Abstract
Cachexia represents a leading cause of morbidity and mortality in various cancers, chronic inflammation and infections. Understanding of the mechanisms that drive cachexia has remained limited, especially for infection-associated cachexia (IAC). In the present paper we describe a model of reversible cachexia in mice with chronic viral infection and identify an essential role for CD8+ T cells in IAC. Cytokines linked to cancer-associated cachexia did not contribute to IAC. Instead, virus-specific CD8+ T cells caused morphologic and molecular changes in the adipose tissue, which led to depletion of lipid stores. These changes occurred at a time point that preceded the peak of the CD8+ T cell response and required T cell–intrinsic type I interferon signaling and antigen-specific priming. Our results link systemic antiviral immune responses to adipose-tissue remodeling and reveal an underappreciated role of CD8+ T cells in IAC.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The accession number for the raw data of the RNA-seq is GSE118819.
References
Von Haehling, S. & Anker, S. D. Treatment of cachexia: an overview of recent developments. Int. J. Cardiol. 184, 726–742 (2014).
Baracos, V. E., Martin, L., Korc, M., Guttridge, D. C. & Fearon, K. C. H. Cancer-associated cachexia. Nat. Rev. Dis. Primers 4, 1–18 (2018).
Fearon, K. et al. Definition and classifi cation of cancer cachexia: an international consensus. Lancet Oncol. 12, 489–495 (2011).
Kotler, D. Challenges to diagnosis of HIV-associated wasting. J. Acquir. Immune Defic. Syndr. 37, S280–S283 (2004).
Tisdale, M. J. Cachexia in cancer patients. Nat. Rev. Cancer 2, 862–871 (2002).
Porporato, P. E. Understanding cachexia as a cancer metabolism syndrome. Oncogenesis 5, 200 (2016).
Morley, J. E., Thomas, D. R. & Wilson, M.-M. G. Cachexia: pathophysiology and clinical relevance. Am. J. Clin. Nutr. 83, 735–743 (2006).
Fearon, K. C. H., Glass, D. J. & Guttridge, D. C. Cancer cachexia: Mediators, signaling, and metabolic pathways. Cell Metab. 16, 153–166 (2012).
Vaitkus, J. A. & Celi, F. S. The role of adipose tissue in cancer-associated cachexia. Exp. Biol. Med. 242, 473–481 (2017).
Petruzzelli, M. & Wagner, E. F. Mechanisms of metabolic dysfunction in cancer-associated cachexia. Genes Dev. 30, 489–501 (2016).
Flint, T. R., Fearon, D. T. & Janowitz, T. Connecting the metabolic and immune responses to cancer. Trends Mol. Med. 23, 451–464 (2017).
Bergthaler, A. et al. Viral replicative capacity is the primary determinant of lymphocytic choriomeningitis virus persistence and immunosuppression. Proc. Natl Acad. Sci. USA 107, 21641–21646 (2010).
Stamm, A., Valentine, L., Potts, R. & Premenko-Lanier, M. An intermediate dose of LCMV clone 13 causes prolonged morbidity that is maintained by CD4+T cells. Virology 425, 122–132 (2012).
Zechner, R. et al. FAT SIGNALS—lipases and lipolysis in lipid metabolism and signaling. Cell Metab. 15, 279–291 (2012).
Tsoli, M. et al. Depletion of white adipose tissue in cancer cachexia syndrome is associated with inflammatory signaling and disrupted circadian regulation. PLoS ONE 9, e92966 (2014).
Das, S. K. et al. Adipose triglyceride lipase contributes to cancer-associated cachexia. Science 333, 233–238 (2011).
Ruud, J. & Brüning, J. C. Metabolism: light on leptin link to lipolysis. Nature 527, 43–44 (2015).
Baatar, D., Patel, K. & Taub, D. D. The effects of ghrelin on inflammation and the immune system. Mol. Cell Endocrinol. 340, 44–58 (2011).
