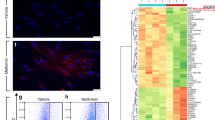
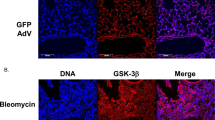
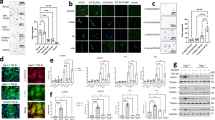
Abstract
Fibrosis is a pathological result of a dysfunctional repair response to tissue injury and occurs in a number of organs, including the lungs1. Cellular metabolism regulates tissue repair and remodelling responses to injury2,3,4. AMPK is a critical sensor of cellular bioenergetics and controls the switch from anabolic to catabolic metabolism5. However, the role of AMPK in fibrosis is not well understood. Here, we demonstrate that in humans with idiopathic pulmonary fibrosis (IPF) and in an experimental mouse model of lung fibrosis, AMPK activity is lower in fibrotic regions associated with metabolically active and apoptosis-resistant myofibroblasts. Pharmacological activation of AMPK in myofibroblasts from lungs of humans with IPF display lower fibrotic activity, along with enhanced mitochondrial biogenesis and normalization of sensitivity to apoptosis. In a bleomycin model of lung fibrosis in mice, metformin therapeutically accelerates the resolution of well-established fibrosis in an AMPK-dependent manner. These studies implicate deficient AMPK activation in non-resolving, pathologic fibrotic processes, and support a role for metformin (or other AMPK activators) to reverse established fibrosis by facilitating deactivation and apoptosis of myofibroblasts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
13 August 2018
In the version of this article originally published, a grant was omitted from the Acknowledgements section. The following sentence should have been included: “R.B.M. was supported by a Department of Veterans Affairs Merit Award (5I01BX003272).” The error has been corrected in the HTML and PDF versions of this article.
References
Wynn, T. A. & Ramalingam, T. R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat. Med. 18, 1028–1040 (2012).
Lumeng, C. N. & Saltiel, A. R. Inflammatory links between obesity and metabolic disease. J. Clin. Invest. 121, 2111–2117 (2011).
O’Neill, L. A. & Hardie, D. G. Metabolism of inflammation limited by AMPK and pseudo-starvation. Nature 493, 346–355 (2013).
Eltzschig, H. K. & Eckle, T. Ischemia and reperfusion—from mechanism to translation. Nat. Med. 17, 1391–1401 (2011).
Hardie, D. G., Ross, F. A. & Hawley, S. A. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 13, 251–262 (2012).
Beers, M. F. & Morrisey, E. E. The three R’s of lung health and disease: repair, remodeling, and regeneration. J. Clin. Invest. 121, 2065–2073 (2011).
Thannickal, V. J., Zhou, Y., Gaggar, A. & Duncan, S. R. Fibrosis: ultimate and proximate causes. J. Clin. Invest. 124, 4673–4677 (2014).
Duffield, J. S., Lupher, M., Thannickal, V. J. & Wynn, T. A. Host responses in tissue repair and fibrosis. Annu. Rev. Pathol. 8, 241–276 (2013).
Bueno, M. et al. PINK1 deficiency impairs mitochondrial homeostasis and promotes lung fibrosis. J. Clin. Invest. 125, 521–538 (2015).
Kobayashi, K. et al. Involvement of PARK2-mediated mitophagy in idiopathic pulmonary fibrosis pathogenesis. J. Immunol. 197, 504–516 (2016).
Bernard, K. et al. Metabolic reprogramming is required for myofibroblast contractility and differentiation. J. Biol. Chem. 290, 25427–25438 (2015).
Ramos, C. et al. Fibroblasts from idiopathic pulmonary fibrosis and normal lungs differ in growth rate, apoptosis, and tissue inhibitor of metalloproteinases expression. Am. J. Respir. Cell Mol. Biol. 24, 591–598 (2001).
Romero, Y. et al. mTORC1 activation decreases autophagy in aging and idiopathic pulmonary fibrosis and contributes to apoptosis resistance in IPF fibroblasts. Aging Cell 15, 1103–1112 (2016).
Ashley, S. L. et al. Targeting inhibitor of apoptosis proteins protects from bleomycin-induced lung fibrosis. Am. J. Respir. Cell Mol. Biol. 54, 482–492 (2016).
