Abstract
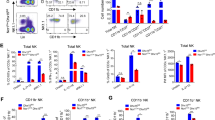
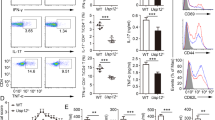
CD8+ T cells and natural killer (NK) cells are central cellular components of immune responses against pathogens and cancer, which rely on interleukin (IL)-15 for homeostasis. Here we show that IL-15 also mediates homeostatic priming of CD8+ T cells for antigen-stimulated activation, which is controlled by a deubiquitinase, Otub1. IL-15 mediates membrane recruitment of Otub1, which inhibits ubiquitin-dependent activation of AKT, a kinase that is pivotal for T cell activation and metabolism. Otub1 deficiency in mice causes aberrant responses of CD8+ T cells to IL-15, rendering naive CD8+ T cells hypersensitive to antigen stimulation characterized by enhanced metabolic reprograming and effector functions. Otub1 also controls the maturation and activation of NK cells. Deletion of Otub1 profoundly enhances anticancer immunity by unleashing the activity of CD8+ T cells and NK cells. These findings suggest that Otub1 controls the activation of CD8+ T cells and NK cells by functioning as a checkpoint of IL-15-mediated priming.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
RNA sequencing datasets were deposited to Gene Expression Omnibus with the accession code GSE126777. Other datasets generated during the current study are available from the corresponding author upon reasonable request. The human skin cutaneous melanoma datasets reported by other studies were downloaded from http://www.oncolnc.org/.
References
Durgeau, A., Virk, Y., Corgnac, S. & Mami-Chouaib, F. Recent advances in targeting CD8 T-cell immunity for more effective cancer immunotherapy. Front. Immunol. 9, 14 (2018).
Chiossone, L., Dumas, P. Y., Vienne, M. & Vivier, E. Natural killer cells and other innate lymphoid cells in cancer. Nat. Rev. Immunol. 18, 671–688 (2018).
Crouse, J., Xu, H. C., Lang, P. A. & Oxenius, A. NK cells regulating T cell responses: mechanisms and outcome. Trends Immunol. 36, 49–58 (2015).
Rosenberg, J. & Huang, J. CD8+ T cells and NK cells: parallel and complementary soldiers of immunotherapy. Curr. Opin. Chem. Eng. 19, 9–20 (2018).
Surh, C. D. & Sprent, J. Homeostasis of naive and memory T cells. Immunity 29, 848–862 (2008).
Castillo, E. F. & Schluns, K. S. Regulating the immune system via IL-15 transpresentation. Cytokine 59, 479–490 (2012).
Schluns, K. S., Kieper, W. C., Jameson, S. C. & Lefrancois, L. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat. Immunol. 1, 426–432 (2000).
Schluns, K. S. & Lefrancois, L. Cytokine control of memory T-cell development and survival. Nat. Rev. Immunol. 3, 269–279 (2003).
Liu, K., Catalfamo, M., Li, Y., Henkart, P. A. & Weng, N. P. IL-15 mimics T cell receptor crosslinking in the induction of cellular proliferation, gene expression, and cytotoxicity in CD8+ memory T cells. Proc. Natl Acad. Sci. USA 99, 6192–6197 (2002).
Deshpande, P. et al. IL-7- and IL-15-mediated TCR sensitization enables T cell responses to self-antigens. J. Immunol. 190, 1416–1423 (2013).
Teague, R. M. et al. Interleukin-15 rescues tolerant CD8+ T cells for use in adoptive immunotherapy of established tumors. Nat. Med. 12, 335–341 (2006).
Hu, H. & Sun, S. C. Ubiquitin signaling in immune responses. Cell Res. 26, 457–483 (2016).
Sun, S. C. Deubiquitylation and regulation of the immune response. Nat. Rev. Immunol. 8, 501–511 (2008).
Juang, Y. C. et al. OTUB1 co-opts Lys48-linked ubiquitin recognition to suppress E2 enzyme function. Mol. Cell 45, 384–397 (2012).
Nakada, S. et al. Non-canonical inhibition of DNA damage-dependent ubiquitination by OTUB1. Nature 466, 941–946 (2010).
Wang, T. et al. Evidence for bidentate substrate binding as the basis for the K48 linkage specificity of otubain 1. J. Mol. Biol. 386, 1011–1023 (2009).
Wiener, R., Zhang, X., Wang, T. & Wolberger, C. The mechanism of OTUB1-mediated inhibition of ubiquitination. Nature 483, 618–622 (2012).
