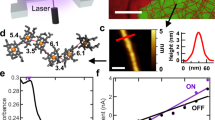
Abstract
Multifunctional living materials are attractive due to their powerful ability to self-repair and replicate. However, most natural materials lack electronic functionality. Here we show that an electric field, applied to electricity-producing Geobacter sulfurreducens biofilms, stimulates production of cytochrome OmcZ nanowires with 1,000-fold higher conductivity (30 S cm−1) and threefold higher stiffness (1.5 GPa) than the cytochrome OmcS nanowires that are important in natural environments. Using chemical imaging-based multimodal nanospectroscopy, we correlate protein structure with function and observe pH-induced conformational switching to β-sheets in individual nanowires, which increases their stiffness and conductivity by 100-fold due to enhanced π-stacking of heme groups; this was further confirmed by computational modeling and bulk spectroscopic studies. These nanowires can transduce mechanical and chemical stimuli into electrical signals to perform sensing, synthesis and energy production. These findings of biologically produced, highly conductive protein nanowires may help to guide the development of seamless, bidirectional interfaces between biological and electronic systems.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are contained in the published article (and its supplementary information), or available from the corresponding author on reasonable request. Source data are provided with this paper.
Code availability
The codes used during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Someya, T., Bao, Z. & Malliaras, G. G. The rise of plastic bioelectronics. Nature 540, 379–385 (2016).
Bathe, M. et al. Roadmap on biological pathways for electronic nanofabrication and materials. Nano Futures 3, 012001 (2019).
Zhang, B., Song, W., Brown, J., Nemanich, R. J. & Lindsay, S. Electronic conductance resonance in non-redox proteins. J. Am. Chem. Soc. 142, 6432–6438 (2020).
Wang, F. et al. Structure of microbial nanowires reveals stacked hemes that transport electrons over micrometers. Cell 177, 361–369 (2019).
Reguera, G. et al. Extracellular electron transfer via microbial nanowires. Nature 435, 1098–1101 (2005).
Mehta, T., Coppi, M. V., Childers, S. E. & Lovley, D. R. Outer membrane c-type cytochromes required for Fe(III) and Mn(IV) oxide reduction in Geobacter sulfurreducens. Appl. Environ. Microbiol. 71, 8634–8641 (2005).
Malvankar, N. S. et al. Tunable metallic-like conductivity in microbial nanowire networks. Nat. Nanotechnol. 6, 573–579 (2011).
Inoue, K. et al. Purification and characterization of omcZ, an outer-surface, octaheme c-type cytochrome essential for optimal current production by Geobacter sulfurreducens. Appl. Environ. Microbiol. 76, 3999–4007 (2010).
Nevin, K. P. et al. Anode biofilm transcriptomics reveals outer surface components essential for high density current production in Geobacter sulfurreducens fuel cells. PLoS ONE 4, e5628 (2009).
Inoue, K. et al. Specific localization of the c-type cytochrome OmcZ at the anode surface in current-producing biofilms of Geobacter sulfurreducens. Environ. Microbiol. Rep. 3, 211–217 (2010).
Malvankar, N. S., Tuominen, M. T. & Lovley, D. R. Lack of cytochrome involvement in long-range electron transport through conductive biofilms and nanowires of Geobacter sulfurreducens. Energy Environ. Sci. 5, 8651–8659 (2012).
Amenabar, I. et al. Structural analysis and mapping of individual protein complexes by infrared nanospectroscopy. Nat. Commun. 4, 2890 (2013).
O’Brien, J. P. & Malvankar, N. S. A simple and low-cost procedure for growing Geobacter sulfurreducens cell cultures and biofilms in bioelectrochemical systems. Curr. Protoc. Microbiol. 43, A.4K.1–A.4K.27 (2017).
Tan, Y. et al. Synthetic biological protein nanowires with high conductivity. Small 12, 4481–4485 (2016).
Richter, L. V., Sandler, S. J. & Weis, R. M. Two isoforms of Geobacter sulfurreducens PilA have distinct roles in pilus biogenesis, cytochrome localization, extracellular electron transfer, and biofilm formation. J. Bacteriol. 194, 2551–2563 (2012).
Liu, X., Zhuo, S., Rensing, C. & Zhou, S. Syntrophic growth with direct interspecies electron transfer between pili-free Geobacter species. ISME J. 12, 2142–2151 (2018).
Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta 1767, 1073–1101 (2007).
