinks containing Lactobacillus casei DN-114001 reduce antibiotic-associated diarrhea. World J. Gastroenterol. 20, 15837–15844 (2014).
Gareau, M. G., Sherman, P. M. & Walker, W. A. Probiotics and the gut microbiota in intestinal health and disease. Nat. Rev. Gastroenterol. Hepatol. 7, 503–514 (2010).
Begley, M., Hill, C. & Gahan, C. G. Bile salt hydrolase activity in probiotics. Appl. Environ. Microbiol. 72, 1729–1738 (2006).
de Vrieze, J. The metawars. Science 361, 1184–1188 (2018).
Moayyedi, P. et al. The efficacy of probiotics in the treatment of irritable bowel syndrome: a systematic review. Gut 59, 325–332 (2010).
Shimizu, M., Hashiguchi, M., Shiga, T., Tamura, H. O. & Mochizuki, M. Meta-analysis: effects of probiotic supplementation on lipid profiles in normal to mildly hypercholesterolemic individuals. PLoS One 10, e0139795 (2015).
Lu, C. et al. Probiotic supplementation does not improve eradication rate of Helicobacter pylori infection compared to placebo based on standard therapy: a meta-analysis. Sci. Rep. 6, 23522 (2016).
Lü, M. et al. Efficacy of probiotic supplementation therapy for Helicobacter pylori eradication: a meta-analysis of randomized controlled trials. PLoS One 11, e0163743 (2016).
Kolber, M. R., Vandermeer, B. & Allan, G. M. Funding may influence trial results examining probiotics and Clostridium difficile diarrhea rates. Am. J. Gastroenterol. 109, 1081–1082 (2014).
Allen, S. J., Martinez, E. G., Gregorio, G. V. & Dans, L. F. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst. Rev. 11, CD003048 (2010).
Feizizadeh, S., Salehi-Abargouei, A. & Akbari, V. Efficacy and safety of Saccharomyces boulardii for acute diarrhea. Pediatrics 134, e176–e191 (2014).
Szajewska, H., Skórka, A., Ruszczyński, M. & Gieruszczak-Białek, D. Meta-analysis: Lactobacillus GG for treating acute gastroenteritis in children—updated analysis of randomised controlled trials. Aliment. Pharmacol. Ther. 38, 467–476 (2013).
Van Niel, C. W., Feudtner, C., Garrison, M. M. & Christakis, D. A. Lactobacillus therapy for acute infectious diarrhea in children: a meta-analysis. Pediatrics 109, 678–684 (2002).
Goldenberg, J. Z. et al. Probiotics for the prevention of pediatric antibiotic-associated diarrhea. Cochrane Database Syst. Rev. 11, CD004827 (2015).
Hempel, S. et al. Probiotics for the prevention and treatment of antibiotic-associated diarrhea: a systematic review and meta-analysis. J. Am. Med. Assoc. 307, 1959–1969 (2012).
Jafarnejad, S. et al. Probiotics reduce the risk of antibiotic-associated diarrhea in adults (18–64 years) but not the elderly (>65 years): a meta-analysis.Nutr. Clin. Pract. 31, 502–513 2016).
Hickson, M. et al. Use of probiotic Lactobacillus preparation to prevent diarrhoea associated with antibiotics: randomised double blind placebo controlled trial. Br. Med. J. 335, 80 (2007).
Olek, A. et al. Efficacy and safety of Lactobacillus plantarum DSM 9843 (LP299V) in the prevention of antibiotic-associated gastrointestinal symptoms in children-randomized, double-blind, placebo-controlled study. J. Pediatr. 186, 82–86 2017).
Allen, S. J. et al. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): a randomised, double-blind, placebo-controlled, multicentre trial. Lancet 382, 1249–1257 (2013).
Freedman, S. B. et al. Multicenter trial of a combination probiotic for children with gastroenteritis. N. Engl. J. Med. 379, 2015–2026 2018).
Schnadower, D. et al. Lactobacillus rhamnosus GG versus placebo for acute gastroenteritis in children. N. Engl. J. Med. 379, 2002–2014 2018).
Freedman, S. B. et al. Gastroenteritis therapies in developed countries: systematic review and meta-analysis. PLoS One 10, e0128754 (2015).
Khanna, R., Lakhanpaul, M., Burman-Roy, S. & Murphy, M. S. Diarrhoea and vomiting caused by gastroenteritis in children under 5 years: summary of NICE guidance. Br. Med. J. 338, b1350 (2009).
Szajewska, H. et al. Use of probiotics for management of acute gastroenteritis: a position paper by the ESPGHAN Working Group for Probiotics and Prebiotics. J. Pediatr. Gastroenterol. Nutr. 58, 531–539 (2014).
Li, S. T., Klein, E. J., Tarr, P. I. & Denno, D. M. Parental management of childhood diarrhea. Clin. Pediatr. (Phila.) 48, 295–303 (2009).
Goldenberg, J. Z. et al. Probiotics for the prevention of Clostridium difficile–associated diarrhea in adults and children. Cochrane Database Syst. Rev. 5, CD006095 (2013).
Shen, N. T. et al. Timely use of probiotics in hospitalized adults prevents Clostridium difficile infection: a systematic review with meta-regression analysis. Gastroenterology 152, 1889–1900 e1889 (2017).
Goldenberg, J. Z. et al. Probiotics for the prevention of Clostridium difficile–associated diarrhea in adults and children. Cochrane Database Syst. Rev. 12, CD006095 (2017).