Gubser, P. M. et al. Rapid effector function of memory CD8+ T cells requires an immediate–early glycolytic switch. Nat. Immunol. 14, 1064–1072 (2013).
Kim, E. H. & Suresh, M. Role of PI3K/Akt signaling in memory CD8 T cell differentiation. Front. Immunol. 4, 20 (2013).
Cammann, C. et al. Early changes in the metabolic profile of activated CD8+ T cells. BMC Cell Biol. 17, 28 (2016).
Hogquist, K. A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Lodolce, J. P., Burkett, P. R., Koka, R. M., Boone, D. L. & Ma, A. Regulation of lymphoid homeostasis by interleukin-15. Cytokine Growth Factor Rev. 13, 429–439 (2002).
Burkett, P. R. et al. IL-15Rα expression on CD8+ T cells is dispensable for T cell memory. Proc. Natl Acad. Sci. USA 100, 4724–4729 (2003).
Schluns, K. S. et al. Distinct cell types control lymphoid subset development by means of IL-15 and IL-15 receptor α expression. Proc. Natl Acad. Sci. USA 101, 5616–5621 (2004).
Goldrath, A. W. et al. Cytokine requirements for acute and basal homeostatic proliferation of naive and memory CD8+ T cells. J. Exp. Med. 195, 1515–1522 (2002).
Guillerey, C., Huntington, N. D. & Smyth, M. J. Targeting natural killer cells in cancer immunotherapy. Nat. Immunol. 17, 1025–1036 (2016).
Chiossone, L. et al. Maturation of mouse NK cells is a 4-stage developmental program. Blood 113, 5488–5496 (2009).
Polansky, J. K. et al. High dose CD11c-driven IL15 is sufficient to drive NK cell maturation and anti-tumor activity in a trans-presentation independent manner. Sci. Rep. 6, 19699 (2016).
Bottcher, J. P. et al. NK cells stimulate recruitment of cDC1 into the tumor microenvironment promoting cancer immune control. Cell 172, 1022–1037 (2018).
Vadlakonda, L., Dash, A., Pasupuleti, M., Anil Kumar, K. & Reddanna, P. The paradox of Akt–mTOR interactions. Front. Oncol. 3, 165 (2013).
Kim, E. H. et al. Signal integration by Akt regulates CD8 T cell effector and memory differentiation. J. Immunol. 188, 4305–4314 (2012).
Waldmann, T. A. The shared and contrasting roles of IL2 and IL15 in the life and death of normal and neoplastic lymphocytes: implications for cancer therapy. Cancer Immunol. Res. 3, 219–227 (2015).
Jethwa, N. et al. Endomembrane PtdIns(3,4,5)P3 activates the PI3K–Akt pathway. J. Cell Sci. 128, 3456–3465 (2015).
Cantley, L. C. The phosphoinositide 3-kinase pathway. Science 296, 1655–1657 (2002).
Carnero, A., Blanco-Aparicio, C., Renner, O., Link, W. & Leal, J. F. The PTEN/PI3K/AKT signalling pathway in cancer, therapeutic implications. Curr. Cancer Drug Targets 8, 187–198 (2008).
Balakirev, M. Y., Tcherniuk, S. O., Jaquinod, M. & Chroboczek, J. Otubains: a new family of cysteine proteases in the ubiquitin pathway. EMBO Rep. 4, 517–522 (2003).
Yang, W. L. et al. The E3 ligase TRAF6 regulates Akt ubiquitination and activation. Science 325, 1134–1138 (2009).
Calleja, V., Laguerre, M., Parker, P. J. & Larijani, B. Role of a novel PH–kinase domain interface in PKB/Akt regulation: structural mechanism for allosteric inhibition. PLoS Biol. 7, e17 (2009).
Roberts, D. J. & Miyamoto, S. Hexokinase II integrates energy metabolism and cellular protection: Akting on mitochondria and TORCing to autophagy. Cell Death Differ. 22, 248–257 (2015).
Pearce, E. L., Poffenberger, M. C., Chang, C. H. & Jones, R. G. Fueling immunity: insights into metabolism and lymphocyte function. Science 342, 1242454 (2013).
Zheng, Y., Delgoffe, G. M., Meyer, C. F., Chan, W. & Powell, J. D. Anergic T cells are metabolically anergic. J. Immunol. 183, 6095–6101 (2009).
McKinney, E. F. & Smith, K. G. C. Metabolic exhaustion in infection, cancer and autoimmunity. Nat. Immunol. 19, 213–221 (2018).
Huang, P. L. et al. Skeletal muscle interleukin 15 promotes CD8+ T-cell function and autoimmune myositis. Skelet. Muscle 5, 33 (2015).