Yalcin, S. E., Legg, B. A., Yeşilbaş, M., Malvankar, N. S. & Boily, J. Direct observation of anisotropic growth of water films on minerals driven by defects and surface tension. Sci. Adv. 6, eaaz9708 (2020).
Berweger, S. et al. Nano-chemical infrared imaging of membrane proteins in lipid bilayers. J. Am. Chem. Soc. 135, 18292–18295 (2013).
Yang, H., Yang, S., Kong, J., Dong, A. & Yu, S. Obtaining information about protein secondary structures in aqueous solution using Fourier transform IR spectroscopy. Nat. Protoc. 10, 382–396 (2015).
Qian, X. et al. Biochemical characterization of purified OmcS, a c-type cytochrome required for insoluble Fe(III) reduction in Geobacter sulfurreducens. Biochim. Biophys. Acta 1807, 404–412 (2011).
Chadwick, G. L., Otero, F. J., Gralnick, J. A., Bond, D. R. & Orphan, V. J. NanoSIMS imaging reveals metabolic stratification within current-producing biofilms. Proc. Natl Acad. Sci. USA 116, 20716–20724 (2019).
Ho Choi, S., Kim, B. & Frisbie, C. D. Electrical resistance of long conjugated molecular wires. Science 320, 1482–1486 (2008).
Lee, K. et al. Metallic transport in polyaniline. Nature 441, 65–68 (2006).
Valasatava, Y., Andreini, C. & Rosato, A. Hidden relationships between metalloproteins unveiled by structural comparison of their metal sites. Sci. Rep. 5, 9486 (2015).
Janiak, C. A critical account on π-π stacking in metal complexes with aromatic nitrogen-containing ligands. J. Chem. Soc., Dalton Trans. 2000, 3885–3896 (2000).
Adhikari, R., Malvankar, N., Tuominen, M. & Lovley, D. Conductivity of individual Geobacter pili. RSC Adv. 6, 8354–8357 (2016).
Malvankar, N. S. et al. Structural basis for metallic-like conductivity in microbial nanowires. mBio 6, e00084–00015 (2015).
Donhauser, Z. J. et al. Conductance switching in single molecules through conformational changes. Science 292, 2303–2307 (2001).
Su, T. A., Li, H., Steigerwald, M. L., Venkataraman, L. & Nuckolls, C. Stereoelectronic switching in single-molecule junctions. Nat. Chem. 7, 215–220 (2015).
Li, C., Wang, Z., Lu, Y., Liu, X. & Wang, L. Conformation-based signal transfer and processing at the single-molecule level. Nat. Nanotechnol. 12, 1071 (2017).
Simone, R. F. et al. Influence of the β‐sheet content on the mechanical properties of aggregates during amyloid fibrillization. Angew. Chem. Int. Ed. 54, 2462–2466 (2015).
Freund, J., Halbritter, J. & Hörber, J. How dry are dried samples? Water adsorption measured by STM. Microsc. Res. Tech. 44, 327–338 (1999).
Rygula, A. et al. Raman spectroscopy of proteins: a review. J. Raman Spectrosc. 44, 1061–1076 (2013).
Sreerama, N. & Woody, R. W. Estimation of protein secondary structure from circular dichroism spectra: comparison of CONTIN, SELCON, and CDSSTR methods with an expanded reference set. Anal. Biochem. 287, 252–260 (2000).
Greenfield, N. J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc. 1, 2876–2890 (2006).
Guterman, T. et al. Formation of bacterial pilus-like nanofibres by designed minimalistic self-assembling peptides. Nat. Commun. 7, 13482 (2016).
Boyken, S. E. et al. De novo design of tunable, pH-driven conformational changes. Science 364, 658–664 (2019).
Koga, T., Taguchi, K., Kobuke, Y., Kinoshita, T. & Higuchi, M. Structural regulation of a peptide‐conjugated graft copolymer: a simple model for amyloid formation. Chem.–A Eur. J. 9, 1146–1156 (2003).
Cerpa, R., Cohen, F. E. & Kuntz, I. D. Conformational switching in designed peptides: the helix/sheet transition. Fold. Des. 1, 91–101 (1996).
Cote, Y. et al. Mechanism of the pH-controlled self-assembly of nanofibers from peptide amphiphiles. J. Phys. Chem. C. 118, 16272–16278 (2014).
