Abstract
Stroke is the second leading cause of death worldwide and accounts for >2 million deaths annually in China1,2. Ischemic stroke (IS) and intracerebral hemorrhage (ICH) account for an equal number of deaths in China, despite a fourfold greater incidence of IS1,2. Stroke incidence and ICH proportion are higher in China than in Western populations3,4,5, despite having a lower mean low-density lipoprotein cholesterol (LDL-C) concentration. Observational studies reported weaker positive associations of LDL-C with IS than with coronary heart disease (CHD)6,7, but LDL-C-lowering trials demonstrated similar risk reductions for IS and CHD8,9,10. Mendelian randomization studies of LDL-C and IS have reported conflicting results11,12,13, and concerns about the excess risks of ICH associated with lowering LDL-C14,15 may have prevented the more widespread use of statins in China. We examined the associations of biochemically measured lipids with stroke in a nested case-control study in the China Kadoorie Biobank (CKB) and compared the risks for both stroke types associated with equivalent differences in LDL-C in Mendelian randomization analyses. The results demonstrated positive associations of LDL-C with IS and equally strong inverse associations with ICH, which were confirmed by genetic analyses and LDL-C-lowering trials. Lowering LDL-C is still likely to have net benefit for the prevention of overall stroke and cardiovascular disease in China.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
R.Clarke, D.B., L.L., and Z.C. have full access to all the study data and take responsibility for the integrity of the data and the accuracy of the data analysis. Data from the baseline survey, first resurvey, and cause-specific mortality are available to all bona fide researchers (www.ckbiobank.org). Additional data can also be made available on a collaborative basis by contacting the study investigators. All data requests are reviewed monthly by the CKB Data Access Committee, which is composed of senior scientists from Beijing and Oxford.
References
Roth, G. A. et al. Demographic and epidemiologic drivers of global cardiovascular mortality. N. Engl. J. Med. 372, 1333–1341 (2015).
Global Burden of Disease Study 2016. Global Burden of Disease Study 2016 (GBD 2016) (Institute for Health Metrics and Evaluation, Seattle, 2016).
Mathers, C. D. & Loncar, D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 3, e442 (2006).
Tsai, C. F., Thomas, B. & Sudlow, C. L. Epidemiology of stroke and its subtypes in Chinese vs white populations: a systematic review. Neurology 81, 264–272 (2013).
Zhou, M. et al. Cause-specific mortality for 240 causes in China during 1990–2013: a systematic subnational analysis for the Global Burden of Disease Study 2013. Lancet 387, 251–272 (2016).
Lewington, S. et al. Blood cholesterol and vascular mortality by age, sex, and blood pressure: a meta-analysis of individual data from 61 prospective studies with 55,000 vascular deaths. Lancet 370, 1829–1839 (2007).
Di Angelantonio, E. et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 302, 1993–2000 (2009).
Collins, R. et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet 388, 2532–2561 (2016).
Sabatine, M. S. et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N. Engl. J. Med. 376, 1713–1722 (2017).
Ridker, P. M. LDL cholesterol: controversies and future therapeutic directions. Lancet 384, 607–617 (2014).
Hopewell, J., Stari, T., Parish, S., Collins, R. & Clarke, R. The impact of genetic variants related to LDL-cholesterol on risk of ischemic stroke and coronary heart disease. Circulation 126, abstr. 11959 (2012).
Ference, B. A. et al. Variation in PCSK9 and HMGCR and risk of cardiovascular disease and diabetes. N. Engl. J. Med. 375, 2144–2153 (2016).
Hopewell, J. C. et al. Differential effects of PCSK9 variants on risk of coronary disease and ischaemic stroke. Eur. Heart J. 39, 354–359 (2018).
Amarenco, P. et al. High-dose atorvastatin after stroke or transient ischemic attack. N. Engl. J. Med. 355, 549–559 (2006).
Fulcher, J. et al. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet 385, 1397–1405 (2015).
Do, R. et al. Common variants associated with plasma triglycerides and risk for coronary artery disease. Nat. Genet. 45, 1345–1352 (2013).
Willer, C. J. et al. Discovery and refinement of loci associated with lipid levels. Nat. Genet. 45, 1274–1283 (2013).
Palmer, T. M. et al. Instrumental variable estimation of causal risk ratios and causal odds ratios in Mendelian randomization analyses. Am. J. Epidemiol. 173, 1392–1403 (2011).
Chen, Z. et al. Serum cholesterol concentration and coronary heart disease in population with low cholesterol concentrations. BMJ 303, 276–282 (1991).
Baigent, C. et al. The effects of lowering LDL cholesterol with simvastatin plus ezetimibe in patients with chronic kidney disease (Study of Heart and Renal Protection): a randomised placebo-controlled trial. Lancet 377, 2181–2192 (2011).
Cannon, C. P. et al. Ezetimibe added to statin therapy after acute coronary syndromes. N. Engl. J. Med. 372, 2387–2397 (2015).
Zhang, X. et al. Cholesterol, coronary heart disease, and stroke in the Asia Pacific region. Int. J. Epidemiol. 32, 563–572 (2003).