McFarland, L. V. Meta-analysis of probiotics for the prevention of antibiotic associated diarrhea and the treatment of Clostridium difficile disease. Am. J. Gastroenterol. 101, 812–822 (2006).
Szajewska, H. & Kołodziej, M. Systematic review with meta-analysis: Saccharomyces boulardii in the prevention of antibiotic-associated diarrhoea. Aliment. Pharmacol. Ther. 42, 793–801 (2015).
Szajewska, H.et al. Probiotics for the prevention of antibiotic-associated diarrhea in children. J. Pediatr. Gastroenterol. Nutr. 62, 495–506 2016).
Georgieva, M. et al. Use of the probiotic Lactobacillus reuteri DSM 17938 in the prevention of antibiotic-associated infections in hospitalIzed Bulgarian children: a randomized, controlled trial. J. IMAB–Annu. Proc. Sci. Pap. 21, 895–900 (2015).
Ouwehand, A. C. et al. Probiotics reduce symptoms of antibiotic use in a hospital setting: a randomized dose response study. Vaccine 32, 458–463 (2014).
Klarin, B. et al. Lactobacillus plantarum 299v reduces colonisation of Clostridium difficile in critically ill patients treated with antibiotics. Acta Anaesthesiol. Scand. 52, 1096–1102 (2008).
Morrow, L. E., Kollef, M. H. & Casale, T. B. Probiotic prophylaxis of ventilator-associated pneumonia: a blinded, randomized, controlled trial. Am. J. Respir. Crit. Care Med. 182, 1058–1064 (2010).
Shan, L. S. et al. Prevention and treatment of diarrhoea with Saccharomyces boulardii in children with acute lower respiratory tract infections. Benef. Microbes 4, 329–334 (2013).
Rafiq, R. et al. in Gastroenterology, Vol. 132. A187 (WB Saunders Co–Elsevier, 2007).
Lemann, M., Cezard, J., Ruemmele, F. & Turck, D. European Society for Paediatric Gastroenterology, Hepatology, and Nutrition Annual Meeting June 3–6, 2009 Budapest, Hungary. J. Pediatr. Gastroenterol. Nutr. 48, E1–E149 (2009).
Viggars, A. P., Gracie, D. J. & Ford, A. C. Use of probiotics in hospitalized adults to prevent Clostridium difficile infection: downgrade the quality of evidence?. Gastroenterology 153, 1451–1452 (2017).
McFarland, L. V. Deciphering meta-analytic results: a mini-review of probiotics for the prevention of paediatric antibiotic-associated diarrhoea and Clostridium difficile infections. Benef. Microbes 6, 189–194 (2015).
Guyonnet, D. et al. Effect of a fermented milk containing Bifidobacterium animalis DN-173 010 on the health-related quality of life and symptoms in irritable bowel syndrome in adults in primary care: a multicentre, randomized, double-blind, controlled trial. Aliment. Pharmacol. Ther. 26, 475–486 (2007).
Ford, A. C., Harris, L. A., Lacy, B. E., Quigley, E. M. M. & Moayyedi, P. Systematic review with meta-analysis: the efficacy of prebiotics, probiotics, synbiotics and antibiotics in irritable bowel syndrome. Aliment. Pharmacol. Ther. 48, 1044–1060 (2018).
McKenzie, Y. A., Thompson, J., Gulia, P. & Lomer, M. C. British Dietetic Association systematic review of systematic reviews and evidence-based practice guidelines for the use of probiotics in the management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 29, 576–592 (2016).
Olsen, R., Greisen, G., Schrøder, M. & Brok, J. Prophylactic probiotics for preterm infants: a systematic review and meta-analysis of observational studies. Neonatology 109, 105–112 (2016).
Rao, S. C., Athalye-Jape, G. K., Deshpande, G. C., Simmer, K. N. & Patole, S. K. Probiotic supplementation and late-onset sepsis in preterm infants: a meta-analysis. Pediatrics 137, e20153684 (2016).
Ganguli, K. et al. Probiotics prevent necrotizing enterocolitis by modulating enterocyte genes that regulate innate immune-mediated inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 304, G132–G141 (2013).
Yan, F. et al. Neonatal colonization of mice with LGG promotes intestinal development and decreases susceptibility to colitis in adulthood. Mucosal Immunol. 10, 117–127 (2017).
Costeloe, K., Hardy, P., Juszczak, E., Wilks, M. & Millar, M. R. Bifidobacterium breve BBG-001 in very preterm infants: a randomised controlled phase 3 trial. Lancet 387, 649–660 (2016).
AlFaleh, K. & Anabrees, J. Probiotics for prevention of necrotizing enterocolitis in preterm infants. Cochrane Database Syst. Rev. 4, CD005496 (2014).
Aceti, A. et al. Probiotics prevent late-onset sepsis in human milk-fed, very low birth weight preterm infants: systematic review and meta-analysis. Nutrients 9, 904 (2017).
Dermyshi, E. et al. The “golden age” of probiotics: a systematic review and meta-analysis of randomized and observational studies in preterm infants. Neonatology 112, 9–23 (2017).
Zhang, G. Q., Hu, H. J., Liu, C. Y., Shakya, S. & Li, Z. Y. Probiotics for preventing late-onset sepsis in preterm neonates: A PRISMA-compliant systematic review and meta-analysis of randomized controlled trials. Med. (Baltim.) 95, e2581 (2016).
Sommer, F. & Bäckhed, F. The gut microbiota—masters of host development and physiology. Nat. Rev. Microbiol. 11, 227–238 (2013).
King, S. et al. Does probiotic consumption reduce antibiotic utilization for common acute infections? A systematic review and meta-analysis. Eur. J. Public Health https://doi.org/10.1093/eurpub/cky185 (2018).