Kandarian, S. C. et al. Tumour-derived leukaemia inhibitory factor is a major driver of cancer cachexia and morbidity in C26 tumour-bearing mice. J. Cachexia Sarcopenia Muscle 9, 1–12 (2018).
Patel, H. J. & Patel, B. M. TNF-α and cancer cachexia: molecular insights and clinical implications. Life Sci. 170, 56–63 (2016).
Bhattacharya, A. et al. Superoxide dismutase 1 protects hepatocytes from type i interferon-driven oxidative damage. Immunity 43, 974–986 (2015).
Curtsinger, J. M., Valenzuela, J. O., Agarwal, P., Lins, D. & Mescher, M. F. Cutting edge: type I IFNS provide a third signal to CD8 T cells to stimulate clonal expansion and differentiation. J. Immunol. 174, 4465–4469 (2005).
Kolumam, G. A., Thomas, S., Thompson, L. J., Sprent, J. & Murali-Krishna, K. Type I interferons act directly on CD8 T cells to allow clonal expansion and memory formation in response to viral infection. J. Exp. Med. 202, 637–650 (2005).
Xu, C. et al. Direct effect of glucocorticoids on lipolysis in adipocytes. Mol. Endocrinol. 23, 1161–1170 (2009).
Pinschewer, D. D. et al. FTY720 immunosuppression impairs effector T cell peripheral homing without affecting induction, expansion, and memory. J. Immunol. 164, 5761–5770 (2000).
Petruzzelli, M. et al. A switch from white to brown fat increases energy expenditure in cancer-associated cachexia. Cell Metab. 20, 433–447 (2014).
Kir, S. et al. PTH/PTHrP receptor mediates cachexia in models of kidney failure and cancer. Cell Metab. 23, 315–323 (2016).
Cao, Y. Angiogenesis and vascular functions in modulation of obesity, adipose metabolism, and insulin sensitivity. Cell Metab. 18, 478–489 (2013).
Vegiopoulos, A., Rohm, M. & Herzig, S. Adipose tissue: between the extremes. EMBO J. 36, 1999–2017 (2017).
Quatrini, L. et al. Endogenous glucocorticoids control host resistance to viral infection through the tissue-specific regulation of PD-1 expression on NK cells. Nat. Immunol. 19, 954–962 (2018).
Miller, A. H. et al. Effects of viral infection on corticosterone secretion and glucocorticoid receptor binding in immune tissues. Psychoneuroendocrinology 22, 455–474 (1997).
Jamieson, A. M., Yu, S., Annicelli, C. H. & Medzhitov, R. Influenza virus-induced glucocorticoids compromise innate host defense against a secondary bacterial infection. Cell Host Microbe 7, 103–114 (2010).
Doherty, P. C., Hou, S. & Southern, P. J. Lymphocytic choriomeningitis virus induces a chronic wasting disease in mice lacking class I major histocompatibility complex glycoproteins. J. Neuroimmunol. 46, 11–17 (1993).
Hildeman, D. & Muller, D. Immunopathologic weight loss in intracranial LCMV infection initiated by the anorexigenic effects of IL-1β. Viral Immunol. 13, 273–285 (2000).
Kamperschroer, C. & Quinn, D. G. The role of proinflammatory cytokines in wasting disease during lymphocytic choriomeningitis virus infection. J. Immunol. 169, 340–349 (2002).
Wang, A. et al. Opposing effects of fasting metabolism on tissue tolerance in bacterial and viral inflammation. Cell 166, 1512–1525.e12 (2016).
Pietrocola, F. et al. Caloric restriction mimetics enhance anticancer immunosurveillance. Cancer Cell 30, 147–160 (2016).
Nencioni, A., Caffa, I., Cortellino, S. & Longo, V. D. Fasting and cancer: molecular mechanisms and clinical application. Nat. Rev. Cancer 18, 707–719 (2018).
