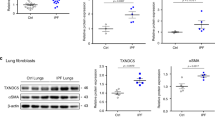
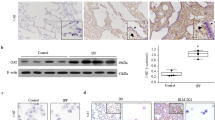
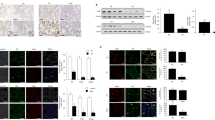
Abstract
Idiopathic pulmonary fibrosis is characterized by excessive deposition of collagen in the lung, leading to chronically impaired gas exchange and death1,2,3. Oxidative stress is believed to be critical in this disease pathogenesis4,5,6, although the exact mechanisms remain enigmatic. Protein S-glutathionylation (PSSG) is a post-translational modification of proteins that can be reversed by glutaredoxin-1 (GLRX)7. It remains unknown whether GLRX and PSSG play a role in lung fibrosis. Here, we explored the impact of GLRX and PSSG status on the pathogenesis of pulmonary fibrosis, using lung tissues from subjects with idiopathic pulmonary fibrosis, transgenic mouse models and direct administration of recombinant Glrx to airways of mice with existing fibrosis. We demonstrate that GLRX enzymatic activity was strongly decreased in fibrotic lungs, in accordance with increases in PSSG. Mice lacking Glrx were far more susceptible to bleomycin- or adenovirus encoding active transforming growth factor beta-1 (AdTGFB1)-induced pulmonary fibrosis, whereas transgenic overexpression of Glrx in the lung epithelium attenuated fibrosis. We furthermore show that endogenous GLRX was inactivated through an oxidative mechanism and that direct administration of the Glrx protein into airways augmented Glrx activity and reversed increases in collagen in mice with TGFB1- or bleomycin-induced fibrosis, even when administered to fibrotic, aged animals. Collectively, these findings suggest the therapeutic potential of exogenous GLRX in treating lung fibrosis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
du Bois, R. M. Strategies for treating idiopathic pulmonary fibrosis. Nat. Rev. Drug Discov. 9, 129–140 (2010).
Barkauskas, C. E. & Noble, P. W. Cellular mechanisms of tissue fibrosis. 7. New insights into the cellular mechanisms of pulmonary fibrosis. Am. J. Physiol. Cell Physiol. 306, C987–996 (2014).
Raghu, G. et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am. J. Respir. Crit. Care Med. 183, 788–824 (2011).
Kinnula, V. L. & Myllarniemi, M. Oxidant–antioxidant imbalance as a potential contributor to the progression of human pulmonary fibrosis. Antioxid. Redox Signal. 10, 727–738 (2008).
Janssen-Heininger, Y. M. et al. Regulation of apoptosis through cysteine oxidation: implications for fibrotic lung disease. Ann. NY Acad. Sci. 1203, 23–28 (2010).
Kurundkar, A. & Thannickal, V. J. Redox mechanisms in age-related lung fibrosis. Redox Biol. 9, 67–76 (2016).
Mieyal, J. J., Gallogly, M. M., Qanungo, S., Sabens, E. A. & Shelton, M. D. Molecular mechanisms and clinical implications of reversible protein S-glutathionylation. Antioxid. Redox Signal. 10, 1941–1988 (2008).
Cantin, A. M., Hubbard, R. C. & Crystal, R. G. Glutathione deficiency in the epithelial lining fluid of the lower respiratory tract in idiopathic pulmonary fibrosis. Am. Rev. Respir. Dis. 139, 370–372 (1989).
Beeh, K. M. et al. Glutathione deficiency of the lower respiratory tract in subjects with idiopathic pulmonary fibrosis. Eur. Respir. J. 19, 1119–1123 (2002).
Rahman, I. et al. Systemic and pulmonary oxidative stress in idiopathic pulmonary fibrosis. Free Radic. Biol. Med. 27, 60–68 (1999).
Meyer, A., Buhl, R. & Magnussen, H. The effect of oral N-acetylcysteine on lung glutathione levels in idiopathic pulmonary fibrosis. Eur. Respir. J. 7, 431–436 (1994).
Shahzeidi, S., Sarnstrand, B., Jeffery, P. K., McAnulty, R. J. & Laurent, G. J. Oral N-acetylcysteine reduces bleomycin-induced collagen deposition in the lungs of mice. Eur. Respir. J. 4, 845–852 (1991).
