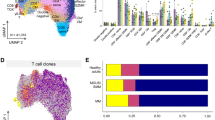
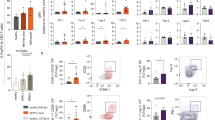
Abstract
T cell dysfunction contributes to tumor immune escape in patients with cancer and is particularly severe amidst glioblastoma (GBM). Among other defects, T cell lymphopenia is characteristic, yet often attributed to treatment. We reveal that even treatment-naïve subjects and mice with GBM can harbor AIDS-level CD4 counts, as well as contracted, T cell–deficient lymphoid organs. Missing naïve T cells are instead found sequestered in large numbers in the bone marrow. This phenomenon characterizes not only GBM but a variety of other cancers, although only when tumors are introduced into the intracranial compartment. T cell sequestration is accompanied by tumor-imposed loss of S1P1 from the T cell surface and is reversible upon precluding S1P1 internalization. In murine models of GBM, hindering S1P1 internalization and reversing sequestration licenses T cell–activating therapies that were previously ineffective. Sequestration of T cells in bone marrow is therefore a tumor-adaptive mode of T cell dysfunction, whose reversal may constitute a promising immunotherapeutic adjunct.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
22 January 2019
In the version of this article originally published, the figure callout in this sentence was incorrect: “Furthermore, in S1P1-KI mice themselves, whereas PD-1 blockade was ineffectual as monotherapy, the effects of 4-1BB agonism and checkpoint blockade proved additive, with the combination prolonging median survival and producing a 50% long-term survival rate (Fig. 6f).” The callout should have been to Supplementary Fig. 6b. The error has been corrected in the PDF and HTML versions of the article.
References
Dunn, G. P., Bruce, A. T., Ikeda, H., Old, L. J. & Schreiber, R. D. Cancer immunoediting: from immunosurveillance to tumor escape. Nat. Immunol. 3, 991–998 (2002).
Dunn, G. P., Old, L. J. & Schreiber, R. D. The three Es of cancer immunoediting. Annu. Rev. Immunol. 22, 329–360 (2004).
Dix, A. R., Brooks, W. H., Roszman, T. L. & Morford, L. A. Immune defects observed in patients with primary malignant brain tumors. J. Neuroimmunol. 100, 216–232 (1999).
Dunn, G. P., Fecci, P. E. & Curry, W. T. Cancer immunoediting in malignant glioma. Neurosurgery 71, 201–222 (2012).
Fecci, P. E., Heimberger, A. B. & Sampson, J. H. Immunotherapy for primary brain tumors: no longer a matter of privilege. Clin. Cancer Res. 20, 5620–5629 (2014).
Woroniecka, K. I., Rhodin, K. E., Chongsathidkiet, P., Keith, K. A. & Fecci, P. E. T-cell dysfunction in glioblastoma: applying a new framework. Clin. Cancer Res. 24, 1–11 (2018).
Ostrom, Q. T. et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2010-2014. Neuro Oncol. 19, v1–v88 (2017).
Brooks, W. H., Roszman, T. L., Mahaley, M. S. & Woosley, R. E. Immunobiology of primary intracranial tumours. II. Analysis of lymphocyte subpopulations in patients with primary brain tumours. Clin. Exp. Immunol. 29, 61–66 (1977).
Garris, C. S., Blaho, V. A., Hla, T. & Han, M. H. Sphingosine-1-phosphate receptor 1 signalling in T cells: trafficking and beyond. Immunology 142, 347–353 (2014).
Rosen, H., Gonzalez-Cabrera, P. J., Sanna, M. G. & Brown, S. Sphingosine 1-phosphate receptor signaling. Annu. Rev. Biochem. 78, 743–768 (2009).
Matloubian, M. et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427, 355–360 (2004).
Drouillard, A. et al. Human naive and memory T cells display opposite migratory responses to sphingosine-1 phosphate. J. Immunol. 200, 551–557 (2018).
Spiegel, S. & Milstien, S. Sphingosine-1-phosphate: an enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 4, 397–407 (2003).
Schwab, S. R. et al. Lymphocyte sequestration through S1P lyase inhibition and disruption of S1P gradients. Science 309, 1735–1739 (2005).
Cyster, J. G. Chemokines, sphingosine-1-phosphate, and cell migration in secondary lymphoid organs. Annu. Rev. Immunol. 23, 127–159 (2005).