Longo, V. D. & Mattson, M. P. Fasting: molecular mechanisms and clinical applications. Cell Metab. 19, 181–192 (2014).
Rouse, B. T. & Sehrawat, S. Immunity and immunopathology to viruses: what decides the outcome? Nat. Rev. Immunol. 10, 514–526 (2010).
Virgin, H. W., Wherry, E. J. & Ahmed, R. Redefining chronic viral infection. Cell 138, 30–50 (2009).
Sullivan, B. M., Teijaro, J. R., De La Torre, J. C. & Oldstone, M. B. A. Early virus–host interactions dictate the course of a persistent infection. PLoS Pathog. 11, 1004588 (2015).
Danai, L. V. et al. Altered exocrine function can drive adipose wasting in early pancreatic cancer. Nature 558, 600–604 (2018).
Wherry, E. J. & Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 15, 486–499 (2015).
Perry, R. J. et al. Hepatic acetyl CoA links adipose tissue inflammation to hepatic insulin resistance and type 2 diabetes. Cell 160, 745–758 (2015).
Norata, G. D. et al. The cellular and molecular basis of translational immunometabolism. Immunity 43, P421–P434 (2015).
Buck, M. D., Sowell, R. T., Kaech, S. M. & Pearce, E. L. Metabolic instruction of immunity. Cell 169, 570–586 (2017).
Huang, S. et al. Immune response in mice that lack the interferon-gamma receptor. Science 259, 1742–1745 (1993).
Pasparakis, M., Alexopoulou, L., Episkopou, V. & Kollias, G. Immune and inflammatory responses in TNF alpha-deficient mice: a critical requirement for TNF alpha in the formation of primary B cell follicles, follicular dendritic cell networks and germinal centers, and in the maturation of the humoral immune response. J. Exp. Med. 184, 1397–1411 (1996).
Peschon, J. J. et al. TNF receptor-deficient mice reveal divergent roles for p55 and p75 in several models of inflammation. J. Immunol. 160, 943–952 (1998).
Muller, U. et al. Functional role of type I and type II interferons in antiviral defense. Science 264, 1918–1921 (1994).
Prigge, J. R. et al. Type I IFNs act upon hematopoietic progenitors to protect and maintain hematopoiesis during pneumocystis lung infection in mice. J. Immunol. 195, 5347–5357 (2015).
Sitnick, M. T. et al. Skeletal muscle triacylglycerol hydrolysis does not influence metabolic complications of obesity. Diabetes 62, 3350–3361 (2013).
Eguchi, J. et al. Transcriptional control of adipose lipid handling by IRF4. Cell Metab. 13, 249–259 (2011).
Sawada, S., Scarborough, J. D., Killeen, N. & Littman, D. R. A lineage-specific transcriptional silencer regulates CD4 gene expression during T lymphocyte development. Cell 77, 917–929 (1994).
Coleman, D. L. & Hummel, K. The influence of genetic background on the expression of the obese (Ob) gene in the mouse. Diabetologia 9, 287–293 (1973).
Komuro, K., Itakura, K., Boyse, E. A. & John, M. Ly-5: a new T-lymphocyte antigen system. Immunogenetics 1, 452–456 (1974).
Fung-Leung, W. et al. CD8 is needed for development of cytotoxic T but not helper T cells. Cell 65, 443–449 (1991).
Shinkai, Y. et al. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell 68, 855–867 (1992).
Hogquist, K. A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Mombaerts, P. et al. RAG-l-deficient mice have no mature B and T lymphocytes. Cell 68, 869–877 (1992).
Kägi, D. et al. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature 369, 31–37 (1994).
Smyth, G. K. Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet. Mol. Biol. 3, 1–25 (2004).