Hao, Q., Dong, B.R. & Wu, T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst. Rev. 2, CD006895 (2015).
Vouloumanou, E. K., Makris, G. C., Karageorgopoulos, D. E. & Falagas, M. E. Probiotics for the prevention of respiratory tract infections: a systematic review. Int. J. Antimicrob. Agents 34, 197.e1–197.e10 (2009).
Merenstein, D. et al. Use of a fermented dairy probiotic drink containing Lactobacillus casei (DN-114 001) to decrease the rate of illness in kids: the DRINK study. A patient-oriented, double-blind, cluster-randomized, placebo-controlled, clinical trial. Eur. J. Clin. Nutr. 64, 669–677 (2010).
de Vrese, M. et al. Effect of Lactobacillus gasseri PA 16/8, Bifidobacterium longum SP 07/3, B. bifidum MF 20/5 on common cold episodes: a double blind, randomized, controlled trial. Clin. Nutr. (Edinb., Scotl.) 24, 481–491 (2005).
Smith, T. J., Rigassio-Radler, D., Denmark, R., Haley, T. & Touger-Decker, R. Effect of Lactobacillus rhamnosus LGG® and Bifidobacterium animalis ssp. lactis BB-12® on health-related quality of life in college students affected by upper respiratory infections. Br. J. Nutr. 109, 1999–2007 (2013).
Shinkai, S. et al. Immunoprotective effects of oral intake of heat-killed Lactobacillus pentosus strain b240 in elderly adults: a randomised, double-blind, placebo-controlled trial. Br. J. Nutr. 109, 1856–1865 (2013).
Hatakka, K. et al. Effect of long term consumption of probiotic milk on infections in children attending day care centres: double blind, randomised trial. Br. Med. J. 322, 1327 (2001).
West, N. P. et al. Lactobacillus fermentum (PCC®) supplementation and gastrointestinal and respiratory-tract illness symptoms: a randomised control trial in athletes. Nutr. J. 10, 30 (2011).
Murata, M. et al. Effects of paraprobiotic Lactobacillus paracasei MCC1849 supplementation on symptoms of the common cold and mood states in healthy adults. Benef. Microbes 9, 855–864 (2018).
Atarashi, K. et al. TH17 cell induction by adhesion of microbes to intestinal epithelial cells. Cell 163, 367–380 (2015).
Thaiss, C. A. et al. Microbiota diurnal rhythmicity programs host transcriptome oscillations. Cell 167, 1495–1510.e12 (2016).
Uchimura, Y. et al. Antibodies set boundaries limiting microbial metabolite penetration and the resultant mammalian host response. Immunity 49, 545–559.e5 (2018).
Mattar, A. F. et al. Probiotics up-regulate MUC-2 mucin gene expression in a Caco-2 cell-culture model. Pediatr. Surg. Int. 18, 586–590 (2002).
Zmora, N. et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174, 1388–1405.e21 (2018).
Turroni, F. et al. Role of sortase-dependent pili of Bifidobacterium bifidum PRL2010 in modulating bacterium-host interactions. Proc. Natl Acad. Sci. USA 110, 11151–11156 (2013).
Van Tassell, M. L. & Miller, M. J. Lactobacillus adhesion to mucus. Nutrients 3, 613–636 (2011).
Fujimura, S. et al. Detection of Lactobacillus gasseri OLL2716 strain administered with yogurt drink in gastric mucus layer in humans. Lett. Appl. Microbiol. 43, 578–581 (2006).
Valeur, N., Engel, P., Carbajal, N., Connolly, E. & Ladefoged, K. Colonization and immunomodulation by Lactobacillus reuteri ATCC 55730 in the human gastrointestinal tract. Appl. Environ. Microbiol. 70, 1176–1181 (2004).
Johansson, M. L. et al. Administration of different Lactobacillus strains in fermented oatmeal soup: in vivo colonization of human intestinal mucosa and effect on the indigenous flora. Appl. Environ. Microbiol. 59, 15–20 (1993).
Shibahara-Sone, H. et al. Living cells of probiotic Bifidobacterium bifidum YIT 10347 detected on gastric mucosa in humans. Benef. Microbes 7, 319–326 (2016).
Yang, Y., Galle, S., Le, M. H., Zijlstra, R. T. & Gänzle, M. G. Feed fermentation with reuteran- and levan-producing Lactobacillus reuteri reduces colonization of weanling pigs by enterotoxigenic Escherichia coli. Appl. Environ. Microbiol. 81, 5743–5752 (2015).
Riboulet-Bisson, E. et al. Effect of Lactobacillus salivarius bacteriocin Abp118 on the mouse and pig intestinal microbiota. PLoS One 7, e31113 (2012).
Crittenden, R. et al. Lactobacillus paracasei subsp. paracasei F19: Survival, ecology and safety in the human intestinal tract-A survey of feeding studies within the PROBDEMO project. Microb. Ecol. Health Dis. 14, 22–26 (2002).
Goossens, D. A., Jonkers, D. M., Russel, M. G., Stobberingh, E. E. & Stockbrügger, R. W. The effect of a probiotic drink with Lactobacillus plantarum 299v on the bacterial composition in faeces and mucosal biopsies of rectum and ascending colon. Aliment. Pharmacol. Ther. 23, 255–263 (2006).
Alander, M. et al. Persistence of colonization of human colonic mucosa by a probiotic strain, Lactobacillus rhamnosus GG, after oral consumption. Appl. Environ. Microbiol. 65, 351–354 (1999).