Lo, C. G., Xu, Y., Proia, R. L. & Cyster, J. G. Cyclical modulation of sphingosine-1-phosphate receptor 1 surface expression during lymphocyte recirculation and relationship to lymphoid organ transit. J. Exp. Med. 201, 291–301 (2005).
Zajac, A. J. & Harrington, L. E. Tissue-resident T cells lose their S1P1 exit visas. Cell. Mol. Immunol. 11, 221–223 (2014).
Maeda, Y., Seki, N., Sato, N., Sugahara, K. & Chiba, K. Sphingosine 1-phosphate receptor type 1 regulates egress of mature T cells from mouse bone marrow. Int. Immunol. 22, 515–525 (2010).
Blaho, V. A. et al. HDL-bound sphingosine-1-phosphate restrains lymphopoiesis and neuroinflammation. Nature 523, 342–346 (2015).
Allende, M. L., Yamashita, T. & Proia, R. L. G-protein-coupled receptor S1P1 acts within endothelial cells to regulate vascular maturation. Blood 102, 3665–3667 (2003).
Arnon, T. I. et al. GRK2-dependent S1PR1 desensitization is required for lymphocytes to overcome their attraction to blood. Science 333, 1898–1903 (2011).
Hailemichael, Y. et al. Persistent antigen at vaccination sites induces tumor-specific CD8(+) T cell sequestration, dysfunction and deletion. Nat. Med. 19, 465–472 (2013).
Schietinger, A. & Greenberg, P. D. Tolerance and exhaustion: defining mechanisms of T cell dysfunction. Trends Immunol. 35, 51–60 (2014).
Chi, H. & Flavell, R. A. Cutting edge: regulation of T cell trafficking and primary immune responses by sphingosine 1-phosphate receptor 1. J. Immunol. 174, 2485–2488 (2005).
Skon, C. N. et al. Transcriptional downregulation of S1pr1 is required for the establishment of resident memory CD8+ T cells. Nat. Immunol. 14, 1285–1293 (2013).
Liu, G., Yang, K., Burns, S., Shrestha, S. & Chi, H. The S1P(1)-mTOR axis directs the reciprocal differentiation of T(H)1 and T(reg) cells. Nat. Immunol. 11, 1047–1056 (2010).
Garris, C. S. et al. Defective sphingosine 1-phosphate receptor 1 (S1P1) phosphorylation exacerbates TH17-mediated autoimmune neuroinflammation. Nat. Immunol. 14, 1166–1172 (2013).
Lee, H. et al. STAT3-induced S1PR1 expression is crucial for persistent STAT3 activation in tumors. Nat. Med. 16, 1421–1428 (2010).
Deng, H. et al. Discovery of clinical candidate GSK1842799 as a selective S1P1 receptor agonist (prodrug) for multiple sclerosis. ACS Med. Chem. Lett. 4, 942–947 (2013).
Jin, J. et al. Development of a selective S1P1 receptor agonist, Syl930, as a potential therapeutic agent for autoimmune encephalitis. Biochem. Pharmacol. 90, 50–61 (2014).
Yamamoto, R. et al. ASP4058, a novel agonist for sphingosine 1-phosphate receptors 1 and 5, ameliorates rodent experimental autoimmune encephalomyelitis with a favorable safety profile. PloS ONE 9, e110819 (2014).
Reeves, P. M., Kang, Y. L. & Kirchhausen, T. Endocytosis of ligand-activated sphingosine 1-phosphate receptor 1 mediated by the clathrin-pathway. Traffic 17, 40–52 (2016).
Park, C. O. & Kupper, T. S. The emerging role of resident memory T cells in protective immunity and inflammatory disease. Nat. Med. 21, 688–697 (2015).
Wakim, L. M. et al. The molecular signature of tissue resident memory CD8 T cells isolated from the brain. J. Immunol. 189, 3462–3471 (2012).
Abuhusain, H. J. et al. A metabolic shift favoring sphingosine 1-phosphate at the expense of ceramide controls glioblastoma angiogenesis. J. Biol. Chem. 288, 37355–37364 (2013).
Omuro, A. et al. Nivolumab with or without ipilimumab in patients with recurrent glioblastoma: Results from exploratory phase 1 cohorts of CheckMate 143. Neuro Oncol. 20, 674–686 (2018).
Brooks, W. H. et al. Immunobiology of primary intracranial tumors. Part 5: Correlation of a lymphocyte index and clinical status. J. Neurosurg. 54, 331–337 (1981).
