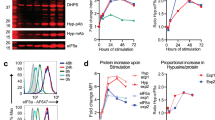
Abstract
In response to pathogenic threats, naive T cells rapidly transition from a quiescent to an activated state, yet the underlying mechanisms are incompletely understood. Using a pulsed SILAC approach, we investigated the dynamics of mRNA translation kinetics and protein turnover in human naive and activated T cells. Our datasets uncovered that transcription factors maintaining T cell quiescence had constitutively high turnover, which facilitated their depletion following activation. Furthermore, naive T cells maintained a surprisingly large number of idling ribosomes as well as 242 repressed mRNA species and a reservoir of glycolytic enzymes. These components were rapidly engaged following stimulation, promoting an immediate translational and glycolytic switch to ramp up the T cell activation program. Our data elucidate new insights into how T cells maintain a prepared state to mount a rapid immune response, and provide a resource of protein turnover, absolute translation kinetics and protein synthesis rates in T cells (https://www.immunomics.ch).
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The MS proteomics raw data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD017159. RNA-seq and ChIP-seq data have been deposited to the NCBI Gene Expression Omnibus (GEO) with the accession numbers GSE147229 and GSE146787. The data that support the findings of this study are available at https://www.immunomics.ch, attached as Supplementary Tables 1–6 or are available from the corresponding author upon request.
References
Crabtree, G. R. Contingent genetic regulatory events in T lymphocyte activation. Science 243, 355–361 (1989).
Vrisekoop, N. et al. Sparse production but preferential incorporation of recently produced naive T cells in the human peripheral pool. Proc. Natl Acad. Sci. USA 105, 6115–6120 (2008).
Chapman, N. M., Boothby, M. R. & Chi, H. Metabolic coordination of T cell quiescence and activation. Nat. Rev. Immunol. 20, 55–70 (2020).
Pearce, E. L., Poffenberger, M. C., Chang, C.-H. & Jones, R. G. Fueling immunity: insights into metabolism and lymphocyte function. Science 342, 1242454 (2013).
Fox, C. J., Hammerman, P. S. & Thompson, C. B. Fuel feeds function: energy metabolism and the T-cell response. Nat. Rev. Immunol. 5, 844–852 (2005).
Araki, K. et al. Translation is actively regulated during the differentiation of CD8+ effector T cells. Nat. Immunol. 18, 1046–1057 (2017).
Sinclair, L. V. et al. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat. Immun. 14, 500–508 (2013).
Gubser, P. M. et al. Rapid effector function of memory CD8 + T cells requires an immediate-early glycolytic switch. Nat. Immunol. 14, 1064–1072 (2013).
Buck, M. D., Sowell, R. T., Kaech, S. M. & Pearce, E. L. Metabolic instruction of immunity. Cell 169, 570–586 (2017).
Geiger, R. et al. L-Arginine modulates T cell metabolism and enhances survival and anti-tumor activity. Cell 167, 829–842.e13 (2016).
Howden, A. J. et al. Quantitative analysis of T cell proteomes and environmental sensors during T cell differentiation. Nat. Immunol. 20, 1542–1554 (2019).
Ron-Harel, N. et al. Mitochondrial biogenesis and proteome remodeling promote one-carbon metabolism for T cell activation. Cell Metab. 24, 104–117 (2016).
Kaech, S. M., Hemby, S., Kersh, E. & Ahmed, R. Molecular and functional profiling of memory CD8 T cell differentiation. Cell 111, 837–851 (2002).
Phan, A. T., Goldrath, A. W. & Glass, C. K. Metabolic and epigenetic coordination of T cell and macrophage immunity. Immunity 46, 714–729 (2017).
Schwanhäusser, B. et al. Global quantification of mammalian gene expression control. Nature 473, 337–342 (2011).
Ong, S.-E. et al. Stable isotope labeling by amino acids in cell culture, SILAC, as a simple and accurate approach to expression proteomics. Mol. Cell. Proteom. 1, 376–386 (2002).
Jovanovic, M. et al. Dynamic profiling of the protein life cycle in response to pathogens. Science 347, 1259038 (2015).
Aebersold, R. & Mann, M. Mass-spectrometric exploration of proteome structure and function. Nature 537, 347–355 (2016).
Wiśniewski, J. R. et al. Extensive quantitative remodeling of the proteome between normal colon tissue and adenocarcinoma. Mol. Syst. Biol. 8, 611 (2012).