Hagiwara, S. I., Ishii, Y. & Kitamura, S. Aerosolized administration of N-acetylcysteine attenuates lung fibrosis induced by bleomycin in mice. Am. J. Respir. Crit. Care Med. 162, 225–231 (2000).
Demedts, M. et al. High-dose acetylcysteine in idiopathic pulmonary fibrosis. N. Engl. J. Med. 353, 2229–2242 (2005).
Martinez, F. J., de Andrade, J. A., Anstrom, K. J., King, T. E. Jr. & Raghu, G. Randomized trial of acetylcysteine in idiopathic pulmonary fibrosis. N. Engl. J. Med. 370, 2093–2101 (2014).
Janssen-Heininger, Y. M. et al. Redox-based regulation of signal transduction: principles, pitfalls, and promises. Free Radic. Biol. Med. 45, 1–17 (2008).
Aesif, S. W. et al. In situ analysis of protein S-glutathionylation in lung tissue using glutaredoxin-1-catalyzed cysteine derivatization. Am. J. Pathol. 175, 36–45 (2009).
Kuwano, K. et al. Essential roles of the Fas–Fas ligand pathway in the development of pulmonary fibrosis. J. Clin. Invest. 104, 13–19 (1999).
Anathy, V. et al. Redox amplification of apoptosis by caspase-dependent cleavage of glutaredoxin 1 and S-glutathionylation of Fas. J. Cell Biol. 184, 241–252 (2009).
Hagimoto, N. et al. Induction of apoptosis and pulmonary fibrosis in mice in response to ligation of Fas antigen. Am. J. Respir. Cell Mol. Biol. 17, 272–278 (1997).
Sisson, T. H. et al. Targeted injury of type II alveolar epithelial cells induces pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 181, 254–263 (2010).
Liang, J. et al. Hyaluronan and TLR4 promote surfactant-protein-C-positive alveolar progenitor cell renewal and prevent severe pulmonary fibrosis in mice. Nat. Med. 22, 1285–1293 (2016).
Korfei, M. et al. Epithelial endoplasmic reticulum stress and apoptosis in sporadic idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 178, 838–846 (2008).
Degryse, A. L. et al. TGFβ signaling in lung epithelium regulates bleomycin-induced alveolar injury and fibroblast recruitment. Am. J. Physiol. Lung Cell Mol. Physiol. 300, L887–897 (2011).
Anathy, V. et al. Glutaredoxin-1 attenuates S-glutathionylation of the death receptor Fas and decreases resolution of Pseudomonas aeruginosa pneumonia. Am. J. Respir. Crit. Care Med. 189, 463–474 (2014).
Sime, P. J., Xing, Z., Graham, F. L., Csaky, K. G. & Gauldie, J. Adenovector-mediated gene transfer of active transforming growth factor-β1 induces prolonged severe fibrosis in rat lung. J. Clin. Invest. 100, 768–776 (1997).
Hashemy, S. I., Johansson, C., Berndt, C., Lillig, C. H. & Holmgren, A. Oxidation and S-nitrosylation of cysteines in human cytosolic and mitochondrial glutaredoxins: effects on structure and activity. J. Biol. Chem. 282, 14428–14436 (2007).
Selman, M. & Pardo, A. Revealing the pathogenic and aging-related mechanisms of the enigmatic idiopathic pulmonary fibrosis. An integral model. Am. J. Respir. Crit. Care Med. 189, 1161–1172 (2014).
Mora, A. L., Rojas, M., Pardo, A. & Selman, M. Emerging therapies for idiopathic pulmonary fibrosis, a progressive age-related disease. Nat. Rev. Drug Discov. 16, 810 (2017).
Hecker, L. et al. Reversal of persistent fibrosis in aging by targeting Nox4–Nrf2 redox imbalance. Sci. Transl. Med. 6, 231ra247 (2014).
Kropski, J. A. & Blackwell, T. S. Endoplasmic reticulum stress in the pathogenesis of fibrotic disease. J. Clin. Invest. 128, 64–73 (2018).