Fecci, P. E. et al. Increased regulatory T-cell fraction amidst a diminished CD4 compartment explains cellular immune defects in patients with malignant glioma. Cancer Res. 66, 3294–3302 (2006).
Morford, L. A., Dix, A. R., Brooks, W. H. & Roszman, T. L. Apoptotic elimination of peripheral T lymphocytes in patients with primary intracranial tumors. J. Neurosurg. 91, 935–946 (1999).
Hughes, M. A., Parisi, M., Grossman, S. & Kleinberg, L. Primary brain tumors treated with steroids and radiotherapy: low CD4 counts and risk of infection. Int. J. Radiat. Oncol. Biol. Phys. 62, 1423–1426 (2005).
Grossman, S. A. et al. Immunosuppression in patients with high-grade gliomas treated with radiation and temozolomide. Clin. Cancer Res. 17, 5473–5480 (2011).
Mahaley, M. S. Jr. et al. Immunobiology of primary intracranial tumors. Part 1: Studies of the cellular and humoral general immune competence of brain-tumor patients. J. Neurosurg. 46, 467–476 (1977).
Jackson, C. M. et al. Systemic tolerance mediated by melanoma brain tumors is reversible by radiotherapy and vaccination. Clin. Cancer Res. 22, 1161–1172 (2016).
Ostrom, Q. T. et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol. 16(Suppl.), iv1–iv63 (2014).
Frej, C. et al. Quantification of sphingosine 1-phosphate by validated LC-MS/MS method revealing strong correlation with apolipoprotein M in plasma but not in serum due to platelet activation during blood coagulation. Anal. Bioanal. Chem. 407, 8533–8542 (2015).
Acknowledgements
We would like to thank G.E. Archer, K.A. Batich, T.A. Chewning, K.L. Congdon, K.A. Keith, R.J. Lefkowitz, P.K. Norberg, E.A. Reap, L.A.M. Rein, K.E. Rhodin, A. Seas, S.H. Shen, R.J. Schmittling, D.J. Snyder, C.M. Suryadevara, A.M. Swartz, W.H. Tomaszewski, D.S. Wilkinson, W. Xie, H. Yang, and members of the Duke Brain Tumor Immunotherapy Program for their insights throughout the study. We would like to thank M. Foster and J.W. Thompson from the Proteomics and Metabolomics Shared Resource, Duke Center for Genomic and Computational Biology for help with liquid chromatography-tandem mass spectrometry analyses. We would like to thank M.R. Llewellyn for the preparation of medical illustrations. This work was supported by institutional start-up funds from Duke University Medical Center, The Sontag Foundation Distinguished Scientist Award, and the National Institutes of Health R01NS099096 to P.E.F.
Author information
Authors and Affiliations
Contributions
P.C., C.J., S.K., F.L., N.C.S., J.-V.C., B.V.N., and P.E.F. obtained and/or analyzed human data. P.C., C.J., S.K., X.C., S.H.F., K.W., A.A.E., C.A.D., H.R.K., L.S.-P., T.A.C., and P.E.F. designed, carried out, and/or analyzed in vitro and in vivo animal experiments. J.I.E. provided pathological characterization and immunohistochemistry analyses. J.E.H. provided biostatistics consultation. J.H.S., M.D.G., R.L.M., G.D., W.T.C., and P.E.F. provided feedback and supervised all research. P.C., C.A.D., and P.E.F. wrote the manuscript. All authors read, revised, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1 and 2
Rights and permissions
About this article
Cite this article
Chongsathidkiet, P., Jackson, C., Koyama, S. et al. Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors. Nat Med 24, 1459–1468 (2018). https://doi.org/10.1038/s41591-018-0135-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0135-2
This article is cited by
-
Systematic review and pooled analysis of the impact of treatment-induced lymphopenia on survival of glioblastoma patients
Radiation Oncology (2024)
-
Novel Clinical Trials and Approaches in the Management of Glioblastoma
Current Oncology Reports (2024)
-
Immunotherapy for Brain Tumors: Where We Have Been, and Where Do We Go From Here?
Current Treatment Options in Oncology (2024)
-
Strategies to Improve Drug Delivery Across the Blood–Brain Barrier for Glioblastoma
Current Neurology and Neuroscience Reports (2024)
-
Tumor-Wirtszellen-Interaktion im Mikromilieu: neue Angriffspunkte für die Therapie?
Der Nervenarzt (2024)