Haldar, S., Sil, P., Thangamuniyandi, M. & Chattopadhyay, K. Conversion of amyloid fibrils of cytochrome c to mature nanorods through a honeycomb morphology. Langmuir 31, 4213–4223 (2015).
Wang, X., Ye, X. & Zhang, G. Investigation of pH-induced conformational change and hydration of poly (methacrylic acid) by analytical ultracentrifugation. Soft Matter 11, 5381–5388 (2015).
Esquerra, R. M. et al. The pH dependence of heme pocket hydration and ligand rebinding kinetics in photodissociated carbonmonoxymyoglobin. J. Biol. Chem. 283, 14165–14175 (2008).
Durchschlag, H. & Zipper, P. Modeling the hydration of proteins at different pH values. Anal. Ultracentrif. VII 127, 98–112 (2004).
Tschirhart, T. et al. Electronic control of gene expression and cell behaviour in Escherichia coli through redox signalling. Nat. Commun. 8, 14030 (2017).
Park, I. & Kim, B.-C. Homologous overexpression of omcZ, a gene for an outer surface c-type cytochrome of Geobacter sulfurreducens by single-step gene replacement. Biotechnol. Lett. 33, 2043 (2011).
Vargas, M. et al. Aromatic amino acids required for pili conductivity and long-range extracellular electron transport in Geobacter sulfurreducens. mBio 4, e00105–e00113 (2013).
Wang, F. et al. Cryoelectron microscopy reconstructions of the Pseudomonas aeruginosa and Neisseria gonorrhoeae type IV pili at sub-nanometer resolution. Structure 25, 1423–1435 (2017).
Silva, J. C., Gorenstein, M. V., Li, G.-Z., Vissers, J. P. & Geromanos, S. J. Absolute quantification of proteins by LCMSE: a virtue of parallel MS acquisition. Mol. Cell. Proteom. 5, 144–156 (2006).
Grossmann, J. et al. Implementation and evaluation of relative and absolute quantification in shotgun proteomics with label-free methods. J. Proteom. 73, 1740–1746 (2010).
Krey, J. F. et al. Accurate label-free protein quantitation with high-and low-resolution mass spectrometers. J. Proteome Res. 13, 1034–1044 (2013).
Erickson, H. P. Size and shape of protein molecules at the nanometer level determined by sedimentation, gel filtration, and electron microscopy. Biol. Proced. 11, 32 (2009).
Qin, N. et al. Nanoscale probing of electron-regulated structural transitions in silk proteins by near-field IR imaging and nano-spectroscopy. Nat. Commun. 7, 1–8 (2016).
Paulite, M. et al. Imaging secondary structure of individual amyloid fibrils of a β2-microglobulin fragment using near-field infrared spectroscopy. J. Am. Chem. Soc. 133, 7376–7383 (2011).
Ramer, G., Ruggeri, F. S., Levin, A., Knowles, T. P. & Centrone, A. Determination of polypeptide conformation with nanoscale resolution in water. ACS Nano 12, 6612–6619 (2018).
Ruggeri, F. et al. Infrared nanospectroscopy characterization of oligomeric and fibrillar aggregates during amyloid formation. Nat. Commun. 6, 1–9 (2015).
Ruggeri, F. et al. Nanoscale studies link amyloid maturity with polyglutamine diseases onset. Sci. Rep. 6, 1–11 (2016).
Kocun, M., Labuda, A., Meinhold, W., Revenko, I. & Proksch, R. Fast, high resolution, and wide modulus range nanomechanical mapping with bimodal tapping mode. ACS Nano 11, 10097–10105 (2017).
Bruckner, S. Estimation of the background in powder diffraction patterns through a robust smoothing procedure. J. Appl. Crystallogr. 33, 977–979 (2000).
