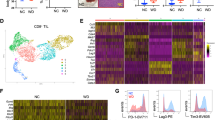
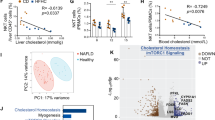
Abstract
Up to 49% of certain types of cancer are attributed to obesity, and potential mechanisms include overproduction of hormones, adipokines, and insulin. Cytotoxic immune cells, including natural killer (NK) cells and CD8+ T cells, are important in tumor surveillance, but little is known about the impact of obesity on immunosurveillance. Here, we show that obesity induces robust peroxisome proliferator-activated receptor (PPAR)-driven lipid accumulation in NK cells, causing complete ‘paralysis’ of their cellular metabolism and trafficking. Fatty acid administration, and PPARα and PPARδ (PPARα/δ) agonists, mimicked obesity and inhibited mechanistic target of rapamycin (mTOR)-mediated glycolysis. This prevented trafficking of the cytotoxic machinery to the NK cell–tumor synapse. Inhibiting PPARα/δ or blocking the transport of lipids into mitochondria reversed NK cell metabolic paralysis and restored cytotoxicity. In vivo, NK cells had blunted antitumor responses and failed to reduce tumor growth in obesity. Our results demonstrate that the lipotoxic obese environment impairs immunosurveillance and suggest that metabolic reprogramming of NK cells may improve cancer outcomes in obesity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Global status report on noncommunicable diseases 2014 (WHO, 2004).
Calle, E. E. & Kaaks, R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat. Rev. Cancer 4, 579–591 (2004).
Renehan, A. G., Tyson, M., Egger, M., Heller, R. F. & Zwahlen, M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet 371, 569–578 (2008).
Falagas, M. E. & Kompoti, M. Obesity and infection. Lancet. Infect. Dis. 6, 438–446 (2006).
Sjostrom, L. et al. Effects of bariatric surgery on cancer incidence in obese patients in Sweden (Swedish Obese Subjects Study): a prospective, controlled intervention trial. Lancet Oncol. 10, 653–662 (2009).
Renehan, A. G., Zwahlen, M. & Egger, M. Adiposity and cancer risk: new mechanistic insights from epidemiology. Nat. Rev. Cancer 15, 484–498 (2015).
Park, J., Morley, T. S., Kim, M., Clegg, D. J. & Scherer, P. E. Obesity and cancer mechanisms underlying tumour progression and recurrence. Nat. Rev. Endocrinol. 10, 455–465 (2014).
Pascual, G. et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 541, 41–45 (2017).
Beyaz, S. et al. High-fat diet enhances stemness and tumorigenicity of intestinal progenitors. Nature 531, 53–58 (2016).
Vivier, E., Tomasello, E., Baratin, M., Walzer, T. & Ugolini, S. Functions of natural killer cells. Nat. Immunol. 9, 503–510 (2008).
Mace, E. M. et al. Cell biological steps and checkpoints in accessing NK cell cytotoxicity. Immunol. Cell Biol. 92, 245–255 (2014).
Orange, J. S. Formation and function of the lytic NK-cell immunological synapse. Nat. Rev. Immunol. 8, 713–725 (2008).
Chowdhury, D. & Lieberman, J. Death by a thousand cuts: granzyme pathways of programmed cell death. Annu. Rev. Immunol. 26, 389–420 (2008).
Donnelly, R. P. et al. mTORC1-dependent metabolic reprogramming is a prerequisite for NK cell effector function. J. Immunol. 193, 4477–4484 (2014).
Marcais, A. et al. The metabolic checkpoint kinase mTOR is essential for IL-15 signaling during the development and activation of NK cells. Nat. Immunol. 15, 749–757 (2014).
Bjorntorp, P., Bergman, H. & Varnauskas, E. Plasma free fatty acid turnover rate in obesity. Acta Med. Scand. 185, 351–356 (1969).
Jensen, M. D., Haymond, M. W., Rizza, R. A., Cryer, P. E. & Miles, J. M. Influence of body fat distribution on free fatty acid metabolism in obesity. J. Clin. Invest. 83, 1168–1173 (1989).
Lynch, L. A. et al. Are natural killer cells protecting the metabolically healthy obese patient? Obesity (Silver Spring) 17, 601–605 (2009).
O’Shea, D., Cawood, T. J., O’Farrelly, C. & Lynch, L. Natural killer cells in obesity: impaired function and increased susceptibility to the effects of cigarette smoke. PloS ONE 5, e8660 (2010).
Jahn, J. et al. Decreased NK cell functions in obesity can be reactivated by fat mass reduction. Obesity (Silver Spring) 23, 2233–2241 (2015).
Perdu, S. et al. Maternal obesity drives functional alterations in uterine NK cells. JCI Insight 1, e85560 (2016).
Laplante, M. & Sabatini, D. M. mTOR signaling in growth control and disease. Cell 149, 274–293 (2012).
Finlay, D. K. et al. PDK1 regulation of mTOR and hypoxia-inducible factor 1 integrate metabolism and migration of CD8+ T cells. J. Exp. Med. 209, 2441–2453 (2012).
Wang, R. & Green, D. R. Metabolic checkpoints in activated T cells. Nat. Immunol. 13, 907–915 (2012).
Sengupta, S., Peterson, T. R., Laplante, M., Oh, S. & Sabatini, D. M. mTORC1 controls fasting-induced ketogenesis and its modulation by ageing. Nature 468, 1100–1104 (2010).