Hecker, L. et al. Reversal of persistent fibrosis in aging by targeting Nox4-Nrf2 redox imbalance. Sci. Transl. Med. 6, 231ra247 (2014).
Inoki, K., Kim, J. & Guan, K. L. AMPK and mTOR in cellular energy homeostasis and drug targets. Annu. Rev. Pharmacol. Toxicol. 52, 381–400 (2012).
Shaw, R. J. Metformin trims fats to restore insulin sensitivity. Nat. Med. 19, 1570–1572 (2013).
Riera, C. E. & Dillin, A. Can aging be ‘drugged’? Nat. Med. 21, 1400–1405 (2015).
Finkel, T. The metabolic regulation of aging. Nat. Med. 21, 1416–1423 (2015).
Burkewitz, K., Zhang, Y. & Mair, W. B. AMPK at the nexus of energetics and aging. Cell Metab. 20, 10–25 (2014).
Salminen, A. & Kaarniranta, K. AMP-activated protein kinase (AMPK) controls the aging process via an integrated signaling network. Ageing Res. Rev. 11, 230–241 (2012).
Park, C. S. et al. Metformin reduces airway inflammation and remodeling via activation of AMP-activated protein kinase. Biochem. Pharmacol. 84, 1660–1670 (2012).
Liu, Z. et al. AMP-activated protein kinase and glycogen synthase kinase 3β modulate the severity of sepsis-induced lung injury. Mol. Med. 21, 937–950 (2015).
Sato, N. et al. Metformin attenuates lung fibrosis development via NOX4 suppression. Respir. Res. 17, 107 (2016).
Mishra, R. et al. AMP-activated protein kinase inhibits transforming growth factor-beta-induced Smad3-dependent transcription and myofibroblast transdifferentiation. J. Biol. Chem. 283, 10461–10469 (2008).
Thakur, S. et al. Activation of AMP-activated protein kinase prevents TGF-β1-induced epithelial-mesenchymal transition and myofibroblast activation. Am. J. Pathol. 185, 2168–2180 (2015).
Lim, J. Y., Oh, M. A., Kim, W. H., Sohn, H. Y. & Park, S. I. AMP-activated protein kinase inhibits TGF-β-induced fibrogenic responses of hepatic stellate cells by targeting transcriptional coactivator p300. J. Cell Physiol. 227, 1081–1089 (2012).
Li, L. et al. Metformin attenuates gefitinib-induced exacerbation of pulmonary fibrosis by inhibition of TGF-β signaling pathway. Oncotarget 6, 43605–43619 (2015).
Park, I. H. et al. Metformin reduces TGF-β1-induced extracellular matrix production in nasal polyp-derived fibroblasts. Otolaryngol. Head Neck Surg. 150, 148–153 (2014).
Kottmann, R. M. et al. Lactic acid is elevated in idiopathic pulmonary fibrosis and induces myofibroblast differentiation via pH-dependent activation of transforming growth factor-beta. Am. J. Respir. Crit. Care Med. 186, 740–751 (2012).
Egan, D. F. et al. Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science 331, 456–461 (2011).
Parsons, M. J. & Green, D. R. Mitochondria in cell death. Essays Biochem. 47, 99–114 (2010).
Tait, S. W. & Green, D. R. Mitochondria and cell death: outer membrane permeabilization and beyond. Nat. Rev. Mol. Cell Biol. 11, 621–632 (2010).
Kim, J. W., Tchernyshyov, I., Semenza, G. L. & Dang, C. V. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 3, 177–185 (2006).
Spoden, G. A. et al. Pyruvate kinase isoenzyme M2 is a glycolytic sensor differentially regulating cell proliferation, cell size and apoptotic cell death dependent on glucose supply. Exp. Cell Res. 315, 2765–2774 (2009).
Horowitz, J. C. et al. Activation of the pro-survival phosphatidylinositol 3-kinase/AKT pathway by transforming growth factor-β1 in mesenchymal cells is mediated by p38 MAPK-dependent induction of an autocrine growth factor. J. Biol. Chem. 279, 1359–1367 (2004).
Foretz, M., Guigas, B., Bertrand, L., Pollak, M. & Viollet, B. Metformin: from mechanisms of action to therapies. Cell Metab. 20, 953–966 (2014).
