Abstract
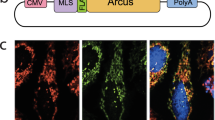
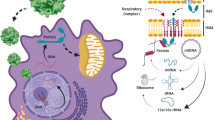
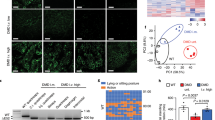
Mutations in the mitochondrial DNA (mtDNA) are responsible for several metabolic disorders, commonly involving muscle and the central nervous system1. Because of the critical role of mtDNA in oxidative phosphorylation, the majority of pathogenic mtDNA mutations are heteroplasmic, co-existing with wild-type molecules1. Using a mouse model with a heteroplasmic mtDNA mutation2, we tested whether mitochondrial-targeted TALENs (mitoTALENs)3,4 could reduce the mutant mtDNA load in muscle and heart. AAV9-mitoTALEN was administered via intramuscular, intravenous, and intraperitoneal injections. Muscle and heart were efficiently transduced and showed a robust reduction in mutant mtDNA, which was stable over time. The molecular defect, namely a decrease in transfer RNAAla levels, was restored by the treatment. These results showed that mitoTALENs, when expressed in affected tissues, could revert disease-related phenotypes in mice.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All the data contained in this manuscript is available to investigators.
Change history
05 October 2018
In the version of this article originally published, there was an error in Fig. 1a. The m.5024C>T mutation, shown as a green T, was displaced by one base. The error has been corrected in the print, HTML and PDF versions of this article.
References
Craven, L., Alston, C. L., Taylor, R. W. & Turnbull, D. M. Recent advances in mitochondrial disease. Annu. Rev. Genomics Hum. Genet. 18, 257–275 (2017).
Kauppila, J. H. et al. A phenotype-driven approach to generate mouse models with pathogenic mtDNA mutations causing mitochondrial disease. Cell Rep. 16, 2980–2990 (2016).
Bacman, S. R., Williams, S. L., Pinto, M., Peralta, S. & Moraes, C. T. Specific elimination of mutant mitochondrial genomes in patient-derived cells by mitoTALENs. Nat. Med. 19, 1111–1113 (2013).
Hashimoto, M. et al. MitoTALEN: a general approach to reduce mutant mtDNA Loads and restore oxidative phosphorylation function in mitochondrial diseases. Mol. Ther. 23, 1592–1599 (2015).
Gammage, P. A. et al. Near-complete elimination of mutant mtDNA by iterative or dynamic dose-controlled treatment with mtZFNs. Nucleic Acids Res. 44, 7804–7816 (2016).
Lehmann, D. et al. Pathogenic mitochondrial mt-tRNA(Ala) variants are uniquely associated with isolated myopathy. Eur. J. Hum. Genet. 23, 1735–1738 (2015).
Cermak, T. et al. Efficient design and assembly of custom TALEN and other TAL effector-based constructs for DNA targeting. Nucleic Acids Res. 39, e82 (2011).
Miller, J. C. et al. An improved zinc-finger nuclease architecture for highly specific genome editing. Nat. Biotechnol. 25, 778–785 (2007).
Li, T. et al. TAL nucleases (TALNs): hybrid proteins composed of TAL effectors and FokI DNA-cleavage domain. Nucleic Acids Res. 39, 359–372 (2011).
Szymczak, A. L. et al. Correction of multi-gene deficiency in vivo using a single ‘self-cleaving’ 2A peptide-based retroviral vector. Nat. Biotechnol. 22, 589–594 (2004).
Yardeni, T., Eckhaus, M., Morris, H. D., Huizing, M. & Hoogstraten-Miller, S. Retro-orbital injections in mice. Lab. Anim. (NY) 40, 155–160 (2011).
Srivastava, S. & Moraes, C. T. Double-strand breaks of mouse muscle mtDNA promote large deletions similar to multiple mtDNA deletions in humans. Hum. Mol. Genet. 14, 893–902 (2005).
Fukui, H. & Moraes, C. T. Mechanisms of formation and accumulation of mitochondrial DNA deletions in aging neurons. Hum. Mol. Genet. 18, 1028–1036 (2009).
Bacman, S. R., Williams, S. L. & Moraes, C. T. Intra- and inter-molecular recombination of mitochondrial DNA after in vivo induction of multiple double-strand breaks. Nucleic Acids Res. 37, 4218–4226 (2009).
El-Hattab, A. W. & Scaglia, F. Mitochondrial cardiomyopathies. Front. Cardiovasc. Med. 3, 25 (2016).
Kelly, R. D., Mahmud, A., McKenzie, M., Trounce, I. A. & St John, J. C. Mitochondrial DNA copy number is regulated in a tissue specific manner by DNA methylation of the nuclear-encoded DNA polymerase gamma A. Nucleic Acids Res. 40, 10124–10138 (2012).
