Abstract
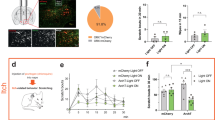
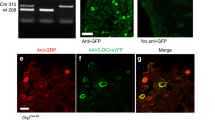
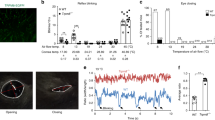
Itch and pain are refractory symptoms of many ocular conditions. Ocular itch is generated mainly in the conjunctiva and is absent from the cornea. In contrast, most ocular pain arises from the cornea. However, the underlying mechanisms remain unknown. Using genetic axonal tracing approaches, we discover distinct sensory innervation patterns between the conjunctiva and cornea. Further genetic and functional analyses in rodent models show that a subset of conjunctival-selective sensory fibers marked by MrgprA3 expression, rather than corneal sensory fibers, mediates ocular itch. Importantly, the actions of both histamine and nonhistamine pruritogens converge onto this unique subset of conjunctiva sensory fibers and enable them to play a key role in mediating itch associated with allergic conjunctivitis. This is distinct from skin itch, in which discrete populations of sensory neurons cooperate to carry itch. Finally, we provide proof of concept that selective silencing of conjunctiva itch-sensing fibers by pruritogen-mediated entry of sodium channel blocker QX-314 is a feasible therapeutic strategy to treat ocular itch in mice. Itch-sensing fibers also innervate the human conjunctiva and allow pharmacological silencing using QX-314. Our results cast new light on the neural mechanisms of ocular itch and open a new avenue for developing therapeutic strategies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ciprandi, G., Buscaglia, S., Cerqueti, P. M. & Canonica, G. W. Drug treatment of allergic conjunctivitis. A review of the evidence. Drugs 43, 154–176 (1992).
Abelson, M. B., Smith, L. & Chapin, M. Ocular allergic disease: mechanisms, disease sub-types, treatment. Ocul. Surf. 1, 127–149 (2003).
Wong, A. H., Barg, S. S. & Leung, A. K. Seasonal and perennial allergic conjunctivitis. Recent Pat. Inflamm. Allergy Drug Discov. 3, 118–127 (2009).
Ono, S. J. & Abelson, M. B. Allergic conjunctivitis: update on pathophysiology and prospects for future treatment. J. Allergy Clin. Immunol. 115, 118–122 (2005).
Yeniad, B., Alparslan, N. & Akarcay, K. Eye rubbing as an apparent cause of recurrent keratoconus. Cornea 28, 477–479 (2009).
Nagaki, Y., Hayasaka, S. & Kadoi, C. Cataract progression in patients with atopic dermatitis. J. Cataract Refract. Surg. 25, 96–99 (1999).
Ambati, B. K. et al. Corneal avascularity is due to soluble VEGF receptor-1. Nature 443, 993–997 (2006).
Cursiefen, C. Immune privilege and angiogenic privilege of the cornea. Chem. Immunol. Allergy 92, 50–57 (2007).
Paus, R., Schmelz, M., Bíró, T. & Steinhoff, M. Frontiers in pruritus research: scratching the brain for more effective itch therapy. J. Clin. Invest. 116, 1174–1186 (2006).
Oetjen, L. K. et al. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell 171, 217–228.e13 (2017).
Bonini, S. et al. Conjunctival provocation test as a model for the study of allergy and inflammation in humans. Int. Arch. Allergy Appl. Immunol. 88, 144–148 (1989).
Leonardi, A. The central role of conjunctival mast cells in the pathogenesis of ocular allergy. Curr. Allergy Asthma Rep. 2, 325–331 (2002).
Belmonte, C., Acosta, M. C., Schmelz, M. & Gallar, J. Measurement of corneal sensitivity to mechanical and chemical stimulation with a CO2 esthesiometer. Invest. Ophthalmol. Vis. Sci. 40, 513–519 (1999).
Müller, L. J., Marfurt, C. F., Kruse, F. & Tervo, T. M. Corneal nerves: structure, contents and function. Exp. Eye Res. 76, 521–542 (2003).
Tanelian, D. L. & Beuerman, R. W. Responses of rabbit corneal nociceptors to mechanical and thermal stimulation. Exp. Neurol. 84, 165–178 (1984).
MacIver, M. B. & Tanelian, D. L. Structural and functional specialization of A delta and C fiber free nerve endings innervating rabbit corneal epithelium. J. Neurosci. 13, 4511–4524 (1993).
Beuerman, R. W. & Tanelian, D. L. Corneal pain evoked by thermal stimulation. Pain 7, 1–14 (1979).
Huang, C. C. et al. A histamine-independent itch pathway is required for allergic ocular itch. J. Allergy Clin. Immunol. 137, 1267–1270.e6 (2016).
Bautista, D. M. et al. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 124, 1269–1282 (2006).
