Abstract
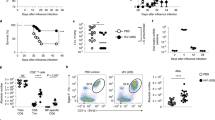
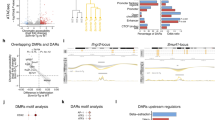
Sepsis and trauma cause inflammation and elevated susceptibility to hospital-acquired pneumonia. As phagocytosis by macrophages plays a critical role in the control of bacteria, we investigated the phagocytic activity of macrophages after resolution of inflammation. After resolution of primary pneumonia, murine alveolar macrophages (AMs) exhibited poor phagocytic capacity for several weeks. These paralyzed AMs developed from resident AMs that underwent an epigenetic program of tolerogenic training. Such adaptation was not induced by direct encounter of the pathogen but by secondary immunosuppressive signals established locally upon resolution of primary infection. Signal-regulatory protein α (SIRPα) played a critical role in the establishment of the microenvironment that induced tolerogenic training. In humans with systemic inflammation, AMs and also circulating monocytes still displayed alterations consistent with reprogramming six months after resolution of inflammation. Antibody blockade of SIRPα restored phagocytosis in monocytes of critically ill patients in vitro, which suggests a potential strategy to prevent hospital-acquired pneumonia.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Bulk RNA-seq data and epigenetic data have been deposited in the ArrayExpress Archive of Functional Genomics Data (accession code GSE147450). Clinical data and data sets generated for the study are stored on the secured server of the University of Nantes and are available from the corresponding authors upon reasonable request.
Change history
24 June 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41590-020-0739-9
References
Bekaert, M. et al. Attributable mortality of ventilator-associated pneumonia: a reappraisal using causal analysis. Am. J. Respir. Crit. Care Med. 184, 1133–1139 (2011).
GBD 2015 LRI Collaborators. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 17, 1133–1161 (2017).
Eber, M. R., Laxminarayan, R., Perencevich, E. N. & Malani, A. Clinical and economic outcomes attributable to health care–associated sepsis and pneumonia. Arch. Intern. Med. 170, 347–353 (2010).
Torres, A. et al. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia. Eur. Respir. J. 50, 1700582 (2017).
Kalil, A. C. et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin. Infect. Dis. 63, e61–e111 (2016).
Weiss, E., Essaied, W., Adrie, C., Zahar, J.-R. & Timsit, J.-F. Treatment of severe hospital-acquired and ventilator-associated pneumonia: a systematic review of inclusion and judgment criteria used in randomized controlled trials. Crit. Care 21, 162 (2017).
Roquilly, A., Marret, E., Abraham, E. & Asehnoune, K. Pneumonia prevention to decrease mortality in intensive care unit: a systematic review and meta-analysis. Clin. Infect. Dis. 60, 64–75 (2015).
Asehnoune, K. et al. Hydrocortisone and fludrocortisone for prevention of hospital-acquired pneumonia in patients with severe traumatic brain injury (Corti-TC): a double-blind, multicentre phase 3, randomised placebo-controlled trial. Lancet Respir. Med. 2, 706–716 (2014).
van Vught, L. A et al. Incidence, risk factors, and attributable mortality of secondary infections in the intensive care unit after admission for sepsis. JAMA 315, 1469–1479 (2016).
Roquilly, A. et al. Pathophysiological role of respiratory dysbiosis in hospital-acquired pneumonia. Lancet Respir. Med. 7, 710–720 (2019).
Roquilly, A. & Villadangos, J. A. The role of dendritic cell alterations in susceptibility to hospital-acquired infections during critical-illness related immunosuppression. Mol. Immunol. 68, 120–123 (2015).
Bouras, M., Asehnoune, K. & Roquilly, A. Contribution of dendritic cell responses to sepsis-induced immunosuppression and to susceptibility to secondary pneumonia. Front. Immunol. 9, 2590 (2018).
Hotchkiss, R. S., Monneret, G. & Payen, D. Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat. Rev. Immunol. 13, 862–874 (2013).
Roquilly, A. et al. Local modulation of antigen-presenting cell development after resolution of pneumonia induces long-term susceptibility to secondary infections. Immunity 47, 135–147.e5 (2017).
Kopf, M., Schneider, C. & Nobs, S. P. The development and function of lung-resident macrophages and dendritic cells. Nat. Immunol. 16, 36–44 (2014).
Roquilly, A. et al. Empiric antimicrobial therapy for ventilator-associated pneumonia after brain injury. Eur. Respir. J. 47, 1219–1228 (2016).
Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38, 792–804 (2013).
Yao, Y. et al. Induction of autonomous memory alveolar macrophages requires T cell help and is critical to trained immunity. Cell 175, 1634–1650.e17 (2018).