Bjørkøy, G. et al. p62/SQSTM1 forms protein aggregates degraded by autophagy and has a protective effect on huntingtin-induced cell death. J. Cell Biol. 171, 603–614 (2005).
Wu, N. et al. AMPK-dependent degradation of TXNIP upon energy stress leads to enhanced glucose uptake via GLUT1. Mol. Cell 49, 1167–1175 (2013).
Weinreich, M. A. et al. KLF2 transcription-factor deficiency in T cells results in unrestrained cytokine production and upregulation of bystander chemokine receptors. Immunity 31, 122–130 (2009).
Muthusamy, N., Barton, K. & Leiden, J. M. Defective activation and survival of T cells lacking the Ets-1 transcription factor. Nature 377, 639–642 (1995).
Bories, J.-C. et al. Increased T-cell apoptosis and terminal B-cell differentiation induced by inactivation of the Ets-1 proto-oncogene. Nature 377, 635–638 (1995).
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y. & Greenleaf, W. J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods 10, 1213–1218 (2013).
Froehlich, J. et al. FAM65B controls the proliferation of transformed and primary T cells. Oncotarget 7, 63215 (2016).
Binnewies, M. et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 24, 541–550 (2018).
Zheng, C. et al. Landscape of infiltrating T cells in liver cancer revealed by single-cell sequencing. Cell 169, e1316 (2017).
Morisaki, T. et al. Real-time quantification of single RNA translation dynamics in living cells. Science 352, 1425–1429 (2016).
Wu, B., Eliscovich, C., Yoon, Y. J. & Singer, R. H. Translation dynamics of single mRNAs in live cells and neurons. Science 352, 1430–1435 (2016).
Yan, X., Hoek, T. A., Vale, R. D. & Tanenbaum, M. E. Dynamics of translation of single mRNA molecules in vivo. Cell 165, 976–989 (2016).
Wang, C., Han, B., Zhou, R. & Zhuang, X. Real-time imaging of translation on single mRNA transcripts in live cells. Cell 165, 990–1001 (2016).
Thoreen, C. C. et al. A unifying model for mTORC1-mediated regulation of mRNA translation. Nature 485, 109–113 (2012).
Thoreen, C. C. et al. An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J. Biol. Chem. 284, 8023–8032 (2009).
Yang, K., Neale, G., Green, D. R., He, W. & Chi, H. The tumor suppressor Tsc1 enforces quiescence of naive T cells to promote immune homeostasis and function. Nat. Immunol. 12, 888–897 (2011).
Iezzi, G., Karjalainen, K. & Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8, 89–95 (1998).
Harndahl, M., Rasmussen, M., Roder, G. & Buus, S. Real-time, high-throughput measurements of peptide–MHC-I dissociation using a scintillation proximity assay. J. Immunol. Methods 374, 5–12 (2011).
Lipford, J. R. & Deshaies, R. J. Diverse roles for ubiquitin-dependent proteolysis in transcriptional activation. Nat. Cell Biol. 5, 845–850 (2003).
Ingolia, N. T., Lareau, L. F. & Weissman, J. S. Ribosome profiling of mouse embryonic stem cells reveals the complexity and dynamics of mammalian proteomes. Cell 147, 789–802 (2011).
Sharova, L. V. et al. Database for mRNA half-life of 19 977 genes obtained by DNA microarray analysis of pluripotent and differentiating mouse embryonic stem cells. DNA Res. 16, 45–58 (2008).
Shiow, L. R. et al. CD69 acts downstream of interferon-α/β to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature 440, 540–544 (2006).
Ricciardi, S. et al. The Translational Machinery of Human CD4+ T Cells Is Poised for Activation and Controls the Switch from Quiescence to Metabolic Remodeling. Cell Metab. 28, 895–906.e5 (2018).
Tan, T. C. et al. Suboptimal T-cell receptor signaling compromises protein translation, ribosome biogenesis, and proliferation of mouse CD8 T cells. Proc. Natl Acad. Sci. USA 114, E6117–E6126 (2017).
Liebmann, M. et al. Nur77 serves as a molecular brake of the metabolic switch during T cell activation to restrict autoimmunity. Proc. Natl Acad. Sci. USA 115, E8017–E8026 (2018).
Kouzine, F. et al. Global regulation of promoter melting in naive lymphocytes. Cell 153, 988–999 (2013).
Shapiro, E., Biezuner, T. & Linnarsson, S. Single-cell sequencing-based technologies will revolutionize whole-organism science. Nat. Rev. Genet. 14, 618–630 (2013).