Overwijk, W. W. et al. Tumor regression and autoimmunity after reversal of a functionally tolerant state of self-reactive CD8+ T cells. J. Exp. Med. 198, 569–580 (2003).
Zhang, P., Cote, A. L., de Vries, V. C., Usherwood, E. J. & Turk, M. J. Induction of postsurgical tumor immunity and T-cell memory by a poorly immunogenic tumor. Cancer Res. 67, 6468–6476 (2007).
Maueroder, C. et al. Tumor immunotherapy: lessons from autoimmunity. Front. Immunol. 5, 212 (2014).
Restifo, N. P., Dudley, M. E. & Rosenberg, S. A. Adoptive immunotherapy for cancer: harnessing the T cell response. Nat. Rev. Immunol. 12, 269–281 (2012).
Mishra, A., Sullivan, L. & Caligiuri, M. A. Molecular pathways: interleukin-15 signaling in health and in cancer. Clin. Cancer Res. 20, 2044–2050 (2014).
Dubois, S., Mariner, J., Waldmann, T. A. & Tagaya, Y. IL-15Rα recycles and presents IL-15 in trans to neighboring cells. Immunity 17, 537–547 (2002).
Yu, J. et al. Regulation of T-cell activation and migration by the kinase TBK1 during neuroinflammation. Nat. Commun. 6, 6074 (2015).
Pearce, E. L. & Shen, H. Generation of CD8 T cell memory is regulated by IL-12. J. Immunol. 179, 2074–2081 (2007).
Reiley, W. W. et al. Deubiquitinating enzyme CYLD negatively regulates the ubiquitin-dependent kinase Tak1 and prevents abnormal T cell responses. J. Exp. Med. 204, 1475–1485 (2007).
Xiao, G., Harhaj, E. W. & Sun, S. C. NF-κB-inducing kinase regulates the processing of NF-κB2 p100. Mol. Cell. 7, 401–409 (2001).
Acknowledgements
We thank S. Dubois (NCI/NIH) and Q. Yi (Cleveland Clinic) for cell lines and D. Durocher (Lunenfeld–Tanenbaum Research Institute) and P. Martin (University of Nice Sophia Antipolis) for plasmids. This study was supported by NIH grant AI64639 (to S.-C.S.) and partially supported by the following grants: National Institutes of Health grants AI057555 and GM84459 (to S.-C.S.), AI121458-01A1 (to K.S.) and AI145287 (to P.L.); Cancer Prevention & Research Institute of Texas grant RP160188 (to K.S.); Bridge Biotherapeutics (to S.-C.S.); Mission Therapeutics (to S.-C.S.); and Nektar Therapeutics (to K.S.). This study was also supported by NIH/NCI grant P30 CA016672 and used the flow cytometry, sequencing and microarray, and animal facility of the Cancer Center Support Grant-shared resources at the MD Anderson Cancer Center.
Author information
Authors and Affiliations
Contributions
X.Z. designed and performed the research, prepared the figures and wrote part of the manuscript; J.Y. performed research; X.C. contributed to the generation and maintenance of Otub1fl/fl mice; G.C.M., L.Z. and J.W. contributed to the RNA sequencing data analysis; K.S., B.Z. and P. L. contributed critical reagents; and S.-C.S. supervised the work and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.Y. is an employee of Therapeutic Tumor Microenvironment Strategies, and S.-C.S. received research funding from Bridge Biotherapeutics and Mission Therapeutics. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–12 and Supplementary table 1
Rights and permissions
About this article
Cite this article
Zhou, X., Yu, J., Cheng, X. et al. The deubiquitinase Otub1 controls the activation of CD8+ T cells and NK cells by regulating IL-15-mediated priming. Nat Immunol 20, 879–889 (2019). https://doi.org/10.1038/s41590-019-0405-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-019-0405-2
This article is cited by
-
Lung cancer cell-intrinsic IL-15 promotes cell migration and sensitizes murine lung tumors to anti-PD-L1 therapy
Biomarker Research (2024)
-
NK cells as powerful therapeutic tool in cancer immunotherapy
Cellular Oncology (2024)
-
Partial otubain 1 deficiency compromises fetal well-being in allogeneic pregnancies despite no major changes in the dendritic cell and T cell compartment
BMC Research Notes (2022)
-
CD4+ T cells are required to improve the efficacy of CIK therapy in non-small cell lung cancer
Cell Death & Disease (2022)
-
OTUB1 suppresses Hippo signaling via modulating YAP protein in gastric cancer
Oncogene (2022)