Acknowledgements
We thank S. Niggemeyer, S. Jungwirth and J. Riede for animal husbandry. The authors would like to thank L. Bakiri, V. Baracos, Y. Belkaid, R. Medzhitov, G. Superti-Furga, E. Wagner and R. M. Zinkernagel for valuable feedback and discussions. This project has received funding from the European Research Council under the European Union’s Seventh Framework Programme and Horizon 2020 research and innovation program (grant agreement no. 677006, ‘CMIL’ to A.B., no. 340896 and ‘LipoCheX’ to R. Z.) from the German Research Council (grant nos. SFB974, KFO217, LA-2558/5-1 and Jürgen Manchot Graduate School MOI III to P.A.L.), from the Austrian Science Fund (grant no. FWF P26766 to T.S.) and from the US National Institutes of Health (grant nos. R01AI032972, U19AI100627 to A. Aderem). A.L. and M. Smyth are supported by DOC fellowships of the Austrian Academy of Sciences.
Author information
Authors and Affiliations
Contributions
H.B. conceived the project, designed and performed experiments, analyzed the data and wrote the manuscript. M. Schweiger, T.S., B.V., A. Aderem and R.Z. contributed to the experimental design, shared reagents and/or contributed to data interpretation. M.M., H.X., K.K., L.K., M. Smyth, A.L. and P.A.L. designed, performed and/or analyzed experiments. A.P. performed the bioinformatic data analyses. S.G., A. Ali and M.H. performed metabolic cage measurements. J.F. and T.H.H. performed MRI. D.M. provided histologic and immunohistochemical staining. A.B. conceived the project, designed experiments, analyzed the data, wrote the manuscript and supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
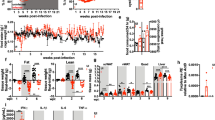
Supplementary Fig. 1 Characterization of the pathophysiologic changes induced by IAC.
a) Activity, oxygen consumption (VO2) and respiratory exchange ratio (RER) of LCMV-infected mice compared to uninfected controls (n = 10). Data are representative of a single experiment. b) Circulating viral load of LCMV-infected mice, measured using focus-forming assay (n = 4). Data are representative of two independent experiments. c,d) LCMV nuclear protein as measured by qPCR from tissue homogenate of inguinal, gonadal, and interscapular-brown adipose tissue up to 8 days post-infection, and 17 days post-infection (column graph) (n = 5), representative of three independent experiments (c), as well as quadriceps, gastrocnemius and soleus muscles (n = 5) representative of two independent experiments. e-g) RNA expression of myoblast differentiation 1 (MoyD1), and the proteasomal degradation markers Atrogin1 (Fbxo32) and Murf1 (Trim63) in the indicated muscle compartment (n = 5) (p-values: **0.0025, **0.0029, *0.0252, **0.0059 (e), **0.0087, **0.0071 (f), ***0.0003, *0.0295 (g) one-way ANOVA, Bonferroni correction). h) Body weight kinetics of mice infected with a titrated dose of LCMV-Cl13, ranging from 2 × 106 FFU to 2 × 102 FFU (n = 3), (p-values: **** < 0.0001, **0.0021 two-way ANOVA). i) Body weight kinetics of mice infected with high dose LCMV-Cl13 compared to LCMV-ARM (n = 4), (p-values: ****< 0.0001 two-way ANOVA). h-i) data are representative of three independent experiments. j) Body weight kinetics Of LCMV-infected mice after gavage supplementation of indicated diet between 4 and 7 days after infection (n = 4) (p-values: **0.0029, ****< 0.0001, two-way ANOVA) Data are representative of a single experiment. All data shows mean ± s.e.m.