Akat, K. et al. Molecular characterization of desmosomes in meningiomas and arachnoidal tissue. Acta Neuropathol. 106, 337–347 (2003).
Hohenberg, H., Mannweiler, K. & Müller, M. High-pressure freezing of cell suspensions in cellulose capillary tubes. J. Microsc. 175, 34–43 (1994).
Cardona, A. et al. An integrated micro-and macroarchitectural analysis of the Drosophila brain by computer-assisted serial section electron microscopy. PLoS Biol. 8, e1000502 (2010).
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 116, 71–76 (1996).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Scheltema, R. A. et al. The Q Exactive HF, a benchtop mass spectrometer with a pre-filter, high-performance quadrupole and an ultra-high-field Orbitrap analyzer. Mol. Cell. Proteom. 13, 3698–3708 (2014).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Cox, J. et al. Andromeda: a peptide search engine integrated into the MaxQuant environment. J. Proteome Res. 10, 1794–1805 (2011).
Blecher-Gonen, R. et al. High-throughput chromatin immunoprecipitation for genome-wide mapping of in vivo protein–DNA interactions and epigenomic states. Nat. Protoc. 8, 539–554 (2013).
Barozzi, I., Termanini, A., Minucci, S. & Natoli, G. Fish the ChIPs: a pipeline for automated genomic annotation of ChIP-Seq data. Biol. Direct 6, 51 (2011).
Machanick, P. & Bailey, T. L. MEME-ChIP: motif analysis of large DNA datasets. Bioinformatics 27, 1696–1697 (2011).
McLean, C. Y. et al. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 28, 495–501 (2010).
Wiśniewski, J. R., Hein, M. Y., Cox, J. & Mann, M. A “proteomic ruler” for protein copy number and concentration estimation without spike-in standards. Mol. Cell. Proteom. 13, 3497–3506 (2014).
Acknowledgements
We thank D. Jarrossay for cell sorting. This work was supported by a grant from the Swiss SystemsX.ch initiative, evaluated by the Swiss National Science Foundation (grant no. 142032 to R.G.), the European Research Council (grant no. 803150 to R.G.), the Swiss National Science Foundation (grant no 170213 to F.S.), the Federal Ministry of Education and Research (GAIN_ 01GM1910A to B.G.) and the German Research Foundation (SFB1160/2_B5, RESIST–EXC 2155–Project ID 39087428 to B.G). The Institute for Research in Biomedicine is supported by the Helmut Horten Foundation.
Author information
Authors and Affiliations
Contributions
R.G. conceived the study, designed experiments, analyzed data and wrote the manuscript. T.W., W.J., G.Z., I.A.V. and J.C.R. designed and performed experiments with human T cells. M.A. and I.K. wrote the R shiny code for the online platform. M.B. helped with MS. C.K.E.B. and T.B. performed the electron microscopy and analysis. N.J.R. carried out ATAC-seq, and S.N. carried out ChIP-seq analysis. D.B., F.M., B.G., M.M., A.L., F.S., I.K. and R.G. supervised the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information L. A. Dempsey was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Viability of resting T cells in culture.
(a) Protein mass of naïve T cells does not change after 72 h of culturing. Proteomes of T cells that were either analyzed immediately after isolation or after 72 h of culturing were analyzed by LC-MS. Protein content was estimated using the proteome ruler approach59. n = 5 for freshly isolated and n = 2 for 72h-cultured T cells from different donors. Bars represent the S.E.M (b) FACS-purified naïve and memory T cells were cultured in complete medium without the addition of growth factors. To measure cell viability, T cells were stained with Annexin-V-FITC directly after sorting or after 24 h and 96 h of culturing and then analyzed by flow cytometry. n = 4, Four independent experiments with T cells from two different donors. Bars represent the S.E.M. (c) FACS-purified naïve T cells were cultured for 24 h in complete medium containing either DMSO, 50 µg/ml CHX or 50 µg/ml CHX together with 10 µM bortezomib (CHX + PS). n = 8 from three different donors. Bars represent the S.E.M.
Extended Data Fig. 2 Analysis of hallmark transcription factors.
(a) Frequency of ATAC-Seq peaks annotated to different genomic regions. (b) Frequency of ETS1 ChIP-Seq peaks annotated to different genomic regions. (c) Examples of activation markers that were up-regulated in TILs (CD69 and PD1) and proteins that were downregulated (KLF2, CD62L, and FAM65B). n = 1627 for T cells from blood, n = 2170 for T cells from tumors. Violin plot width is based on a Gaussian kernel density estimate of the data (estimated by the density function with standard parameters), scaled to have maximum width = 1. Data are from Zheng et al. 2016.