Acknowledgements
We thank D. Lovley (University of Massachusetts, Amherst) and K. Inoue (University of Miyazaki) for providing strains and OmcZ antibody as well as E. Yan, E. Martz, F. Samatey, C. Salgueiro, C. Shipps and Y. Xiong for helpful discussions. We also thank C. Leang for providing the protocol for immunogold labeling, T. Gokus from Neaspec for help with nanoscale IR imaging and M. Shahid Mansuri, J. Kanyo and T. Lam for help with mass-spectrometry analysis. A portion of the research was performed using Environmental Molecular Sciences Laboratory (EMSL, Ringgold ID 130367), a Department of Energy (DOE) Office of Science User Facility sponsored by the Office of Biological and Environmental Research. S.E.Y. thanks M. Raschke, J. Atkin and S. Lea for help with building the IR s-SNOM setup and C. Smallwood for help with bacteriorhodopsin sample preparation. We thank T. Walsh from Asylum Research for help with stiffness measurements. At Yale, we thank W. Gray from C. Jacobs-Wagner’s laboratory for help with fluorescence microscopy and Z. Wu from H. Wang’s laboratory for help with Raman studies. Computational work was supported by the Air Force Office of Scientific Research Grant FA9550-17-0198 (V.S.B.) and high-performance computing time from the National Energy Research Scientific Computing Center and from the high-performance computing facilities at Yale as well as supercomputer time from the Extreme Science and Engineering Discovery Environment under grant no. TG-CHE170024 (A.A.). Anton 2 computer time was provided by the Pittsburgh Supercomputing Center (PSC) through Grant R01GM116961 from the National Institutes of Health (NIH). The Anton 2 machine at PSC was generously made available by D.E. Shaw Research. This research was supported by the Career Award at the Scientific Interfaces from Burroughs Welcome Fund (N.S.M.), the NIH Director’s New Innovator award no. 1DP2AI138259-01 (N.S.M.), the National Science Foundation (NSF) CAREER award no. 1749662 (N.S.M.). Research was sponsored by the Defense Advanced Research Project Agency Army Research Office and was accomplished under Cooperative Agreement Number W911NF-18-2-0100 (N.S.M. and V.S.B). This research was supported by NSF Graduate Research Fellowship awards 2017224445 (J.P.O.). Research in the laboratory is also supported by the Charles H. Hood Foundation Child Health Research Award (N.S.M.) and The Hartwell Foundation Individual Biomedical Research Award (N.S.M.).
Author information
Authors and Affiliations
Contributions
S.E.Y. and N.S.M. conceived and designed the study. S.E.Y. built the quantum cascade laser-coupled IR s-SNOM detection interferometer, prepared samples, performed AFM, IR s-SNOM measurements, imaged immunogold-labeled nanowires with AFM along with imaging and analysis of reduction in nanowire diameter. J.P.O. grew biofilms in microbial fuel cell and analyzed protein content with R.J. K.R. built the OmcZ model and performed simulations, with help from P.J.D., under the guidance of V.S.B. J.P.O. and W.H. purified nanowires from bacteria, performed CD experiments and conducted analysis. J.P.O. performed FTIR and fluorescence emission spectroscopy and analyzed data with S.E.Y. W.H. and S.E.Y. performed principal component analysis on the IR s-SNOM data. V.S. and Y.G. imaged immunogold-labeled nanowires with TEM. Y.G. also carried out CP–AFM measurements and analyzed data with P.J.D. Y.G. and S.E.Y. performed and analyzed nanowire stiffness measurements. S.M.Y. carried out mass spectroscopy as well as Raman spectroscopy and analyzed Raman data with S.E.Y. Electrode fabrication using electron-beam lithography was carried out by D.V. and Y.G. S.E.Y. and T.V. performed XRD measurements and analyzed data with Y.G. A.A. and S.C. performed initial molecular dynamics simulations under the guidance of V.S.B. A.A. constructed the models, coded the analysis scripts and performed the analysis of molecular dynamics data. N.S.M. supervised the project. S.E.Y. and N.S.M. cowrote the manuscript with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Multimodal imaging and nanospectoscopy platform to determine the structure of individual OmcZ and OmcS nanowires as well as their electrical and mechanical properties.
a, Multimodal platform with OmcS nanowire structure showing stacked hemes providing electron transport path (PDB ID: 6EF8). b, c, OmcZ nanowires produced by ΔomcS strain grown under conditions that overexpress OmcZ. b, AFM images of OmcZ nanowires of ΔomcS strain. c, Zoomed image of OmcZ nanowire shown in white square in b. d, AFM Height profile of the OmcZ nanowire taken at the a location shown by a red line in c. Scale bars: b, 100 nm c, 20 nm.