Wilson, S. R. et al. TRPA1 is required for histamine-independent, Mas-related G protein–coupled receptor–mediated itch. Nat. Neurosci. 14, 595–602 (2011).
Usoskin, D. et al. Unbiased classification of sensory neuron types by large-scale single-cell RNA sequencing. Nat. Neurosci. 18, 145–153 (2015).
Cavanaugh, D. J. et al. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc. Natl Acad. Sci. USA 106, 9075–9080 (2009).
Kremer, A. E. et al. Lysophosphatidic acid is a potential mediator of cholestatic pruritus. Gastroenterology 139, 1008–1018.e1 (2010).
Liu, Q. et al. Mechanisms of itch evoked by β-alanine. J. Neurosci. 32, 14532–14537 (2012).
Liu, Q. et al. Sensory neuron-specific GPCR Mrgprs are itch receptors mediating chloroquine-induced pruritus. Cell 139, 1353–1365 (2009).
Han, L. et al. A subpopulation of nociceptors specifically linked to itch. Nat. Neurosci. 16, 174–182 (2013).
Liu, Q. et al. The distinct roles of two GPCRs, MrgprC11 and PAR2, in itch and hyperalgesia. Sci. Signal. 4, ra45 (2011).
Sikand, P., Dong, X. & LaMotte, R. H. BAM8-22 peptide produces itch and nociceptive sensations in humans independent of histamine release. J. Neurosci. 31, 7563–7567 (2011).
Reddy, V. B. et al. Redefining the concept of protease-activated receptors: cathepsin S evokes itch via activation of Mrgprs. Nat. Commun. 6, 7864 (2015).
Stantcheva, K. K. et al. A subpopulation of itch-sensing neurons marked by Ret and somatostatin expression. EMBO Rep. 17, 585–600 (2016).
Seal, R. P. et al. Injury-induced mechanical hypersensitivity requires C-low threshold mechanoreceptors. Nature 462, 651–655 (2009).
Lou, S., Duan, B., Vong, L., Lowell, B. B. & Ma, Q. Runx1 controls terminal morphology and mechanosensitivity of VGLUT3-expressing C-mechanoreceptors. J. Neurosci. 33, 870–882 (2013).
Zylka, M. J., Rice, F. L. & Anderson, D. J. Topographically distinct epidermal nociceptive circuits revealed by axonal tracers targeted to Mrgprd. Neuron 45, 17–25 (2005).
Knop, E., Korb, D. R., Blackie, C. A. & Knop, N. The lid margin is an underestimated structure for preservation of ocular surface health and development of dry eye disease. Dev. Ophthalmol. 45, 108–122 (2010).
Cevikbas, F. et al. A sensory neuron-expressed IL-31 receptor mediates T helper cell–dependent itch: involvement of TRPV1 and TRPA1. J. Allergy Clin. Immunol. 133, 448–460 (2014).
Lee, M. G. et al. Agonists of the MAS-related gene (Mrgs) orphan receptors as novel mediators of mast cell-sensory nerve interactions. J. Immunol. 180, 2251–2255 (2008).
Morita, T. et al. HTR7 mediates serotonergic acute and chronic itch. Neuron 87, 124–138 (2015).
Kim, Y. S. et al. Central terminal sensitization of TRPV1 by descending serotonergic facilitation modulates chronic pain. Neuron 81, 873–887 (2014).
Otsuki, J. A., Grassick, R., Seymour, D. & Kind, L. S. The use of 3H serotonin release from mast cells of the mouse as an assay for mediator liberation. Immunol. Commun. 5, 27–39 (1976).
Razin, E. et al. IgE-mediated release of leukotriene C4, chondroitin sulfate E proteoglycan, β-hexosaminidase, and histamine from cultured bone marrow-derived mouse mast cells. J. Exp. Med. 157, 189–201 (1983).
Panneton, W. M., Hsu, H. & Gan, Q. Distinct central representations for sensory fibers innervating either the conjunctiva or cornea of the rat. Exp. Eye Res. 90, 388–396 (2010).
Zhao, Z. Q. et al. Cross-inhibition of NMBR and GRPR signaling maintains normal histaminergic itch transmission. J. Neurosci. 34, 12402–12414 (2014).
Fleming, M. S. et al. The majority of dorsal spinal cord gastrin releasing peptide is synthesized locally whereas neuromedin B is highly expressed in pain- and itch-sensing somatosensory neurons. Mol. Pain 8, 52 (2012).
Mishra, S. K., Holzman, S. & Hoon, M. A. A nociceptive signaling role for neuromedin B. J. Neurosci. 32, 8686–8695 (2012).
Binshtok, A. M., Bean, B. P. & Woolf, C. J. Inhibition of nociceptors by TRPV1-mediated entry of impermeant sodium channel blockers. Nature 449, 607–610 (2007).