Kim, K.-W., Zhang, N., Choi, K. & Randolph, G. J. Homegrown macrophages. Immunity 45, 468–470 (2016).
van de Laar, L. et al. Yolk sac macrophages, fetal liver, and adult monocytes can colonize an empty niche and develop into functional tissue-resident macrophages. Immunity 44, 755–768 (2016).
Machiels, B. et al. A gammaherpesvirus provides protection against allergic asthma by inducing the replacement of resident alveolar macrophages with regulatory monocytes. Nat. Immunol. 18, 1310–1320 (2017).
Ma, K. C., Schenck, E. J., Pabon, M. A. & Choi, A. M. K. The role of danger signals in the pathogenesis and perpetuation of critical illness. Am. J. Respir. Crit. Care Med. 197, 300–309 (2018).
Cegelski, L., Marshall, G. R., Eldridge, G. R. & Hultgren, S. J. The biology and future prospects of antivirulence therapies. Nat. Rev. Microbiol. 6, 17–27 (2008).
Vega-Ramos, J. et al. Inflammation conditions mature dendritic cells to retain the capacity to present new antigens but with altered cytokine secretion function. J. Immunol. 193, 3851–3859 (2014).
Spörri, R. & Reis e Sousa, C. Inflammatory mediators are insufficient for full dendritic cell activation and promote expansion of CD4+ T cell populations lacking helper function. Nat. Immunol. 6, 163–170 (2005).
Kim, T. S., Gorski, S. A., Hahn, S., Murphy, K. M. & Braciale, T. J. Distinct dendritic cell subsets dictate the fate decision between effector and memory CD8+ T cell differentiation by a CD24-dependent mechanism. Immunity 40, 400–413 (2014).
Park, S. H. et al. Type I interferons and the cytokine TNF cooperatively reprogram the macrophage epigenome to promote inflammatory activation. Nat. Immunol. 18, 1104–1116 (2017).
Murray, P. J. et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41, 14–20 (2014).
Svedberg, F. R. et al. The lung environment controls alveolar macrophage metabolism and responsiveness in type 2 inflammation. Nat. Immunol. 20, 571–580 (2019).
Liu, M. et al. Metabolic rewiring of macrophages by CpG potentiates clearance of cancer cells and overcomes tumor-expressed CD47−mediated ‘don’t-eat-me’ signal. Nat. Immunol. 20, 265–275 (2019).
Novakovic, B. et al. β-glucan reverses the epigenetic state of LPS-induced immunological tolerance. Cell 167, 1354–1368.e14 (2016).
Netea, M. G et al. Defining trained immunity and its role in health and disease. Nat. Rev. Immunol. https://doi.org/10.1038/s41577-020-0285-6 (2020).
Saeed, S. et al. Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science 345, 1251086 (2014).
Bekkering, S. et al. Metabolic induction of trained immunity through the mevalonate pathway. Cell 172, 135–146.e9 (2018).
Netea, M. G. & Joosten, L. A. B. Trained immunity and local innate immune memory in the lung. Cell 175, 1463–1465 (2018).
Veillette, A. & Chen, J. SIRPα–CD47 immune checkpoint blockade in anticancer therapy. Trends Immunol. 39, 173–184 (2018).
Barclay, A. N. & Brown, M. H. The SIRP family of receptors and immune regulation. Nat. Rev. Immunol. 6, 457–464 (2006).
Bao, Y., Gao, Y., Shi, Y. & Cui, X. Dynamic gene expression analysis in a H1N1 influenza virus mouse pneumonia model. Virus Genes 53, 357–366 (2017).
Berrien-Elliott, M. M. et al. MicroRNA-142 is critical for the homeostasis and function of type 1 innate lymphoid cells. Immunity 51, 479–490.e6 (2019).
Mildner, A. et al. Mononuclear phagocyte miRNome analysis identifies miR-142 as critical regulator of murine dendritic cell homeostasis. Blood 121, 1016–1027 (2013).
Schliehe, C. et al. The methyltransferase Setdb2 mediates virus-induced susceptibility to bacterial superinfection. Nat. Immunol. 16, 67–74 (2014).
Sieweke, M. H. & Allen, J. E. Beyond stem cells: self-renewal of differentiated macrophages. Science 342, 1242974 (2013).
Amit, I., Winter, D. R. & Jung, S. The role of the local environment and epigenetics in shaping macrophage identity and their effect on tissue homeostasis. Nat. Immunol. 17, 18–25 (2016).
Davies, L. C., Jenkins, S. J., Allen, J. E. & Taylor, P. R. Tissue-resident macrophages. Nat. Immunol. 14, 986–995 (2013).
Reyfman, P. A. et al. Single-cell transcriptomic analysis of human lung provides insights into the pathobiology of pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 199, 1517–1536 (2019).
Hussell, T. & Bell, T. J. Alveolar macrophages: plasticity in a tissue-specific context. Nat. Rev. Immunol. 14, 81–93 (2014).