Knowler, W. C. et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 346, 393–403 (2002).
Yu, G. et al. Thyroid hormone inhibits lung fibrosis in mice by improving epithelial mitochondrial function. Nat. Med. 24, 39–49 (2018).
Jiang, S. et al. Mitochondria and AMP-activated protein kinase-dependent mechanism of efferocytosis. J. Biol. Chem. 288, 26013–26026 (2013).
Jian, M. Y., Alexeyev, M. F., Wolkowicz, P. E., Zmijewski, J. W. & Creighton, J. R. Metformin-stimulated AMPK-α1 promotes microvascular repair in acute lung injury. Am. J. Physiol. Lung Cell Mol. Physiol. 305, L844–855 (2013).
Hecker, L. et al. NADPH oxidase-4 mediates myofibroblast activation and fibrogenic responses to lung injury. Nat. Med. 15, 1077–1081 (2009).
Jiang, S. et al. Human resistin promotes neutrophil proinflammatory activation and neutrophil extracellular trap formation and increases severity of acute lung injury. J. Immunol. 192, 4795–4803 (2014).
Franco-Barraza, J., Beacham, D. A., Amatangelo, M. D., & Cukierman, E. Preparation of extracellular matrices produced by cultured and primary fibroblasts. Curr. Protoc. Cell Biol. 71, 10.9.1–10.9.34 (2016).
Hill, B. G. et al. Integration of cellular bioenergetics with mitochondrial quality control and autophagy. Biol. Chem. 393, 1485–1512 (2012).
Graham, L. & Orenstein, J.M. Processing tissue and cells for transmission electron microscopy in diagnostic pathology and research. Nat. Protoc. 2, 2439–2450 (2007).
Acknowledgements
The authors thank B. Viollet (INSERM) and G. Shailendra (Henry Ford Health System) for providing AMPK α1/2−/− MEFs and AMPKα1−/− mice. The authors thank Y. Liu (Medicine, UAB) for technical support, and J. Creighton (Anesthesiology, UAB) and the Neuroscience Molecular Detection and Stereology Core P30 NS047466 (UAB) for help with lung tissue samples/processing. This work was supported in part by the National Institutes of Health (NIH, HL107585), the US Department of Defense (W81XWH-17-1-0577) and the Pulmonary, Allergy and Critical Care Medicine (UAB) Translational Program for ARDS grants to J.W.Z. V.J.T. was supported by NIH grants P01 HL114470 and R01 AG046210, and a Department of Veterans Affairs Merit Award I01BX003056. Su.R. was supported by NIH K08 (HL135399). V.D.-U. received support from UAB Nathan Shock Center (P30 AG 050886). R.B.M. was supported by a Department of Veterans Affairs Merit Award (5I01BX003272).
Author information
Authors and Affiliations
Contributions
Conception and design was provided by V.J.T. and J.W.Z. Experiments, data analysis and interpretation were carried out by Su.R., S.J., D.W.P., N.B.B., K.B., J.D., A.A.Z., R.B.M., M.L.L., Sa.R., E.A., V.D.-U., V.J.T. and J.W.Z. Drafting and revision of the manuscript was carried out by Su.R., V.J.T. and J.W.Z.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1 and 2
Rights and permissions
About this article
Cite this article
Rangarajan, S., Bone, N.B., Zmijewska, A.A. et al. Metformin reverses established lung fibrosis in a bleomycin model. Nat Med 24, 1121–1127 (2018). https://doi.org/10.1038/s41591-018-0087-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0087-6
This article is cited by
-
Activation of senescence in critically ill patients: mechanisms, consequences and therapeutic opportunities
Annals of Intensive Care (2024)
-
Amifostine ameliorates bleomycin-induced murine pulmonary fibrosis via NAD+/SIRT1/AMPK pathway-mediated effects on mitochondrial function and cellular metabolism
European Journal of Medical Research (2024)
-
Diabetes mellitus and idiopathic pulmonary fibrosis: a Mendelian randomization study
BMC Pulmonary Medicine (2024)
-
Association between statin use and the risk for idiopathic pulmonary fibrosis and its prognosis: a nationwide, population-based study
Scientific Reports (2024)
-
Emerging therapeutic targets in systemic sclerosis
Journal of Molecular Medicine (2024)