Thannickal, V. J. & Horowitz, J. C. Evolving concepts of apoptosis in idiopathic pulmonary fibrosis. Proc. Am. Thorac. Soc. 3, 350–356 (2006).
King, T. E. Jr et al. A phase 3 trial of pirfenidone in subjects with idiopathic pulmonary fibrosis. N. Engl. J. Med. 370, 2083–2092 (2014).
Richeldi, L. et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N. Engl. J. Med. 370, 2071–2082 (2014).
Lillig, C. H., Berndt, C. & Holmgren, A. Glutaredoxin systems. Biochim. Biophys. Acta 1780, 1304–1317 (2008).
Wu, R. F., Ma, Z., Liu, Z. & Terada, L. S. Nox4-derived H2O2 mediates endoplasmic reticulum signaling through local Ras activation. Mol. Cell. Biol. 30, 3553–3568 (2010).
Carnesecchi, S. et al. A key role for NOX4 in epithelial cell death during development of lung fibrosis. Antioxid. Redox Signal. 15, 607–619 (2011).
Anathy, V. et al. Oxidative processing of latent Fas in the endoplasmic reticulum controls the strength of apoptosis. Mol. Cell. Biol. 32, 3464–3478 (2012).
Bueno, M. et al. PINK1 deficiency impairs mitochondrial homeostasis and promotes lung fibrosis. J. Clin. Invest. 125, 521–538 (2015).
Mora, A. L., Bueno, M. & Rojas, M. Mitochondria in the spotlight of aging and idiopathic pulmonary fibrosis. J. Clin. Invest. 127, 405–414 (2017).
Perl, A. K., Zhang, L. & Whitsett, J. A. Conditional expression of genes in the respiratory epithelium in transgenic mice: cautionary notes and toward building a better mouse trap. Am. J. Respir. Cell Mol. Biol. 40, 1–3 (2009).
Perl, A. K. et al. Conditional recombination reveals distinct subsets of epithelial cells in trachea, bronchi, and alveoli. Am. J. Respir. Cell Mol. Biol. 33, 455–462 (2005).
Salmena, L. et al. Essential role for caspase 8 in T-cell homeostasis and T-cell-mediated immunity. Genes Dev. 17, 883–895 (2003).
Watanabe-Fukunaga, R., Brannan, C. I., Copeland, N. G., Jenkins, N. A. & Nagata, S. Lymphoproliferation disorder in mice explained by defects in Fas antigen that mediates apoptosis. Nature 356, 314–317 (1992).
Ho, Y. S. et al. Targeted disruption of the glutaredoxin 1 gene does not sensitize adult mice to tissue injury induced by ischemia/reperfusion and hyperoxia. Free Radic. Biol. Med. 43, 1299–1312 (2007).
Bauer, Y. et al. A novel genomic signature with translational significance for human idiopathic pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 52, 217–231 (2015).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, 2011).
Woessner, J. F. Jr. The determination of hydroxyproline in tissue and protein samples containing small proportions of this imino acid. Arch. Biochem. Biophys. 93, 440–447 (1961).
Fonseca, C., Taatjes, D. J., Callas, P., Ittleman, F. & Bovill, E. G. The effects of aging on the intimal region of the human saphenous vein: insights from multimodal microscopy and quantitative image analysis. Histochem. Cell Biol. 138, 435–445 (2012).
Reynaert, N. L., Wouters, E. F. & Janssen-Heininger, Y. M. Modulation of glutaredoxin-1 expression in a mouse model of allergic airway disease. Am. J. Respir. Cell Mol. Biol. 36, 147–151 (2007).
Coppo, L., Montano, S. J., Padilla, A. C. & Holmgren, A. Determination of glutaredoxin enzyme activity and protein S-glutathionylation using fluorescent eosin-glutathione. Anal. Biochem. 499, 24–33 (2016).
Micsonai, A. et al. Accurate secondary structure prediction and fold recognition for circular dichroism spectroscopy. Proc. Natl Acad. Sci. USA 112, E3095–3103 (2015).
Rahman, I., Kode, A. & Biswas, S. K. Assay for quantitative determination of glutathione and glutathione disulfide levels using enzymatic recycling method. Nat. Protoc. 1, 3159–3165 (2006).