Netea, M. G., Schlitzer, A., Placek, K., Joosten, L. A. B. & Schultze, J. L. Innate and adaptive immune memory: an evolutionary continuum in the host’s response to pathogens. Cell Host Microbe 25, 13–26 (2019).
Foster, S. L., Hargreaves, D. C. & Medzhitov, R. Gene-specific control of inflammation by TLR-induced chromatin modifications. Nature 447, 972–978 (2007).
Medzhitov, R., Schneider, D. S. & Soares, M. P. Disease tolerance as a defense strategy. Science 335, 936–941 (2012).
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000).
Lahl, K. et al. Selective depletion of Foxp3+ regulatory T cells induces a scurfy-like disease. J. Exp. Med. 204, 57–63 (2007).
Ramalingam, R. et al. Dendritic cell-specific disruption of TGF-β receptor II leads to altered regulatory T cell phenotype and spontaneous multiorgan autoimmunity. J. Immunol. 189, 3878–3893 (2012).
Caton, M. L., Smith-Raska, M. R. & Reizis, B. Notch–RBP-J signaling controls the homeostasis of CD8− dendritic cells in the spleen. J. Exp. Med. 204, 1653–1664 (2007).
Inagaki, K. et al. SHPS-1 regulates integrin-mediated cytoskeletal reorganization and cell motility. EMBO J. 19, 6721–6731 (2000).
Malone, C. L. et al. Fluorescent reporters for Staphylococcus aureus. J. Microbiol Methods 77, 251–260 (2009).
Urban, J. H. & Vogel, J. Translational control and target recognition by Escherichia coli small RNAs in vivo. Nucleic Acids Res. 35, 1018–1037 (2007).
Janssen, W. J. et al. Fas determines differential fates of resident and recruited macrophages during resolution of acute lung injury. Am. J. Respir. Crit. Care Med. 184, 547–560 (2011).
Sun, W. et al. Histone acetylome-wide association study of autism spectrum disorder. Cell 167, 1385–1397.e11 (2016).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
McLean, C. Y. et al. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 28, 495–501 (2010).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 38, 576–589 (2010).
Allhoff, M., Seré, K., Pires, J. F., Zenke, M. & Costa, I. G.Differential peak calling of ChIP–seq signals with replicates with THOR. Nucleic Acids Res 44, e153 (2016).
Acknowledgements
We thank the Biological Resource Centre for biobanking (CHU Nantes, Hôtel Dieu, Centre de Ressources Biologiques (CRB), Nantes, France (BRIF: BB-0033-00040)), the Cytometry Facilty ‘Cytocell’, University of Nantes, and the Genomics and Bioinformatics Core Facility of Nantes (GenoBiRD, Biogenouest) for its technical support. OSE Immunotherapeutics provided Sirpa−/− mice and anti-Sirpa antibody but had no role in data analyses or revision of the manuscript. A.R. and J.P. received funding from the Region Pays de la Loire. This work was funded with grants from the National Health and Medical Research Council of Australia (NHMRC) to J.V., the Sylvia and Charles Viertel Foundation (Senior Medical Research Fellowship to A.K.), the Victorian State Government Operational Infrastructure Support and the Australian Government NHMRC Independent Research Institute Infrastructure Support scheme.
Author information
Authors and Affiliations
Contributions
C.J. performed experiments and contributed to the study design, data analyses, interpretation of results and writing and revision of the manuscript. M.D. and P.R. performed experiments and contributed to interpretation of results and revision of the manuscript. C.F. generated ChIP–seq and RNA-seq data. A.M., A.S. and J.P. performed bioinformatics analysis. A.B., B.M.-A., T.C., M.V., R.C., N.M., V.G., H.E.G.M. and F.A. contributed to interpretation of results and revision of the manuscript. A.R., C.J., J.P., J.A.V. and K.A. contributed to the study design, data analyses, interpretation of results and revision of the manuscript. All authors have approved the final manuscript for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Zoltan Fehervari was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Roquilly, A., Jacqueline, C., Davieau, M. et al. Alveolar macrophages are epigenetically altered after inflammation, leading to long-term lung immunoparalysis. Nat Immunol 21, 636–648 (2020). https://doi.org/10.1038/s41590-020-0673-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-020-0673-x
This article is cited by
-
Alveolar macrophage-expressed Plet1 is a driver of lung epithelial repair after viral pneumonia
Nature Communications (2024)
-
Physiological and immunological barriers in the lung
Seminars in Immunopathology (2024)
-
Fatal BK polyomavirus-associated pneumonia: report of two cases with literature review
BMC Infectious Diseases (2023)
-
RGT: a toolbox for the integrative analysis of high throughput regulatory genomics data
BMC Bioinformatics (2023)
-
Innate immune responses in pneumonia
Pneumonia (2023)