Gianotti, L. et al. A randomized double-blind trial on perioperative administration of probiotics in colorectal cancer patients. World J. Gastroenterol. 16, 167–175 (2010).
Suez, J., Zmora, N. & Elinav, E. Probiotics in the next-generation sequencing era. Gut Microbes 5, 1–17 (2019).
Zhang, C. et al. Ecological robustness of the gut microbiota in response to ingestion of transient food-borne microbes. ISME J. 10, 2235–2245 (2016).
Charbonneau, D., Gibb, R. D. & Quigley, E. M. Fecal excretion of Bifidobacterium infantis 35624 and changes in fecal microbiota after eight weeks of oral supplementation with encapsulated probiotic. Gut Microbes 4, 201–211 (2013).
Alander, M. et al. Effect of galacto-oligosaccharide supplementation on human faecal microflora and on survival and persistence of Bifidobacterium lactis Bb-12 in the gastrointestinal tract. Int. Dairy J. 11, 817–825 (2001).
Firmesse, O., Mogenet, A., Bresson, J. L., Corthier, G. & Furet, J. P. Lactobacillus rhamnosus R11 consumed in a food supplement survived human digestive transit without modifying microbiota equilibrium as assessed by real-time polymerase chain reaction. J. Mol. Microbiol. Biotechnol. 14, 90–99 (2008).
Rochet, V. et al. Effects of orally administered Lactobacillus casei DN-114 001 on the composition or activities of the dominant faecal microbiota in healthy humans. Br. J. Nutr. 95, 421–429 (2006).
Garrido, D., Suau, A., Pochart, P., Cruchet, S. & Gotteland, M. Modulation of the fecal microbiota by the intake of a Lactobacillus johnsonii La1-containing product in human volunteers. FEMS Microbiol. Lett. 248, 249–256 (2005).
Goossens, D. et al. The effect of Lactobacillus plantarum 299v on the bacterial composition and metabolic activity in faeces of healthy volunteers: a placebo-controlled study on the onset and duration of effects. Aliment. Pharmacol. Ther. 18, 495–505 (2003).
Smith, T. J., Anderson, D., Margolis, L. M., Sikes, A. & Young, A. J. Persistence of Lactobacillus reuteri DSM17938 in the human intestinal tract: response to consecutive and alternate-day supplementation. J. Am. Coll. Nutr. 30, 259–264 (2011).
Jacobsen, C. N. et al. Screening of probiotic activities of forty-seven strains of Lactobacillus spp. by in vitro techniques and evaluation of the colonization ability of five selected strains in humans. Appl. Environ. Microbiol. 65, 4949–4956 (1999).
Sierra, S. et al. Intestinal and immunological effects of daily oral administration of Lactobacillus salivarius CECT5713 to healthy adults. Anaerobe 16, 195–200 (2010).
Frese, S. A., Hutkins, R. W. & Walter, J. Comparison of the colonization ability of autochthonous and allochthonous strains of lactobacilli in the human gastrointestinal tract. Adv. Microbiol. 2, 399 (2012).
Tannock, G. W. et al. Analysis of the fecal microflora of human subjects consuming a probiotic product containing Lactobacillus rhamnosus DR20. Appl. Environ. Microbiol. 66, 2578–2588 (2000).
Maldonado-Gómez, M. X. et al. Stable engraftment of Bifidobacterium longum AH1206 in the human gut depends on individualized features of the resident microbiome. Cell Host Microbe 20, 515–526 (2016).
Suez, J. et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 174, 1406–1423.e16 (2018).
Chung, H. et al. Gut immune maturation depends on colonization with a host-specific microbiota. Cell 149, 1578–1593 (2012).
Dogi, C. A. & Perdigón, G. Importance of the host specificity in the selection of probiotic bacteria. J. Dairy Res. 73, 357–366 (2006).
Marcobal, A. et al. A metabolomic view of how the human gut microbiota impacts the host metabolome using humanized and gnotobiotic mice. ISME J. 7, 1933–1943 (2013).
de Vrese, M. et al. Probiotics—compensation for lactase insufficiency. Am. J. Clin. Nutr. 73, 421S–429S (2001).
Bonder, M. J. et al. The effect of host genetics on the gut microbiome. Nat. Genet. 48, 1407–1412 (2016).
Macho Fernandez, E. et al. Anti-inflammatory capacity of selected lactobacilli in experimental colitis is driven by NOD2-mediated recognition of a specific peptidoglycan-derived muropeptide. Gut 60, 1050–1059 (2011).
Lin, Y. P., Thibodeaux, C. H., Peña, J. A., Ferry, G. D. & Versalovic, J. Probiotic Lactobacillus reuteri suppress proinflammatory cytokines via c-Jun. Inflamm. Bowel Dis. 14, 1068–1083 (2008).
Lavasani, S. et al. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLoS One 5, e9009 (2010).
Thomas, C. M. & Versalovic, J. Probiotics–host communication: modulation of signaling pathways in the intestine. Gut Microbes 1, 148–163 (2010).
van Baarlen, P. et al. Differential NF-κB pathways induction by Lactobacillus plantarum in the duodenum of healthy humans correlating with immune tolerance. Proc. Natl Acad. Sci. USA 106, 2371–2376 (2009).
Matsuguchi, T. et al. Lipoteichoic acids from Lactobacillus strains elicit strong tumor necrosis factor alpha-inducing activities in macrophages through Toll-like receptor 2. Clin. Diagn. Lab. Immunol. 10, 259–266 (2003).