Jones, P. W., Quirk, F. H., Baveystock, C. M. & Littlejohns, P. A self-complete measure of health status for chronic airflow limitation. The St. George’s Respiratory Questionnaire. Am. Rev. Respir. Dis. 145, 1321–1327 (1992).
Acknowledgements
This work was funded by NIH R35HL135828, R01HL079331, R01HL060014 and R03HL095404 (Y.M.W.J.-H.), an ATS unrestricted grant and NIH R01HL122383 (V.A.), Parker B. Francis fellowships (V.A. and J.L.J.v.d.V.), NIH R01HL05646 (A.v.d.V.), T32HL076122 (D.H.M. and J.T.J.), NCRR 1S10RR019246 (D.J.T.), American Heart Association 16GRNT27660006, NIH R01DK103750 and 1UL1TR001430 (M.M.B.), NIH R01HL133013 and R03 AG051857 (R.M.), P30GM103532 (C.G.I.), FAMRI Young Clinical Scientist Award 113393 and 1K01HL125474-01 (J.L.G.) and U01HL108642 and RC2 HL101715 (N.K.). Analysis of biological specimens and data provided by the Lung Tissue Research Consortium (LTRC) was supported by R03HL095404 (Y.M.W.J.-H.). We would like to thank J. Whitsett (University of Cincinnati) for generously providing the CCSP-rtTA, TetOP-Cre mice, J. Gauldie for providing AdTGFB1, N. Bishop for help with imaging of GLRX and collagen, J. M. Siddesha for technical support and P. Vacek for statistical support.
Author information
Authors and Affiliations
Contributions
V.A., K.G.L., S.M.H., D.H.M., J.T.J., E.E., J.D.N., D.T.C., J.L.J.v.d.V. and R.S. performed the animal experiments. S.A., S.B.C. and K.G.L. prepared and tested recombinant GLRX; V.A., S.M.H., E.C.R., R.A. and K.G.L. performed S-glutathionylation and glutaredoxin assays; K.G.L. and D.T.C. performed fibrosis analyses; K.J.B. reviewed histopathology; D.J.T. provided critical suggestions regarding imaging and image analysis. Y.-S.H. generated Glrx−/− mice and helped with data interpretation; R.A. and S.B.C. performed collagenolytic activity assays; M.D.L. and M.E.C. performed and analyzed CD studies; R.H. generated Casp8loxP/loxP mice and helped with data interpretation. R.M. and M.M.B. conducted the ageing study and participated in data analyses. A.v.d.V. and R.C.B. supported the studies with key experimental suggestions and helped interpret data. D.J.S., D.G.C., C.G.I., K.J.B., J.L.G., N.K. and K.K.B. evaluated and analyzed clinical data. Y.M.W.J.-H. and V.A. designed the research, interpreted the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Y.M.W.J.-H. and V.A. hold patents entitled: ‘Treatments involving glutaredoxins and similar agents’, ‘Treatments of oxidative stress conditions’ (US patent no. 8,679,811 and 9,907,828; Y.M.W.J.-H. and V.A.) and ‘Detection of glutathionylated proteins’ (US patent no. 8,877,447; Y.M.W.J.-H.).
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13 and Supplementary Tables 1 and 2
Supplementary Dataset
F, t and df values
Rights and permissions
About this article
Cite this article
Anathy, V., Lahue, K.G., Chapman, D.G. et al. Reducing protein oxidation reverses lung fibrosis. Nat Med 24, 1128–1135 (2018). https://doi.org/10.1038/s41591-018-0090-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0090-y
This article is cited by
-
Local fractal dimension of collagen detects increased spatial complexity in fibrosis
Histochemistry and Cell Biology (2024)
-
Structural and functional fine mapping of cysteines in mammalian glutaredoxin reveal their differential oxidation susceptibility
Nature Communications (2023)
-
ADRB2 inhibition combined with antioxidant treatment alleviates lung fibrosis by attenuating TGFβ/SMAD signaling in lung fibroblasts
Cell Death Discovery (2023)
-
Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology
Nature Reviews Molecular Cell Biology (2022)
-
Oxidative stress-induced FABP5 S-glutathionylation protects against acute lung injury by suppressing inflammation in macrophages
Nature Communications (2021)