Extended Data Fig. 3 Increased turnover of glycolytic enzymes following activation.
(a) Naïve or 6h-activated T cells were analyzed on a Seahorse analyzer. Shown is the Extra Cellular Acidification Rate (ECAR), which is a measure of the glycolytic rate. n = 6 from three donors. Bars represent the S.E.M (b) Gene ontology analysis of the proteome of naïve T cells. Glycolytic proteins (blue) contributed 11% to the cytosolic protein mass. (c) Fraction of newly synthetized (heavily labeled) glycolytic proteins after a 12-hours pulse in naïve or 12h-activated T cells. n = 3 from three different donors. Box plot elements are defined as in Fig. 2b.
Extended Data Fig. 4 3D reconstructions of seven naïve and eight 72 h-activated T cells.
(a) Reconstructions of naïve CD4+ T cells. For the first two cells every layer of the plasma membrane (purple) was drawn, while for the other cells only every third layer was drawn. Scale bar = 2 μm (b) Reconstructions of 72 h-activated CD4+ T cells. For the first four cells every layer of the plasma membrane (purple) was drawn, while for the other cells only every third layer was drawn. Scale bar = 2 μm.
Extended Data Fig. 5 Estimation of the number of ribosomes.
(a) Copy numbers of 82 ribosomal proteins in naïve T cells. n = 7 from seven different donors. Box plot elements are defined as in Fig. 2b (b) Distribution of the copy numbers of ribosomal proteins in naïve T cells. Average values from n = 7 are shown. Dashed line shows the median, which was used as an approximation of total ribosomes. (c) Total RNA in naïve and activated T cells. To estimate the number of ribosomes, it was assumed that 83% of total RNA was ribosomal RNA. n = 7 for naïve, 6 h and 12h-activated T cells. n = 4 for 24 h, 48 h and 72 h activated T cells from different donors. Bars represent the S.E.M (d) mRNA to protein ratio. n = 7 for naïve, n = 3 for 6 h and n = 4 for 24 h activated T cells from different donors. Bars represent the S.E.M (e) Total amount of mRNA per T cell. n = 9 for naïve and 24 h activated T cells, n = 7 for 6 h and 12 h activated T cells from different donors. Bars represent the S.E.M (f) Average mRNA copy numbers per T cell. (g) mRNA processing rate in naïve and 6h-activated T cells. Average values from n = 3 from different donors are shown.
Extended Data Fig. 6 Posttranscriptional regulations.
(a) Absolute copy numbers of CD40LG and JUNB proteins in resting and activated T cells. n = 7 for naïve T cells, n = 3 for 6 h, 12 h, 48 h, 120h-activated T cells, n = 4 for 24 h, 72 h, 96 h activated T cells. Box plot elements are defined as in Fig. 2b (b) Absolute protein synthesis rates in resting and 6h-activated T cells that were untreated or treated with Torin-1. n = 3 from three donors. Bars represent the S.E.M.
Extended Data Fig. 7 Rapidly turned over proteins in naïve and memory T cells.
(a) Comparison of protein turnover kinetics in resting naïve and memory CD4+ T cells of selected proteins. n = 3 for naïve 6 h and naïve 12 h; n = 4 for naïve 24 h, memory 6 h, memory 12 h and memory 24 h from different donors.
Supplementary information
Supplementary Tables
Supplementary Tables 1–6.
Rights and permissions
About this article
Cite this article
Wolf, T., Jin, W., Zoppi, G. et al. Dynamics in protein translation sustaining T cell preparedness. Nat Immunol 21, 927–937 (2020). https://doi.org/10.1038/s41590-020-0714-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-020-0714-5
This article is cited by
-
The cell biology of HIV-1 latency and rebound
Retrovirology (2024)
-
Mitochondrial ATP generation is more proteome efficient than glycolysis
Nature Chemical Biology (2024)
-
The YAP–TEAD complex promotes senescent cell survival by lowering endoplasmic reticulum stress
Nature Aging (2023)
-
Spatially resolved mapping of proteome turnover dynamics with subcellular precision
Nature Communications (2023)
-
Dhx33 promotes B-cell growth and proliferation by controlling activation-induced rRNA upregulation
Cellular & Molecular Immunology (2023)