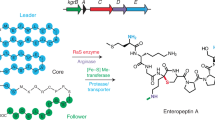
Abstract
Microbial natural products comprise diverse architectures that are generated by equally diverse biosynthetic strategies. In peptide natural products, amino acid sidechains are frequently used as sites of modification to generate macrocyclic motifs. Backbone amide groups, among the most stable of biological moieties, are rarely used for this purpose. Here we report the discovery and biosynthesis of bicyclostreptins—peptide natural products from Streptococcus spp. with an unprecedented structural motif consisting of a macrocyclic β-ether and a heterocyclic sp3–sp3 linkage between a backbone amide nitrogen and an adjacent α-carbon. Both reactions are installed, in that order, by two radical S-adenosylmethionine (RaS) metalloenzymes. Bicyclostreptins are produced at nM concentrations and are potent growth regulation agents in Streptococcus thermophilus. Our results add a distinct and unusual chemotype to the growing family of ribosomal peptide natural products, expand the already impressive catalytic scope of RaS enzymes, and provide avenues for further biological studies in human-associated streptococci.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Plasmids and strains used in this study are described in Supplementary Tables 1 and 2. All oligonucleotides are shown in Supplementary Table 3. The sequences of codon-optimized gene fragments are provided in Supplementary Note 1. Other relevant data supporting the findings of this study are available within the paper and the supplementary material. Raw NMR data used to elucidate natural product structures are available from the corresponding author upon reasonable request.
References
Bentley, R. Microbial secondary metabolites play important roles in medicine; prospects for discovery of new drugs. Perspect. Biol. Med. 40, 364–394 (1997).
Clardy, J. & Walsh, C. Lessons from natural molecules. Nature 432, 829–837 (2004).
Demain, A. L. & Sanchez, S. Microbial drug discovery: 80 years of progress. J. Antibiot. 62, 5–16 (2009).
Newman, D. J. & Cragg, G. M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 83, 770–803 (2020).
Miller, S. J. & Clardy, J. Natural products: beyond grind and find. Nat. Chem. 1, 261–263 (2009).
Ziemert, N., Alanjary, M. & Weber, T. The evolution of genome mining in microbes—a review. Nat. Prod. Rep. 33, 988–1005 (2016).
Katz, L. & Baltz, R. H. Natural product discovery: past, present, and future. J. Ind. Microbiol. Biotechnol. 43, 155–176 (2016).
Ting, C. P. et al. Use of a scaffold peptide in the biosynthesis of amino acid-derived natural products. Science 365, 280–284 (2019).
Velásquez, J. E. & van der Donk, W. A. Genome mining for ribosomally synthesized natural products. Curr. Opin. Chem. Biol. 15, 11–21 (2011).
Russell, A. H. & Truman, A. W. Genome mining strategies for ribosomally synthesised and post-translationally modified peptides. Comput. Struct. Biotechnol. J. 18, 1838–1851 (2020).
Montalban-Lopez, M. et al. New developments in RiPP discovery, enzymology and engineering. Nat. Prod. Rev. 38, 130–239 (2021).
Bushin, L. B., Clark, K. A., Pelczer, I. & Seyedsayamdost, M. R. Charting an unexplored streptococcal biosynthetic landscape reveals a unique peptide cyclization motif. J. Am. Chem. Soc. 140, 17674–17684 (2018).
Fleuchot, B. et al. Rgg proteins associated with internalized small hydrophobic peptides: a new quorum-sensing mechanism in streptococci. Mol. Microbiol. 80, 1102–1119 (2011).
Frey, P. A., Hegeman, A. D. & Ruzicka, F. J. The radical SAM superfamily. Crit. Rev. Biochem. Mol. Biol. 43, 63–88 (2008).
Landgraf, B. J., McCarthy, E. L. & Booker, S. J. Radical S-adenosylmethionine enzymes in human health and disease. Annu. Rev. Biochem. 85, 485–514 (2016).
Broderick, J. B., Duffus, B. R., Duschene, K. S. & Shepard, E. M. Radical S-adenosylmethionine enzymes. Chem. Rev. 114, 4229–4317 (2014).
Bassler, B. L. & Losick, R. Bacterially speaking. Cell 125, 237–246 (2006).
Gerlt, J. A. Genomic enzymology: web tools for leveraging protein family sequence–function space and genome context to discover novel functions. Biochemistry 56, 4293–4308 (2017).
Caruso, A., Bushin, L. B., Clark, K. A., Martinie, R. J. & Seyedsayamdost, M. R. Radical approach to enzymatic β-thioether bond formation. J. Am. Chem. Soc. 141, 990–997 (2019).
Clark, K. A., Bushin, L. B. & Seyedsayamdost, M. R. Aliphatic ether bond formation expands the scope of radical SAM enzymes in natural product biosynthesis. J. Am. Chem. Soc. 141, 10610–10615 (2019).