Medina, M., Izquierdo, E., Ennahar, S. & Sanz, Y. Differential immunomodulatory properties of Bifidobacterium logum strains: relevance to probiotic selection and clinical applications. Clin. Exp. Immunol. 150, 531–538 (2007).
Schiavi, E. et al. The surface-associated exopolysaccharide of Bifidobacterium longum 35624 plays an essential role in dampening host proinflammatory responses and repressing local Th17 responses. Appl. Environ. Microbiol. 82, 7185–7196 (2016).
von Ossowski, I. et al. Using recombinant Lactococci as an approach to dissect the immunomodulating capacity of surface piliation in probiotic Lactobacillus rhamnosus GG. PLoS One 8, e64416 (2013).
Ardita, C. S. et al. Epithelial adhesion mediated by pilin SpaC is required for Lactobacillus rhamnosus GG–induced cellular responses. Appl. Environ. Microbiol. 80, 5068–5077 (2014).
Yanagihara, S. et al. Uromodulin-SlpA binding dictates Lactobacillus acidophilus uptake by intestinal epithelial M cells. Int. Immunol. 29, 357–363 (2017).
Konieczna, P. et al. Bifidobacterium infantis 35624 administration induces Foxp3 T regulatory cells in human peripheral blood: potential role for myeloid and plasmacytoid dendritic cells. Gut 61, 354–366 (2012).
Fukushima, Y., Kawata, Y., Hara, H., Terada, A. & Mitsuoka, T. Effect of a probiotic formula on intestinal immunoglobulin A production in healthy children. Int. J. Food Microbiol. 42, 39–44 (1998).
Galdeano, C. M. & Perdigón, G. The probiotic bacterium Lactobacillus casei induces activation of the gut mucosal immune system through innate immunity. Clin. Vaccin. Immunol. 13, 219–226 (2006).
Gueimonde, M., Margolles, A., de los Reyes-Gavilán, C. G. & Salminen, S. Competitive exclusion of enteropathogens from human intestinal mucus by Bifidobacterium strains with acquired resistance to bile—a preliminary study. Int. J. Food Microbiol. 113, 228–232 (2007).
Tsai, C. C. et al. Antagonistic activity against Salmonella infection in vitro and in vivo for two Lactobacillus strains from swine and poultry. Int. J. Food Microbiol. 102, 185–194 (2005).
Kim, Y., Kim, S. H., Whang, K. Y., Kim, Y. J. & Oh, S. Inhibition of Escherichia coli O157:H7 attachment by interactions between lactic acid bacteria and intestinal epithelial cells. J. Microbiol. Biotechnol. 18, 1278–1285 (2008).
Fukuda, S. et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547 (2011).
Cotter, P. D., Hill, C. & Ross, R. P. Bacteriocins: developing innate immunity for food. Nat. Rev. Microbiol. 3, 777–788 (2005).
Corr, S. C. et al. Bacteriocin production as a mechanism for the antiinfective activity of Lactobacillus salivarius UCC118. Proc. Natl Acad. Sci. USA 104, 7617–7621 (2007).
Medellin-Peña, M. J., Wang, H., Johnson, R., Anand, S. & Griffiths, M. W. Probiotics affect virulence-related gene expression in Escherichia coli O157:H7. Appl. Environ. Microbiol. 73, 4259–4267 (2007).
Yun, B., Oh, S. & Griffiths, M. W. Lactobacillus acidophilus modulates the virulence of Clostridium difficile. J. Dairy Sci. 97, 4745–4758 (2014).
Li, J., Wang, W., Xu, S. X., Magarvey, N. A. & McCormick, J. K. Lactobacillus reuteri-produced cyclic dipeptides quench agr-mediated expression of toxic shock syndrome toxin-1 in staphylococci. Proc. Natl Acad. Sci. USA 108, 3360–3365 (2011).
Miller, M. B. & Bassler, B. L. Quorum sensing in bacteria. Annu. Rev. Microbiol. 55, 165–199 (2001).
Lagrafeuille, R. et al. Opposing effect of Lactobacillus on in vitro Klebsiella pneumoniae in biofilm and in an in vivo intestinal colonisation model. Benef. Microbes 9, 87–100 (2018).
Thompson, J. A., Oliveira, R. A., Djukovic, A., Ubeda, C. & Xavier, K. B. Manipulation of the quorum sensing signal AI-2 affects the antibiotic-treated gut microbiota. Cell Rep. 10, 1861–1871 (2015).
Ohland, C. L. & Macnaughton, W. K. Probiotic bacteria and intestinal epithelial barrier function. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G807–G819 (2010).
Miyamoto, J. et al. A gut microbial metabolite of linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, ameliorates intestinal epithelial barrier impairment partially via GPR40–MEK–ERK pathway. J. Biol. Chem. 290, 2902–2918 (2015).
Kaikiri, H. et al. Supplemental feeding of a gut microbial metabolite of linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, alleviates spontaneous atopic dermatitis and modulates intestinal microbiota in NC/nga mice. Int. J. Food Sci. Nutr. 68, 941–951 (2017).
Yamada, M. et al. A bacterial metabolite ameliorates periodontal pathogen-induced gingival epithelial barrier disruption via GPR40 signaling. Sci. Rep. 8, 9008 (2018).
Yan, F. et al. Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology 132, 562–575 (2007).
Mack, D. R., Ahrne, S., Hyde, L., Wei, S. & Hollingsworth, M. A. Extracellular MUC3 mucin secretion follows adherence of Lactobacillus strains to intestinal epithelial cells in vitro. Gut 52, 827–833 (2003).