Roberson, D. P. et al. Activity-dependent silencing reveals functionally distinct itch-generating sensory neurons. Nat. Neurosci. 16, 910–918 (2013).
McNeil, B. D. et al. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 519, 237–241 (2015).
Mousli, M. et al. Activation of rat peritoneal mast cells by substance P and mastoparan. J. Pharmacol. Exp. Ther. 250, 329–335 (1989).
Parra, A. et al. Ocular surface wetness is regulated by TRPM8-dependent cold thermoreceptors of the cornea. Nat. Med. 16, 1396–1399 (2010).
Belmonte, C., Aracil, A., Acosta, M. C., Luna, C. & Gallar, J. Nerves and sensations from the eye surface. Ocul. Surf. 2, 248–253 (2004).
Abila, B., Ezeamuzie, I. C., Igbigbi, P. S., Ambakederemo, A. W. & Asomugha, L. Effects of two antihistamines on chloroquine and histamine induced weal and flare in healthy African volunteers. Afr. J. Med. Med. Sci. 23, 139–142 (1994).
McCoy, E. S. et al. Peptidergic CGRPα primary sensory neurons encode heat and itch and tonically suppress sensitivity to cold. Neuron 78, 138–151 (2013).
Liu, Q. et al. Molecular genetic visualization of a rare subset of unmyelinated sensory neurons that may detect gentle touch. Nat. Neurosci. 10, 946–948 (2007).
Dell, R. B., Holleran, S. & Ramakrishnan, R. Sample size determination. ILAR J. 43, 207–213 (2002).
Acknowledgements
We are grateful to M. W. Panneton, H. Hu, B. Kim, Z. F. Chen, T. P. Margolis and X. Dong for insightful discussions and comments on the manuscript, and we thank A. S. Yoo and Y. Liu for technical support. Mrgpra3GFP-cre, PirtGCaMP3/+ and MRGPRX1;Mrgpr-clusterΔ−/− mice were generous gifts from X. Dong of Johns Hopkins University. MrgprdeGFP/+ mice were from D. J. Anderson of the California Institute of Technology. Trpm8GFP/+ mice were from G. Story. Nav1.8cre, Nmb−/−, Nmbr−/− and NmbrGFP transgenic mice were from Z. -F. Chen of Washington University in St. Louis. Slc17a8Cre/+ tissues were from Q. Ma of Dana-Farber Cancer Institute. This work was supported by the “Research to Prevent Blindness” (RPB) unrestricted grant to the Department of Ophthalmology (A.J.W.H. and Q.L.), and the National Institutes of Health (R01EY024704 and 1R01AI125743; Q.L.) and the Pew Scholar Award (Q.L.).
Author information
Authors and Affiliations
Contributions
C.-C.H. performed genetic axonal tracing, pharmacological and behavioral assays, calcium imaging experiments, immunofluorescence staining and data analysis and participated in manuscript preparation. W.Y. conducted retrograde tracing of ocular afferent neurons and single-cell picking, pharmacological and behavioral assays, calcium imaging, immunofluorescence and H&E staining and data analysis and participated in manuscript preparation. C.G. conducted single-cell qRT-PCR and immunofluorescence staining and assisted with mouse breeding strategy design, genetic ablation tests and manuscript preparation. H.J. performed electrophysiological recordings and data analysis. F.L. conducted TRPM8-GFP axonal tracing and ocular pain tests. M.X., in collaboration with W.Y, examined itch-sensing afferent fibers in human conjunctiva. S.D. performed the electrophysiological recordings and data analysis. G.Y. conducted calcium imaging of culture DRG neurons. B.D. and T.H. perfused Slc17a8cre/+; Rosa26tdTomato/+ mice and provided tissues for imaging. A.J.W.H. provided human tissues and contributed to experimental design and manuscript preparation. Q.L. planned and directed all of the experiments and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 and Supplementary Tables 1 and 2
Rights and permissions
About this article
Cite this article
Huang, CC., Yang, W., Guo, C. et al. Anatomical and functional dichotomy of ocular itch and pain. Nat Med 24, 1268–1276 (2018). https://doi.org/10.1038/s41591-018-0083-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0083-x
This article is cited by
-
Neuronal FcεRIα directly mediates ocular itch via IgE-immune complex in a mouse model of allergic conjunctivitis
Journal of Neuroinflammation (2022)
-
Mucosal immunology of the ocular surface
Mucosal Immunology (2022)
-
Exploration of sensory and spinal neurons expressing gastrin-releasing peptide in itch and pain related behaviors
Nature Communications (2020)
-
Itch in Organs Beyond the Skin
Current Allergy and Asthma Reports (2020)
-
Factors affecting pain in patients undergoing bilateral cataract surgery
International Ophthalmology (2020)