Hindy, G. et al. Role of blood lipids in the development of ischemic stroke and its subtypes: a Mendelian randomization study. Stroke 49, 820–827 (2018).
Rao, A. S. et al. Large-scale phenome-wide association study of PCSK9 variants demonstrates protection against ischemic stroke. Circ. Genom. Precis. Med. 11, e002162 (2018).
Iso, H., Jacobs, D. R. Jr., Wentworth, D., Neaton, J. D. & Cohen, J. D. Serum cholesterol levels and six-year mortality from stroke in 350,977 men screened for the multiple risk factor intervention trial. N. Engl. J. Med. 320, 904–910 (1989).
Ebrahim, S. et al. Serum cholesterol, haemorrhagic stroke, ischaemic stroke, and myocardial infarction: Korean national health system prospective cohort study. BMJ 333, 22 (2006).
Peto, R. Current misconception 3: that subgroup-specific trial mortality results often provide a good basis for individualising patient care. Br. J. Cancer 104, 1057–1058 (2011).
Sever, P. S. et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial—Lipid Lowering Arm (ASCOT-LLA): a multicentre randomised controlled trial. Lancet 361, 1149–1158 (2003).
Ooneda, G. et al. Smooth muscle cells in the development of plasmatic arterionecrosis, arteriosclerosis, and arterial contraction. Blood Vessels 15, 148–156 (1978).
Konishi, M. et al. Associations of serum total cholesterol, different types of stroke, and stenosis distribution of cerebral arteries. The Akita Pathology Study. Stroke 24, 954–964 (1993).
MacKenzie, J. M. Intracerebral haemorrhage. J. Clin. Pathol. 49, 360–364 (1996).
Yusuf, S. et al. Use of secondary prevention drugs for cardiovascular disease in the community in high-income, middle-income, and low-income countries (the PURE Study): a prospective epidemiological survey. Lancet 378, 1231–1243 (2011).
Liu, M. et al. Stroke in China: epidemiology, prevention, and management strategies. Lancet Neurol. 6, 456–464 (2007).
Wang, W. et al. Prevalence, incidence and mortality of stroke in china: results from a nationwide population-based survey of 480,687 adults. Circulation 135, 759–771 (2017).
Zhao, D. et al. Epidemiological transition of stroke in China: twenty-one-year observational study from the Sino-MONICA-Beijing Project. Stroke 39, 1668–1674 (2008).
Chen, Z. et al. Cohort profile: the Kadoorie Study of Chronic Disease in China (KSCDC). Int. J. Epidemiol. 34, 1243–1249 (2005).
Chen, Z. et al. China Kadoorie Biobank of 0.5 million people: survey methods, baseline characteristics and long-term follow-up. Int. J. Epidemiol. 40, 1652–1666 (2011).
Clarke, R. et al. Underestimation of risk associations due to regression dilution in long-term follow-up of prospective studies. Am. J. Epidemiol. 150, 341–353 (1999).
Peto, R. The marked differences between carotenoids and retinoids: methodological implications for biochemical epidemiology. Cancer Surv. 2, 327–340 (1983).
Athyros, V. G. et al. Treatment with atorvastatin to the National Cholesterol Educational Program goal versus ‘usual’ care in secondary coronary heart disease prevention. The GREek Atorvastatin and Coronary-heart-disease Evaluation (GREACE) study.Curr. Med. Res. Opin. 18, 220–228 (2002).
Rossebø, A. B. et al. Intensive lipid lowering with simvastatin and ezetimibe in aortic stenosis. N. Engl. J. Med. 359, 1343–1356 (2008).
Yusuf, S. et al. Cholesterol lowering in intermediate-risk persons without cardiovascular disease. N. Engl. J. Med. 374, 2021–2031 (2016).
Schwartz, G. G. et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N. Engl. J. Med. 379, 2097–2107 (2018).
Easton, D. F., Peto, J. & Babiker, A. G. Floating absolute risk: an alternative to relative risk in survival and case-control analysis avoiding an arbitrary reference group. Stat. Med. 10, 1025–1035 (1991).
Di Angelantonio, E. et al. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 388, 776–786 (2016).
Wald, A. The fitting of straight lines if both variables are subject to error.Ann. Math. Statist. 11, 284–300 (1940).
Yusuf, S., Peto, R., Lewis, J., Collins, R. & Sleight, P. Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog. Cardiovasc. Dis. 27, 335–371 (1985).
Lewington, S. et al. The burden of hypertension and associated risk for cardiovascular mortality in china. JAMA Intern. Med. 176, 524–532 (2016).