Nissanka, N., Bacman, S. R., Plastini, M. J. & Moraes, C. T. The mitochondrial DNA polymerase gamma degrades linear DNA fragments precluding the formation of deletions. Nat. Commun. 9, 2491 (2018).
Peeva, V. et al. Linear mitochondrial DNA is rapidly degraded by components of the replication machinery. Nat. Commun. 9, 1727 (2018).
Gammage, P. A. et al. Genome editing in mitochondria corrects a pathogenic mtDNA mutation in vivo. Nat. Med. https://doi.org/10.1038/s41591-018-0165-9 (2018).
Petruzzella, V. et al. Extremely high levels of mutant mtDNAs co-localize with cytochrome c oxidase-negative ragged-red fibers in patients harboring a point mutation at nt 3243. Hum. Mol. Genet. 3, 449–454 (1994).
Doyon, Y. et al. Enhancing zinc-finger-nuclease activity with improved obligate heterodimeric architectures. Nat. Methods 8, 74–79 (2011).
Lochmuller, H., Johns, T. & Shoubridge, E. A. Expression of the E6 and E7 genes of human papillomavirus (HPV16) extends the life span of human myoblasts. Exp. Cell Res. 248, 186–193 (1999).
Lamb, B. M., Mercer, A. C. & Barbas, C. F. 3rd Directed evolution of the TALE N-terminal domain for recognition of all 5’ bases. Nucleic Acids Res. 41, 9779–9785 (2013).
Bacman, S. R., Williams, S. L., Garcia, S. & Moraes, C. T. Organ-specific shifts in mtDNA heteroplasmy following systemic delivery of a mitochondria-targeted restriction endonuclease. Gene Ther. 17, 713–720 (2010).
Moraes, C. T., Ricci, E., Bonilla, E., DiMauro, S. & Schon, E. A. The mitochondrial tRNA(Leu(UUR)) mutation in mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes (MELAS): genetic, biochemical, and morphological correlations in skeletal muscle. Am. J. Hum. Genet. 50, 934–949 (1992).
Pinto, M., Nissanka, N. & Moraes, C. T. Lack of parkin anticipates the phenotype and affects mitochondrial morphology and mtDNA levels in a mouse model of Parkinson’s disease. J. Neurosci. 38, 1042–1053 (2018).
Davies, S. M. et al. MRPS27 is a pentatricopeptide repeat domain protein required for the translation of mitochondrially encoded proteins. FEBS Lett. 586, 3555–3561 (2012).
Acknowledgements
This work was supported primarily by the National Institutes of Health Grant 5R01EY010804, with additional support from 1R01AG036871 and 1R01NS079965 (NIH) and the Muscular Dystrophy Association (CTM). We also acknowledge support from The JDM Fund for Mitochondrial Research and the Biscardi family. N.N. is supported by an American Heart Association predoctoral fellowship (16PRE30480009). We are grateful to the University of Miami Flow Cytometry core facility (SCCC) for expert assistance.
Author information
Authors and Affiliations
Contributions
S.R.B. was involved with the concept, design and execution of the cell and mouse experiments, and wrote and revised the manuscript. J.H.K.K. characterized the mouse model and the tRNA alanine northern experiments. C.V.P. produced and characterized the cell lines with high levels of the m.5024C>T mtDNA mutation. N.N. performed the experiments with mouse and the detection of mtDNA deletions. M.M. performed the tRNA alanine northern experiments. M.P. prepared the positive controls for mtDNA deletions. S.L.W. was involved with the concept, troubleshooting and recombinant virus administration. N.-G.L. characterized the mouse model. J.B.S. characterized the mouse model and the tRNA alanine northern experiments. C.T.M. was involved with the concept and design, and also wrote and revised the manuscript. All authors read and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8
Rights and permissions
About this article
Cite this article
Bacman, S.R., Kauppila, J.H.K., Pereira, C.V. et al. MitoTALEN reduces mutant mtDNA load and restores tRNAAla levels in a mouse model of heteroplasmic mtDNA mutation. Nat Med 24, 1696–1700 (2018). https://doi.org/10.1038/s41591-018-0166-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0166-8
This article is cited by
-
Focusing on mitochondria in the brain: from biology to therapeutics
Translational Neurodegeneration (2024)
-
Strand-selective base editing of human mitochondrial DNA using mitoBEs
Nature Biotechnology (2024)
-
One-pot, degradable, silica nanocarriers with encapsulated oligonucleotides for mitochondrial specific targeting
Discover Nano (2023)
-
Mitochondrial gene editing
Nature Reviews Methods Primers (2023)
-
Correction of a homoplasmic mitochondrial tRNA mutation in patient-derived iPSCs via a mitochondrial base editor
Communications Biology (2023)