Supplementary Fig. 2 IAC triggers severe adipose tissue remodeling and alters leptin expression and concentration.
a) Inguinal fat pad in LCMV-infected mice at 6 and 8 days post-infection compared to pair-fed mice. Similar results were observed across all experiments using LCMV-infected wild-type mice. b,c) Representative H/E staining taken from inguinal fat pad (b) and gonadal fat pad (c) at 6 and 8 days post-infection compared to uninfected controls (n = 3). d,e) Serum concentration of ghrelin (n = 5) (d) and leptin (n = 6) (e) as measured by ELISA in infected and uninfected mice (p-values: ***0.0002, ****<0.00001 one-way ANOVA, Bonferroni correction). Data are pooled from two independent experiments. f) Leptin (Lep) and Adiponectin (Adipoq) mRNA expression in inguinal fat pad of infected and uninfected mice, calculated from arbitrary units normalized to Ribosomal protein (Rplp0) and body weight (n = 4) (p-values: **0.0018 one-way ANOVA, Bonferroni correction). Data are representative of a single experiment. g,h) Infection of leptin knockout mice (LepOb/Ob) and heterogeneous control (LepOb/+) showing body weight kinetics and food intake (n = 4), representative of two independent experiments (g) and body composition as measured in live un-anesthetized mice using EchoMRI (n = 4), representative of a single experiment (h). d–h) data shows mean ± s.e.m.
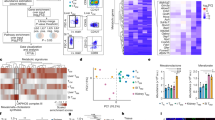
Supplementary Fig. 3 The role of infection-induced proinflammatory cytokines and T cells in mediating weight loss during LCMV clone 13 and influenza infection.
a) Serum cytokines of LCMV-infected mice measured using Luminex multiplexing immunoassays (n = 4). Data are representative of a single experiment. b) Percent of initial body weight at 8 days post infection in genetic knockout and neutralizing antibody-treated mice for the indicated cytokines and cytokine receptors (n = 4) (p-values: *0.0122 unpaired two-tailed Student’s t-test). Data shows a summary of figure 2b-2d. c) Circulating viral load as measured using focus-forming assay at 8 days after infection. (n = 5) for antibody depletion and (n = 4) for Ifnar1−/− (p-values: **0.0092, *0.0371, **0.0015 unpaired two-tailed t-test). d) Percent of circulating CD4+ T cells or CD8+ as indicated, following treatment with either CD4 blocking antibody (n = 3) or CD8 blocking antibody (n = 4) respectively. e) Percent of initial body weight of mice treated with either anti-CD4 or anti-CD8 depleting antibodies, as well as CD8−/− (n = 4). f) Splenic viral load as measured with focus-forming assay at 8 days after infection. (n = 3) for anti-CD4 treated mice and (n = 4) for others. c-f) Data are representative of two independent experiments for antibody depletions and Ifnar1−/−, and a single experiment for Ifng−/−, Tnf−/−, TnfrI−/− and Cd8−/−. g,h) Body weight kinetics of WT and Rag2−/− mice infected with LCMV-Cl13 (n = 5) (g) or Influenza PR/8 (n = 4) (h) (p-values: ****< 0.0001 two-way ANOVA). Data are representative of two independent experiments. i) Body weight kinetics and j) food intake of influenza-infected mice compared to pair-fed uninfected mice up to 8 days post-infection (n = 4), data represent a single experiment. Data shows mean ± s.e.m.
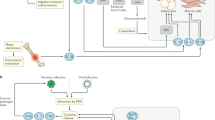
Supplementary Fig. 4 Loss of T cell–intrinsic type I IFN signaling abrogates infection-induced adipose tissue lipolysis.