Gaudier, E., Michel, C., Segain, J. P., Cherbut, C. & Hoebler, C. The VSL# 3 probiotic mixture modifies microflora but does not heal chronic dextran-sodium sulfate-induced colitis or reinforce the mucus barrier in mice. J. Nutr. 135, 2753–2761 (2005).
Caballero-Franco, C., Keller, K., De Simone, C. & Chadee, K. The VSL#3 probiotic formula induces mucin gene expression and secretion in colonic epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 292, G315–G322 (2007).
Persborn, M. et al. The effects of probiotics on barrier function and mucosal pouch microbiota during maintenance treatment for severe pouchitis in patients with ulcerative colitis. Aliment. Pharmacol. Ther. 38, 772–783 (2013).
Jones, C. et al. Modulation of gut barrier function in patients with obstructive jaundice using probiotic LP299v. Eur. J. Gastroenterol. Hepatol. 25, 1424–1430 (2013).
Zeng, J. et al. Clinical trial: effect of active lactic acid bacteria on mucosal barrier function in patients with diarrhoea-predominant irritable bowel syndrome. Aliment. Pharmacol. Ther. 28, 994–1002 (2008).
Sabico, S. et al. Effects of a multi-strain probiotic supplement for 12 weeks in circulating endotoxin levels and cardiometabolic profiles of medication naïve T2DM patients: a randomized clinical trial. J. Transl. Med. 15, 249 (2017).
Wilms, E. et al. Effects of supplementation of the synbiotic ecologic® 825/FOS P6 on intestinal barrier function in healthy humans: a randomized controlled trial. PLoS One 11, e0167775 (2016).
Horvath, A. et al. Randomised clinical trial: the effects of a multispecies probiotic vs. placebo on innate immune function, bacterial translocation and gut permeability in patients with cirrhosis. Aliment. Pharmacol. Ther. 44, 926–935 (2016).
Stadlbauer, V. et al. Lactobacillus casei shirota supplementation does not restore gut microbiota composition and gut barrier in metabolic syndrome: a randomized pilot study. PLoS One 10, e0141399 (2015).
Joyce, S. A. et al. Regulation of host weight gain and lipid metabolism by bacterial bile acid modification in the gut. Proc. Natl Acad. Sci. USA 111, 7421–7426 (2014).
Costabile, A. et al. An in vivo assessment of the cholesterol-lowering efficacy of Lactobacillus plantarum ECGC 13110402 in normal to mildly hypercholesterolaemic adults. PLoS One 12, e0187964 (2017).
Sun, L. et al. Gut microbiota and intestinal FXR mediate the clinical benefits of metformin. Nat. Med. 24, 1919–1929 (2018).
Sarkar, A. et al. Psychobiotics and the manipulation of bacteria–gut–brain signals. Trends Neurosci. 39, 763–781 (2016).
Bravo, J. A. et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl Acad. Sci. USA 108, 16050–16055 (2011).
Kelly, J. R. et al. Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects. Brain Behav. Immun. 61, 50–59 (2017).
Buffington, S. A. et al. Microbial reconstitution reverses maternal diet-induced social and synaptic deficits in offspring. Cell 165, 1762–1775 (2016).
Perez-Burgos, A. et al. The TRPV1 channel in rodents is a major target for antinociceptive effect of the probiotic Lactobacillus reuteri DSM 17938. J. Physiol. (Lond.) 593, 3943–3957 (2015).
Rousseaux, C. et al. Lactobacillus acidophilus modulates intestinal pain and induces opioid and cannabinoid receptors. Nat. Med. 13, 35–37 (2007).
Reis, D. J., Ilardi, S. S. & Punt, S. E. W. The anxiolytic effect of probiotics: a systematic review and meta-analysis of the clinical and preclinical literature. PLoS One 13, e0199041 (2018).
Kristensen, N. B. et al. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: a systematic review of randomized controlled trials. Genome Med. 8, 52 (2016).
McFarland, L. V. Use of probiotics to correct dysbiosis of normal microbiota following disease or disruptive events: a systematic review. BMJ Open 4, e005047 (2014).
Jakobsson, H. E. et al. Short-term antibiotic treatment has differing long-term impacts on the human throat and gut microbiome. PLoS One 5, e9836 (2010).
Sommer, F., Anderson, J. M., Bharti, R., Raes, J. & Rosenstiel, P. The resilience of the intestinal microbiota influences health and disease. Nat. Rev. Microbiol. 15, 630–638 (2017).
Bruzzese, E. et al. Disrupted intestinal microbiota and intestinal inflammation in children with cystic fibrosis and its restoration with Lactobacillus GG: a randomised clinical trial. PLoS One 9, e87796 (2014).
Zoppi, G., Cinquetti, M., Benini, A., Bonamini, E. & Minelli, E. B. Modulation of the intestinal ecosystem by probiotics and lactulose in children during treatment with ceftriaxone. Curr. Ther. Res. Clin. Exp. 62, 418–435 (2001).
Wang, Z. J. et al. Effects of anti–Helicobacter pylori concomitant therapy and probiotic supplementation on the throat and gut microbiota in humans. Microb. Pathog. 109, 156–161 (2017).
Khalesi, S. et al. A review of probiotic supplementation in healthy adults: helpful or hype?. Eur. J. Clin. Nutr. 73, 24–37, doi: (2019).
Ricci, A. et al. Update of the list of QPS‐recommended biological agents intentionally added to food or feed as notified to EFSA 8: suitability of taxonomic units notified to EFSA until March 2018. EFSA Journal 16, e05315 (2018).
Quin, C. et al. Probiotic supplementation and associated infant gut microbiome and health: a cautionary retrospective clinical comparison. Sci. Rep. 8, 8283 (2018).