Caruso, A., Martinie, R. J., Bushin, L. B. & Seyedsayamdost, M. R. Macrocyclization via an arginine-tyrosine crosslink broadens the reaction scope of radical S-adenosylmethionine enzymes. J. Am. Chem. Soc. 141, 16610–16614 (2019).
Bushin, L. B., Covington, B. C., Rued, B. E., Federle, M. J. & Seyedsayamdost, M. R. Discovery and biosynthesis of streptosactin, a sactipeptide with an alternative topology encoded by commensal bacteria in the human microbiome. J. Am. Chem. Soc. 142, 16265–16275 (2020).
Rued, B. E. et al. Quorum sensing in Streptococcus mutans regulates production of tryglysin, a novel RaS-RiPP antimicrobial compound. mBio 12, e02688–20 (2021).
Schramma, K. R., Bushin, L. B. & Seyedsayamdost, M. R. Structure and biosynthesis of a macrocyclic peptide containing an unprecedented lysine-to-tryptophan crosslink. Nat. Chem. 7, 431–437 (2015).
Imai, Y. et al. A new antibiotic selectivey kills Gram-negative pathogens. Nature 576, 459–464 (2019).
Freeman, M. F. et al. Metagenome mining reveals polytheonamides as posttranslationally modified ribosomal peptides. Science 338, 387–390 (2012).
Nguyen, T. Q. N. et al. Post-translational formation of strained cyclophanes in bacteria. Nat. Chem. 12, 1042–1053 (2020).
Burkhart, B. J., Hudson, G. A., Dunbar, K. L. & Mitchell, D. A. A prevalent peptide-binding domain guides ribosomal natural product biosynthesis. Nat. Chem. Biol. 11, 564–570 (2015).
Delorme, C. et al. Complete genome sequence of the pigmented Streptococcus thermophilus strain JIM8232. J. Bacteriol. 193, 5581–5582 (2011).
Sørensen, U. B. S., Wiegan, I. C., Boes, J. & Farre, M. The distribution of clones of Streptococcus agalactiae (group B streptococci) among herdspersons and dairy cows demonstrates lack of host specificity for some lineages. Vet. Microbiol. 235, 71–79 (2019).
Fontaine, L. et al. Novel pheromone quorum-sensing system controls the development of natural competence in Streptococcus thermophilus and Streptococcus salivarius. J. Bacteriol. 192, 1444–1454 (2010).
Claverys, J.-P. & Håvarstein, L. S. Cannibalism and fratricide: mechanisms and raisons dʼêtre. Nat. Rev. Microbiol. 5, 219–229 (2007).
Claverys, J.-P., Martin, B. & Håvarstein, L. S. Competence-induced fratricide in streptococci. Mol. Microbiol. 64, 1423–1433 (2007).
Prozorov, A. A. & Danilenko, V. N. Allolysis in bacteria. Microbiology 80, 1–9 (2011).
Guiral, S., Mitchell, T. J., Martin, B. & Claverys, J.-P. Competence-programmed predation of noncompetent cells in the human pathogen Streptococcus pneumoniae: genetic requirements. Proc. Natl Acad. Sci. USA 102, 8710–8715 (2005).
Lewis, K. Persister cells, dormancy, and infectious disease. Nat. Rev. Microbiol. 5, 48–56 (2007).
Rittershaus, E. S. C., Baek, S.-H. & Sassetti, C. M. The normalcy of dormancy: common themes in microbial quiescence. Cell Host Microbe 13, 643–651 (2013).
Hayes, C. S. & Low, D. A. Signals of growth regulation in bacteria. Curr. Opin. Microbiol. 12, 667–673 (2009).
Flühe, L. et al. The radical SAM enzyme AlbA catalyzes thioether bond formation in subtilosin A. Nat. Chem. Biol. 8, 350–357 (2012).
Huo, L., Rachid, S., Stadler, M., Wenzel, S. C. & Müller, R. Synthetic biotechnology to study and engineer ribosomal bottromycin biosynthesis. Chem. Biol. 19, 1278–1287 (2012).
Barr, I. et al. Demonstration that the radical S-adenosylmethionine (SAM) enzyme PqqE catalyzes de novo carbon-carbon cross-linking within a peptide substrate PqqA in the presence of the peptide chaperone PqqD. J. Biol. Chem. 291, 8877–8884 (2016).
Khaliullin, B., Ayikpoe, R., Tuttle, M. & Latham, J. A. Mechanistic elucidation of the mycofactocin-biosynthetic radical S-adenosylmethionine protein, MftC. J. Biol. Chem. 292, 13022–13033 (2017).
Benjdia, A., Guillot, A., Ruffié, P., Leprince, J. & Berteau, O. Post-translational modification of ribosomally synthesized peptides by a radical SAM epimerase in Bacillus subtilis. Nat. Chem. 9, 698–707 (2017).
Parent, A. et al. The B12-radical SAM enzyme PoyC catalyzes valine Cβ-methylation during polytheonamide biosynthesis. J. Am. Chem. Soc. 138, 15515–15518 (2016).