a) Body weight kinetics of Ifnar1fl/flAdipoqCre/+ mice in comparison to Ifnar1fl/fl controls (n = 4) Data are representative of two independent experiments. b, c) FACS analysis of CD8+ T cells in spleen (n = 4) (p-values: ***0.0005, **0.0017, **0.0059, *0.0124 two-way ANOVA, Bonferroni correction) (b) and inguinal LN (n = 4) (p-values: **0.0049 two-way ANOVA, Bonferroni correction) (c), harvested on 6 days post-infection from Ifnar1fl/flCD4Cre/+ and Ifnar1fl/fl controls. Data represents a single experiment. d) Schematic representation of fasting lipolysis, showing circulating cortisol (p-values: **0.0010, *0.137 two-way ANOVA, Bonferroni correction), corticosterone (p-values: ***0.0001, ***0.0004 two-way ANOVA, Bonferroni correction), norepinephrine (p-values: ***0.0004 two-way ANOVA, Bonferroni correction), free T3 (p-values: **0.0036 two-way ANOVA, Bonferroni correction) and free T4 levels, as well as adipose tissue norepinephrine (n = 3). Data are representative of two independent experiments. RNA-seq data (n = 3) shows mRNA expression of β-AR (Abrd2) (p-values: ***0.0001 one way ANOVA, Bonferroni correction), GNAS (p-values: ****< 0.0001, **0.0014 one-way ANOVA, Bonferroni correction), ATGL (Pnpla2), CGI-58 (Abhd5) (p-values: *0.0271, *0.048 one-way ANOVA, Bonferroni correction), G0S2 (G0s2) and HSL (Lipe), in addition to protein expression of ATGL, HSL, pHSL and Perilipin. Western blot data are representative of two independent experiments were (n = 3). Data shows mean ± s.e.m. for bar graphs and (a).
Supplementary Fig. 5 CD8 T cell egress from lymph nodes is dispensable for the induction of IAC, but antigen-specific activation is required.
a–d) Virus-specific CD3+CD8+ T cells of LCMV-infected and uninfected mice after daily gavage administration of either FTY720 or water. Cell were isolated from blood at day 6 (n = 5) (p-values: **** < 0.0001, **0.0083 two-way ANOVA, Bonferroni correction) (a) and day 8 post infection (n = 5) (p-values: ****<0.0001, **0.0013, **0.0012 two-way ANOVA, Bonferroni correction) (b). At day 8 post-infection, cells were also isolated from inguinal LN (n = 5) (p-values: **** < 0.0001, **0.0036, *0.0142 two way ANOVA, Bonferroni correction) (c) and spleen (n = 5) (p-values: ****<0.0001, ***0.0002 two way ANOVA, Bonferroni correction) (d). e, f) Flow cytometry analysis showing the percent of CD8+ T cells carrying CD45.1+ vs CD45.2+ congenic markers after bone marrow reconstitution in indicated chimeras (n = 6) (p-values: ****<0.0001 unpaired two-tailed Student’s t-test). g,h) Percentage and total number of GP33+CD8+ T cells in chimeric mice at 12 days after LCMV infection (n = 6) (p-values: **0.0038, *0.0282 unpaired two-tailed Student’s t-test). e-h) Data are pooled from two independent experiments. All data shows mean ± s.e.m.
Supplementary Fig. 6 Unprocessed images of all western blots.
(left) unprocessed image acquired for indicated antibodies. (right) merge images show the chemo-luminescence image automatically merged with ladder image as acquired using Bio-Rad ChemiDocTM XRS+ system.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6 and Supplementary Table 1
Rights and permissions
About this article
Cite this article
Baazim, H., Schweiger, M., Moschinger, M. et al. CD8+ T cells induce cachexia during chronic viral infection. Nat Immunol 20, 701–710 (2019). https://doi.org/10.1038/s41590-019-0397-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-019-0397-y
This article is cited by
-
Low muscle mass in lung cancer is associated with an inflammatory and immunosuppressive tumor microenvironment
Journal of Translational Medicine (2023)
-
Adipocyte lipolysis protects mice against Trypanosoma brucei infection
Nature Microbiology (2023)
-
IFNγ-induction of TH1-like regulatory T cells controls antiviral responses
Nature Immunology (2023)
-
Pyrimidine de novo synthesis inhibition selectively blocks effector but not memory T cell development
Nature Immunology (2023)
-
Pathological features of tissues and cell populations during cancer cachexia
Cell Regeneration (2022)