Topcuoglu, S., Gursoy, T., Ovali, F., Serce, O. & Karatekin, G. A new risk factor for neonatal vancomycin-resistant Enterococcus colonisation: bacterial probiotics. J. Matern. Fetal Neonatal Med. 28, 1491–1494 (2015).
Didari, T., Solki, S., Mozaffari, S., Nikfar, S. & Abdollahi, M. A systematic review of the safety of probiotics. Expert Opin. Drug Saf. 13, 227–239 (2014).
Carvour, M. L. et al. Predictors of Clostridium difficile infection and predictive impact of probiotic use in a diverse hospital-wide cohort. Am. J. Infect. Control 47, 2–8, doi: (2019).
Besselink, M. G. et al. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double-blind, placebo-controlled trial. Lancet 371, 651–659 (2008).
Hempel, S. et al. Safety of probiotics used to reduce risk and prevent or treat disease. Evid. Rep. Technol. Assess. (Full Rep.) 1–645 (2011).
Bafeta, A., Koh, M., Riveros, C. & Ravaud, P. Harms reporting in randomized controlled trials of interventions aimed at modifying microbiota: a systematic review. Ann. Intern. Med. 169, 240–247 (2018). .
US Food & Drug Administration. Development & approval process (drugs). https://www.fda.gov/drugs/developmentapprovalprocess/default.htm (2018).
Grazul, H., Kanda, L. L. & Gondek, D. Impact of probiotic supplements on microbiome diversity following antibiotic treatment of mice. Gut Microbes 7, 101–114 (2016).
Kabbani, T. A. et al. Prospective randomized controlled study on the effects of Saccharomyces boulardii CNCM I-745 and amoxicillin-clavulanate or the combination on the gut microbiota of healthy volunteers. Gut Microbes 8, 17–32 (2017).
De Wolfe, T. J. et al. Oral probiotic combination of Lactobacillus and Bifidobacterium alters the gastrointestinal microbiota during antibiotic treatment for Clostridium difficile infection. PLoS One 13, e0204253 (2018).
Brecht, M., Garg, A., Longstaff, K., Cooper, C. & Andersen, C. Lactobacillus sepsis following a laparotomy in a preterm infant: a note of caution. Neonatology 109, 186–189 (2016).
Spinler, J. K. et al. Administration of probiotic kefir to mice with Clostridium difficile infection exacerbates disease. Anaerobe 40, 54–57 (2016).
Oliveira, B. C. M. & Widmer, G. Probiotic product enhances susceptibility of mice to cryptosporidiosis. Appl. Environ. Microbiol. 84, e01408–18 (2018).
He, F. et al. Differences in composition and mucosal adhesion of Bifidobacteria isolated from healthy adults and healthy seniors. Curr. Microbiol. 43, 351–354 (2001).
Kankaanpää, P. E., Salminen, S. J., Isolauri, E. & Lee, Y. K. The influence of polyunsaturated fatty acids on probiotic growth and adhesion. FEMS Microbiol. Lett. 194, 149–153 (2001).
Shepherd, E. S., DeLoache, W. C., Pruss, K. M., Whitaker, W. R. & Sonnenburg, J. L. An exclusive metabolic niche enables strain engraftment in the gut microbiota. Nature 557, 434–438 (2018).
Andriantsoanirina, V., Teolis, A. C., Xin, L. X., Butel, M. J. & Aires, J. Bifidobacterium longum and Bifidobacterium breve isolates from preterm and full term neonates: comparison of cell surface properties. Anaerobe 28, 212–215 (2014).
Roessler, A. et al. The immune system in healthy adults and patients with atopic dermatitis seems to be affected differently by a probiotic intervention. Clin. Exp. Allergy 38, 93–102 (2008).
Pelto, L., Isolauri, E., Lilius, E. M., Nuutila, J. & Salminen, S. Probiotic bacteria down-regulate the milk-induced inflammatory response in milk-hypersensitive subjects but have an immunostimulatory effect in healthy subjects. Clin. Exp. Allergy 28, 1474–1479 (1998).
Hod, K. et al. The effect of a multispecies probiotic on microbiota composition in a clinical trial of patients with diarrhea-predominant irritable bowel syndrome. Neurogastroenterol. Motil. 30, e13456 (2018).
Suwal, S. et al. The probiotic effectiveness in preventing experimental colitis is correlated with host gut microbiota. Front. Microbiol. 9, 2675 (2018).
Abildgaard, A., et al. The antidepressant-like effect of probiotics and their faecal abundance may be modulated by the cohabiting gut microbiota in rats. Eur. Neuropsychopharmacol. 29, 98–110 (2019).
Ferrario, C. et al. Modulation of fecal Clostridiales bacteria and butyrate by probiotic intervention with Lactobacillus paracasei DG varies among healthy adults. J. Nutr. 144, 1787–1796 (2014).
Knight, R. et al. Best practices for analysing microbiomes. Nat. Rev. Microbiol. 16, 410–422 (2018).
Degirolamo, C., Rainaldi, S., Bovenga, F., Murzilli, S. & Moschetta, A. Microbiota modification with probiotics induces hepatic bile acid synthesis via downregulation of the Fxr–Fgf15 axis in mice. Cell Rep. 7, 12–18 (2014).
García-Albiach, R. et al. Molecular analysis of yogurt containing Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus in human intestinal microbiota. Am. J. Clin. Nutr. 87, 91–96 (2008).
Ouwehand, A. C. et al. Bifidobacterium microbiota and parameters of immune function in elderly subjects. FEMS Immunol. Med. Microbiol. 53, 18–25 (2008).