Acknowledgements
We wish to thank the participants, project staff, and staff of the China Center for Disease Control and its regional offices for access to death and disease registries. The Chinese National Health Insurance scheme provided electronic linkage to all hospitalization data. The CKB study is jointly coordinated by the University of Oxford and the Chinese Academy of Medical Sciences. The funding body for the baseline survey was the Kadoorie Charitable Foundation, Hong Kong, China. Z.C. was funded for the long-term continuation of the study by Wellcome Trust grants (nos. 202922/Z/16/Z, 104085/Z/14/Z, and 088158/Z/09/Z). L.L. was funded by the National Natural Science Foundation of China (grant nos. 81390540, 81390541, and 81390544) and the National Key Research and Development Program of China (grant nos. 2016YFC0900500, 2016YFC0900501, 2016YFC0900504, and 2016YFC1303904). Core funding was also provided to the Clinical Trial Service Unit, University of Oxford, by the British Heart Foundation, the UK Medical Research Council, and Cancer Research UK. L.S. received a Clarendon Scholarship from the University of Oxford. The funders played no role in the design or conduct of the study, including data collection, management, analysis, or interpretation of the results; the preparation, review, or approval of the manuscript; or the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Consortia
Contributions
L.S., R.Clarke, D.B., R.P., and Z.C. designed and planned the analysis and manuscript. L.S. performed the data analyses and wrote the first draft of the manuscript. R.Clarke, D.B., S.P., R.P., and Z.C. provided scientific interpretation of the results and revised the manuscript. R.Clarke, Z.C., L.L., R.P., R.Collins, R.W., J.L., and J.C., as members of the CKB steering committee, designed and supervised the overall conduct of the study and obtained the funding. Y.G., Y.C., Z.B., C.Y., and Z.C. coordinated the data acquisition (for baseline, resurveys, and long-term follow-up). R.W., Y.G., I.M., Z.B., and M.H. coordinated the genotyping analyses in China and the laboratory analyses in Oxford. All authors provided critical comments on the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Effect of progressive adjustment for potential confounders on the risk of IS and ICH with usual LDL-C.
Cox regression was used to estimate adjusted RRs (95% confidence intervals (CI)) for the risk of different stroke types per 1 mmol l−1 higher concentrations of usual LDL-C. Each square has an area inversely proportional to the variance of the log risk. The horizontal lines represent the 95% CIs.
Extended Data Fig. 2 Associations of usual LDL-C with risk of IS and ICH in population subgroups at baseline.
Cox regression was used to estimate the adjusted RRs (95% CIs) for the risk of different stroke types per 1 mmol l−1 higher concentrations of usual LDL-C. Chi-squared tests were used to assess heterogeneity and trend; the d.f. are provided as subscripts. All two-sided P values were uncorrected for multiple testing. Symbols and conventions as in Extended Data Fig. 1.
Extended Data Fig. 3 Adjusted RRs for the risk of IS by usual concentrations of LDL-C and HDL-C in observational analyses in the CKB.
Symbols and conventions as in Extended Data Fig. 1. The number of IS cases and controls were 5,475 and 6,290, respectively.
Extended Data Fig. 4 Adjusted RRs for the risk of IS and ICH by usual concentrations of apolipoprotein B and A1, and lipoprotein(a) in observational analyses in the CKB.
a–c, Cox regression was used to estimate the RRs (95% CIs) for IS (N = 5,475) and ICH (N = 4,776) by fifths of usual apolipoprotein B (a), usual apolipoprotein A1 (b), and usual lipoprotein(a) (c), respectively. The line represents the slope from a weighted linear regression with the weights based on the inverse variance of the log RR. Symbols and conventions as in Extended Data Fig. 1.
Extended Data Fig. 5 Associations of the GRS for LDL-C with major vascular risk factors.
The analyses were conducted in 17,567 CKB participants with available data, adjusted for sex, age, age2, and case status. General linear regression was used to estimate s.d. differences in all traits (after rank inverse normal transformation) per 1 s.d. higher GRS. All two-sided P values were uncorrected for multiple testing.
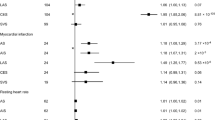
Extended Data Fig. 6 Meta-analysis of randomized trials of LDL-C-lowering treatment with statins, ezetimibe, or PCSK9 inhibitor and risk of IS and ICH.
Study-specific RRs (95% CI) were obtained from the published results of the LDL-C-lowering trials. The overall RRs (95% CIs) were obtained by inverse variance-weighted meta-analysis of the study-specific RRs per 1 mmol l−1 lower LDL-C concentration.
Supplementary information
Supplementary Information
Supplementary Tables 1–4
Rights and permissions
About this article
Cite this article
Sun, L., Clarke, R., Bennett, D. et al. Causal associations of blood lipids with risk of ischemic stroke and intracerebral hemorrhage in Chinese adults. Nat Med 25, 569–574 (2019). https://doi.org/10.1038/s41591-019-0366-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-019-0366-x
This article is cited by
-
Dietary amino acids and risk of stroke subtypes: a prospective analysis of 356,000 participants in seven European countries
European Journal of Nutrition (2024)
-
Effects of air pollution control policies on intracerebral hemorrhage mortality among residents in Tianjin, China
BMC Public Health (2023)
-
A nested case–control study investigating short-term prognosis, clinical and imaging features in acute ischemic stroke patients with systemic lupus erythematosus
Acta Neurologica Belgica (2023)
-
Therapeutic Potentials of MicroRNA-126 in Cerebral Ischemia
Molecular Neurobiology (2023)
-
Identification of three potential novel biomarkers for early diagnosis of acute ischemic stroke via plasma lipidomics
Metabolomics (2023)