Extended Data Fig. 2 Mass spectrometry and immunoblotting of filament preparations confirm electric field induces overexpression of OmcZ in biofilms.
a, b, Strategy to evaluate the effect of an electric field on the production of OmcZ nanowires. Wild-type G. sulfurreducens cells were grown with a continuous supply of fumarate on graphite electrodes as anodes in a microbial fuel cell. a, An electric field was supplied during the growth of current-producing biofilms by connecting anode to cathode via a potentiostat. b, The electric-field was absent after disconnecting the anodes from the cathodes. OmcZ peptide coverage (blue) in filament preparations of c, Wild-type (WT) and d, W51W57 strain confirming the presence of an extracellular (30 kDa) form of OmcZ that forms nanowires. Comparison of OmcZ abundance in filament preparations of WT and W51W57 strains using e, Immunoblotting and f, Mass spectrometry showing higher level of OmcZ in W51W57 strain than WT. Data represent mean ± standard deviation (n = 3 biologically independent samples overlaid as black circles).
Extended Data Fig. 3 Immunogold labeling confirms the identity of OmcZ nanowires.
a, AFM image of immunogold-labelled OmcZ nanowire. b, heights of nanowire (red) and gold nanoparticle (blue) at locations shown in a. c–f, TEM images of OmcZ nanowires of ZKI strain in the c, absence of OmcZ antibody and d, in the presence of anti-OmcZ antibody. Secondary antibody with gold nanoparticles was used in both c–e, No OmcZ labelling was found for filaments of ΔomcZ strain f, Labelling for OmcZ nanowires of W51W57 strain. Scale bars, a, 100 nm, c, 50 nm, d, 25 nm, e, 100 nm, f, 25 nm.
Extended Data Fig. 4 Low pH reduces the diameter of individual OmcS and OmcZ nanowires.
Histograms of AFM measurements of OmcS and OmcZ nanowire heights showed that lowering the pH reduced the diameter of a, OmcS and b, OmcZ nanowires. Values represent mean ± standard deviation (s.d.) (n = 100 measurements of nanowires over 3 biologically independent samples).
Extended Data Fig. 5 IR s-SNOM imaging of bacteriorhodopsin (bR) confirms α-helical structure.
a, b Schematic of IR s-SNOM. a, The interferometer is comprised of a tunable quantum cascade laser (QCL) for tip illumination, a beam splitter (BS), a detector (Mercury Cadmium Telluride, MCT), a parabolic mirror (PM), and a reference mirror. b, Schematic of the IR s-SNOM setup used for bR imaging (PDB ID: 1m0l). All helices are parallel to electric field lines that enhance the amide I signal. c, AFM topography and corresponding height profile for bR taken at a location shown by a black line. d, IR s-SNOM near-field phase (absorption) images for bR at various IR excitations. At 1660 cm-1 (iii, on-resonant IR), the amide I absorption is enhanced in the near-field phase data. However, when changed to other frequencies (i-ii & iv-v, off-resonance), the phase signal decreases and drops to zero. e, Spatiospectral analysis of near-field phase data for the amide I mode of bacteriorhodopsin. The blue line corresponds to a fit of the imaginary part of a Lorentzian (Lor) with peak at 1663 cm−1 and line width of 25 cm−1. Data represent mean ± standard deviation for individual bR proteins (n = 3 biologically independent samples).
Extended Data Fig. 6 Bulk FTIR and IR s-SNOM confirm lysozyme structure and IR s-SNOM spectroscopy of OmcS nanowires agrees with Cryo-EM structure.
Multi-peak fitting function was used to fit the data using a, Gaussian profile for bulk FTIR and b, a Lorentzian profile for IR s-SNOM data. α-helix corresponds to 1662 cm−1, β-sheet corresponds to 1618 cm−1 and 1678 cm−1, and the loop (D) region corresponds to 1635 cm−1. c, Schematic of the IR s-SNOM setup for nanowire imaging. Secondary structure of the OmcS nanowire at pH 10.5 is shown in a (PDB ID: 6EF8) with α-helices in red, 310 helices in pink and beta strands in green. d, At pH 10.5 used to solve the structure of OmcS nanowires, spatiospectral analysis of near-field phase data of the amide I mode of OmcS nanowire. The blue line corresponds to a fit of imaginary parts of a Lorentzian, with peak positions at 1669 cm−1 and 1643 cm−1 corresponding to α-helical and loop regions respectively. Data represent mean ± standard deviation for individual OmcS nanowires (n = 3 biologically independent samples).