Lefebvre, P., Chinetti, G., Fruchart, J. C. & Staels, B. Sorting out the roles of PPAR alpha in energy metabolism and vascular homeostasis. J. Clin. Invest. 116, 571–580 (2006).
Barber, D. F., Faure, M. & Long, E. O. LFA-1 contributes an early signal for NK cell cytotoxicity. J. Immunol. 173, 3653–3659 (2004).
Mace, E. M., Monkley, S. J., Critchley, D. R. & Takei, F. A dual role for talin in NK cell cytotoxicity: activation of LFA-1-mediated cell adhesion and polarization of NK cells. J. Immunol. 182, 948–956 (2009).
James, A. M. et al. Rapid activation receptor- or IL-2-induced lytic granule convergence in human natural killer cells requires Src, but not downstream signaling. Blood 121, 2627–2637 (2013).
Mentlik, A. N., Sanborn, K. B., Holzbaur, E. L. & Orange, J. S. Rapid lytic granule convergence to the MTOC in natural killer cells is dependent on dynein but not cytolytic commitment. Mol. Biol. Cell 21, 2241–2256 (2010).
Liu, D., Martina, J. A., Wu, X. S., Hammer, J. A. 3rd & Long, E. O. Two modes of lytic granule fusion during degranulation by natural killer cells. Immunol. Cell Biol. 89, 728–738 (2011).
Menager, M. M. et al. Secretory cytotoxic granule maturation and exocytosis require the effector protein hMunc13-4. Nat. Immunol. 8, 257–267 (2007).
Tuli, A. et al. Arf-like GTPase Arl8b regulates lytic granule polarization and natural killer cell-mediated cytotoxicity. Mol. Biol. Cell 24, 3721–3735 (2013).
Kupfer, A., Dennert, G. & Singer, S. J. Polarization of the Golgi apparatus and the microtubule-organizing center within cloned natural killer cells bound to their targets. Proc. Natl Acad. Sci. USA 80, 7224–7228 (1983).
Boden, G. & Shulman, G. I. Free fatty acids in obesity and type 2 diabetes: defining their role in the development of insulin resistance and beta-cell dysfunction. Eur. J. Clin. Invest. 32, 14–23 (2002).
Boulenouar, S. et al. Adipose type one innate lymphoid cells regulate macrophage homeostasis through targeted cytotoxicity. Immunity 46, 273–286 (2017).
Wensveen, F. M. et al. NK cells link obesity-induced adipose stress to inflammation and insulin resistance. Nat. Immunol. 16, 376–385 (2015).
O’Sullivan, T. E. et al. Adipose-resident group 1 innate lymphoid cells promote obesity-associated insulin resistance. Immunity 45, 428–441 (2016).
Lee, B. C. et al. Adipose natural killer cells regulate adipose tissue macrophages to promote insulin resistance in obesity. Cell Metab. 23, 685–698 (2016).
Muller, A. J. et al. Chronic inflammation that facilitates tumor progression creates local immune suppression by inducing indoleamine 2,3 dioxygenase. Proc. Natl Acad. Sci. USA 105, 17073–17078 (2008).
Allison, D. J. & Ditor, D. S. Immune dysfunction and chronic inflammation following spinal cord injury. Spinal Cord 53, 14–18 (2015).
Gentile, L. F. et al. Persistent inflammation and immunosuppression: a common syndrome and new horizon for surgical intensive care. J. Trauma Acute Care Surg. 72, 1491–1501 (2012).
Cheng, S. C. et al. Broad defects in the energy metabolism of leukocytes underlie immunoparalysis in sepsis. Nat. Immunol. 17, 406–413 (2016).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–945 (2015).
Cohen, N. R. et al. Shared and distinct transcriptional programs underlie the hybrid nature of iNKT cells. Nat. Immunol. 14, 90–99 (2013).
Acknowledgements
We thank M. Wilk and J. Barrett for assistance with experiments. This research was supported by National Institutes of Health (NIH) grant R01 AI11304603 (M.B.B.), European Research Council (ERC) Starting Grant 679173, a Cancer Research Institute CLIP grant and 16/FRL/3865 (L.L.).
Author information
Authors and Affiliations
Contributions
X.M., L.D., and L.L. conceived and designed the experiments, and wrote the manuscript. X.M., L.D., A.H., R.M.L., D.D., K.W., R.D., M.R., and L.L. performed the experiments. C.F. performed the RNA-seq analysis. A.T., A.V., W.P., D.O.’S., and B.S.N. obtained patient samples and coordinated the clinical investigations. S.B., C.O.F., K.H.G.M., M.B.B., and D.F. provided advice, reagents and critical insight.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6
Rights and permissions
About this article
Cite this article
Michelet, X., Dyck, L., Hogan, A. et al. Metabolic reprogramming of natural killer cells in obesity limits antitumor responses. Nat Immunol 19, 1330–1340 (2018). https://doi.org/10.1038/s41590-018-0251-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-018-0251-7
This article is cited by
-
Cancer immunometabolism: advent, challenges, and perspective
Molecular Cancer (2024)
-
Single-cell transcriptome analysis reveals immunosuppressive landscape in overweight and obese colorectal cancer
Journal of Translational Medicine (2024)
-
Targeting natural killer cells: from basic biology to clinical application in hematologic malignancies
Experimental Hematology & Oncology (2024)
-
Advances in metabolic reprogramming of NK cells in the tumor microenvironment on the impact of NK therapy
Journal of Translational Medicine (2024)
-
Real-time ex vivo monitoring of NK cell migration toward obesity-associated oesophageal adenocarcinoma following modulation of CX3CR1
Scientific Reports (2024)