Grell, T. A. J. et al. Structural and spectroscopic analyses of the sporulation killing factor biosynthetic enzyme SkfB, a bacterial adomet radical sactisynthase. J. Biol. Chem. 293, 17349–17361 (2018).
Morinaka, B. I. et al. Natural noncanonical protein splicing yields products with diverse β-amino acid residues. Science 359, 779–782 (2018).
Ayikpoe, R. S. & Latham, J. A. MftD catalyzes the formation of a biologically active redox center in the biosynthesis of the ribosomally synthesized and post-translationally modified redox cofactor mycofactocin. J. Am. Chem. Soc. 141, 13582–13591 (2019).
Hudson, G. A. et al. Bioinformatic mapping of radical S-adenosylmethionine-dependent ribosomally synthesized and post-translationally modified peptides identifies new Cα, Cβ, and Cγ-linked thioether-containing peptides. J. Am. Chem. Soc. 141, 8228–8238 (2019).
Flores, A. R. et al. Sequence type 1 group B Streptococcus, an emerging cause of invasive disease in adults, evolves by small genetic changes. Proc. Natl Acad. Sci. USA 112, 6431–6436 (2015).
Bushin, L. B. & Seyedsayamdost, M. R. Guidelines for determining the structures of radical SAM enzyme-catalyzed modifications in the biosynthesis of RiPP natural products. Methods Enzymol. 606, 439–460 (2018).
Wiegand, I., Hilpert, K. & Hancock, R. E. W. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 3, 163–175 (2008).
Acknowledgements
We thank B. A. Johnson for technical assistance with isolation of bicyclostreptin A as well as the Eli Lilly Edward C. Taylor Fellowship in Chemistry (to K.A.C.) and the National Science Foundation (NSF GRFP to L.B.B. and NSF CAREER Award to M.R.S.) for financial support.
Author information
Authors and Affiliations
Contributions
L.B.B. and M.R.S. conceived of the study. L.B.B. carried out in vivo characterization and structural elucidation of the reactions of all enzymes. B.C.C. identified the mature natural products from the native organism. K.A.C. conducted in vitro analysis of the radical SAM enzymes. L.B.B., B.C.C. and A.L. carried out bioactivity assays. L.B.B. and M.R.S. wrote the manuscript, with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 HR-MS/MS analysis of S. agalactiae HghCD and HghBCD reaction products.
(a) All y-ions N-terminal to His34 and b-ions C-terminal to His34 for the HghCD product are -2 Da relative to substrate. The same y-ions from the HghBCD product are -4 Da, while b40 remains 2 Da lighter. This pattern points to modifications at His34 within the ‘HGH’ motif. (b) Trypsinolysis of the HghBD or HghBCD products gives the fragments shown. The C-terminal 8mer captures both -2 Da (HghBD) and -4 Da (HghBCD) products. Cleavage does not occur at the Lys residue in red.
Extended Data Fig. 2 HghC and HghB are competent in reductive cleavage of SAM to generate 5’-dA.
(a) Reaction of HghC or HghB with SAM in the absence of HghA. Shown are HR-MS extracted ion chromatograms for 5’-dA after an overnight reaction (18 h). The cleaved 5ꞌ-dA product is seen with both reductants but not in the absence of enzyme. (b) Detection of 5ꞌ-dA after the reaction of HghC or HghB with substrate and SAM after 18 h. Note, different elution programs were carried out in panels a and b, thus explaining the different retention times for 5’-dA (see Methods).
Extended Data Fig. 3 Time-dependent formation of the HghC product as measured by HPLC-Qtof-MS extracted ion chromatography.
The average of three independent samples are shown; bars represent standard error.
Extended Data Fig. 4 Sequence alignment of HghD from S. agalactiae or S. thermophilus with PqqD and WgkC.
No substantial homology is observed.
Extended Data Fig. 5 Biosynthetic pathway of bicyclostreptins A and B from S. thermophilus JIM 8232.
Blue spheres represent unmodified amino acids in the core region with the indicated one-letter code. Gray spheres are amino acids in the leader sequence.
Extended Data Fig. 6 Summary of growth inhibition assays of bicyclostreptins A and C and the variants shown against select bacteria.
Shown are MICs in μM (black and red) or IC50 values in μM (blue), which were calculated using the averages of two independent biological samples.
Extended Data Fig. 7 Effect of bicyclostreptin C on the growth of S. thermophilus JIM 8232.
The averages of two biologically independent samples are shown.
Supplementary information
Supplementary Information
Supplementary Tables 1–21, Supplementary Figs. 1–7 and Supplementary Note 1.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bushin, L.B., Covington, B.C., Clark, K.A. et al. Bicyclostreptins are radical SAM enzyme-modified peptides with unique cyclization motifs. Nat Chem Biol 18, 1135–1143 (2022). https://doi.org/10.1038/s41589-022-01090-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-022-01090-8