Korpela, K. et al. Probiotic supplementation restores normal microbiota composition and function in antibiotic-treated and in caesarean-born infants. Microbiome 6, 182 (2018).
Wang, C. et al. Intestinal microbiota profiles of healthy pre-school and school-age children and effects of probiotic supplementation. Ann. Nutr. Metab. 67, 257–266 (2015).
Mohan, R. et al. Effects of Bifidobacterium lactis Bb12 supplementation on intestinal microbiota of preterm infants: a double-blind, placebo-controlled, randomized study. J. Clin. Microbiol. 44, 4025–4031 (2006).
Veiga, P. et al. Changes of the human gut microbiome induced by a fermented milk product. Sci. Rep. 4, 6328 (2014).
Brahe, L. K. et al. Dietary modulation of the gut microbiota—a randomised controlled trial in obese postmenopausal women. Br. J. Nutr. 114, 406–417 (2015).
McNulty, N. P. et al. The impact of a consortium of fermented milk strains on the gut microbiome of gnotobiotic mice and monozygotic twins. Sci. Transl. Med. 3, 106ra106 (2011).
Eloe-Fadrosh, E. A. et al. Functional dynamics of the gut microbiome in elderly people during probiotic consumption. mBio 6, e00231-15 (2015).
Martin, F. P. et al. Probiotic modulation of symbiotic gut microbial–host metabolic interactions in a humanized microbiome mouse model. Mol. Syst. Biol. 4, 157 (2008).
Burton, K. J. et al. Probiotic yogurt and acidified milk similarly reduce postprandial inflammation and both alter the gut microbiota of healthy, young men. Br. J. Nutr. 117, 1312–1322 (2017).
Kajander, K. et al. Effects of multispecies probiotic supplementation on intestinal microbiota in irritable bowel syndrome. Aliment. Pharmacol. Ther. 26, 463–473 (2007).
Donaldson, G. P., Lee, S. M. & Mazmanian, S. K. Gut biogeography of the bacterial microbiota. Nat. Rev. Microbiol. 14, 20–32 (2016).
Uronis, J. M. et al. Gut microbial diversity is reduced by the probiotic VSL#3 and correlates with decreased TNBS-induced colitis. Inflamm. Bowel Dis. 17, 289–297 (2011).
Arthur, J. C. et al. VSL#3 probiotic modifies mucosal microbial composition but does not reduce colitis-associated colorectal cancer. Sci. Rep. 3, 2868 (2013).
Kühbacher, T. et al. Bacterial and fungal microbiota in relation to probiotic therapy (VSL#3) in pouchitis. Gut 55, 833–841 (2006).
Acknowledgements
We thank the members of the Elinav and Segal laboratories for discussions and apologize to authors whose work was not included due to space constraints. J.S. is the recipient of the Strauss Institute research fellowship. N.Z. is supported by the Gilead Sciences International Research Scholars Program in Liver Disease. E.S. is supported by the Crown Human Genome Center; the Else Kroener Fresenius Foundation; Donald L. Schwarz, Sherman Oaks, CA; Jack N. Halpern, NY, NY; Leesa Steinberg, Canada; and grants funded by the European Research Council and the Israel Science Foundation. E.E. is supported by Y. and R. Ungar, the Abisch Frenkel Foundation for the Promotion of Life Sciences, the Gurwin Family Fund for Scientific Research, the Leona M. and Harry B. Helmsley Charitable Trust, the Crown Endowment Fund for Immunological Research, the estate of J. Gitlitz, the estate of L. Hershkovich, the Benoziyo Endowment Fund for the Advancement of Science, the Adelis Foundation, J. L. and V. Schwartz, A. and G. Markovitz, A. and C. Adelson, the French National Center for Scientific Research (CNRS), D.L. Schwarz, the V.R. Schwartz Research Fellow Chair, L. Steinberg, J. N. Halpern, A. Edelheit, grants funded by the European Research Council, a Marie Curie Integration grant, the German-Israeli Foundation for Scientific Research and Development, the Israel Science Foundation, the Minerva Foundation, the Rising Tide Foundation, the Helmholtz Foundation, and the European Foundation for the Study of Diabetes.
Author information
Authors and Affiliations
Contributions
All authors have researched data for the article, made substantial contributions to discussion of content and wrote, reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Declaration of interests
E.S. and E.E. are paid consultants at DayTwo and BiomX. None of their work on microbial therapies is related to, funded or endorsed by, shared or discussed with or licensed to any commercial entity.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Suez, J., Zmora, N., Segal, E. et al. The pros, cons, and many unknowns of probiotics. Nat Med 25, 716–729 (2019). https://doi.org/10.1038/s41591-019-0439-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-019-0439-x
This article is cited by
-
Probiotic supplementation and risk of necrotizing enterocolitis and mortality among extremely preterm infants—the Probiotics in Extreme Prematurity in Scandinavia (PEPS) trial: study protocol for a multicenter, double-blinded, placebo-controlled, and registry-based randomized controlled trial
Trials (2024)
-
Does Torulaspora delbrueckii has some probiotic capabilities? In vitro and in vivo assessment
Nutrire (2024)
-
Intratumoural microbiota: a new frontier in cancer development and therapy
Signal Transduction and Targeted Therapy (2024)
-
Association between oral and fecal microbiome dysbiosis and treatment complications in pediatric patients undergoing allogeneic hematopoietic stem cell transplantation
Scientific Reports (2024)
-
From sustainable feedstocks to microbial foods
Nature Microbiology (2024)