Extended Data Fig. 7 Model of OmcZ structure reveals highly stacked hemes and beta sheets in agreement with experiments.
a, Computational model of OmcZ. b, Superposition of hemes from five 8-heme cytochromes (PDB ID:4QO5:Green, 1FGJ:Blue, 3GM6:Cyan, 6H5L:Gray, and 6HIF:Pink). Both c, computed and d, experimental cryo-EM density for OmcZ show similar shape and size. e-h, Consecutive histidines cause tight heme T-junction. The His-His binding motif found in OmcZ also exists in c3 cytochromes, (PDB ID: e, 1gyo, f, 2bq4, and g, 2e84). In OmcZ model (h), the three pairs of adjacent histidines that bind to T-stacked hemes are all distal histidine ligands bound opposite to the proximal histidine in the CXXCH sequence. In the c3 cytochromes, all the heme-binding adjacent histidine pairs are mixed, containing both a proximal and a distal histidine. Despite this difference, the distance between the heme pairs for c3 and OmcZ is similar (~6.0 Å).
Extended Data Fig. 8 Reduction in nanowire diameter enhances their stiffness.
AFM topography, Young’s modulus, and the stiffness distribution for OmcS nanowires at a, pH 7 and b, pH 2 and for OmcZ nanowires c, pH 7 and d, pH 2. Scale bars: a and d, 100 nm; b and c, 200 nm. Values represent mean ± (s. d.) for n = 512 ×512 measurements of nanowires over three biologically independent samples.
Extended Data Fig. 9 Bulk FTIR spectra of OmcS and OmcZ nanowires show transition to β-sheets at pH 2 and conformation change is independent of buffers.
FTIR spectra at pH 7 and pH 2 for a, OmcS nanowires and b, OmcZ nanowires showing a red shift, consistent with transition to β-sheets. c, Water does not contribute to the amide I spectra because OmcS nanowires at pH 7 under air-dried and D2O conditions display similar spectra. d, e, OmcS nanowires at pH 7 and pH 2 in 10 mM Potassium Phosphate and 20 mM Citrate buffer characterized by d, Solution CD spectra and e, Solid-state CD spectra. f, FTIR spectra for OmcS nanowires in Citrate buffer at pH 2 showing a red shift, consistent with transition to β-sheets. g, Representative AFM image and h, corresponding height profile at a location shown by a red line in g for OmcS nanowires in citrate buffer at pH 2 under air-dried conditions. Scale bar, 200 nm.
Extended Data Fig. 10 Purified OmcS and OmcZ nanowires from ΔomcZ and KN400 strains respectively show conformational change to β-sheets similar to nanowires of wild-type and W51W57 strains.
SDS-PAGE gel of filament preparations showing a single band corresponding to a, OmcS purified from ΔomcZ strain and c, OmcZ from KN400 strain. Corresponding TEM images of b, OmcS and e, OmcZ nanowires. Scale bars, 400 nm. Solution CD spectra of c, OmcS and f, OmcZ is similar to CD spectra of OmcS and OmcZ nanowires purified from wild-type and W51W57 strains respectively (Fig. 6c, d). M1 and M2 represent nanowires sheared from cells by two different methods – vortexing (M1) and blending (M2).
Supplementary information
Supplementary Information
Supplementary Figs. 1–3 and Tables 1 and 2.
Supplementary Data
PDB file for OmcZ model
Source data
Source Data Fig. 1
Uncropped immunoblot images shown in Fig. 1b
Source Data Fig. 2
Statistical Source Data for Fig. 2a,b
Source Data Fig. 3
Statistical Source Data for Fig. 3b–d
Source Data Fig. 4
Statistical Source Data for Fig. 4a,b
Source Data Fig. 5
Statistical Source Data for Fig. 5a–e
Source Data Fig. 6
Statistical Source Data for Fig. 6a–d, f
Source Data Extended Data Fig. 2
Statistical Source Data for Extended Data Fig. 2f
Source Data Extended Data Fig. 4
Statistical Source Data for Extended Data Fig. 4a,b
Source Data Extended Data Fig. 5
Statistical Source Data for Extended Data Fig. 5e
Source Data Extended Data Fig. 6
Statistical Source Data for Extended Data Fig. 6a,b,d
Source Data Extended Data Fig. 8
Statistical Source Data for Extended Data Fig. 8a–d
Source Data Extended Data Fig. 9
Statistical Source Data for Extended Data Fig. 9a–f
Source Data Extended Data Fig. 10
Uncropped gel mages shown in Extended Data Fig. 10a,d
Source Data Extended Data Fig. 10
Statistical Source Data for Extended Data Fig. 10c,f
Rights and permissions
About this article
Cite this article
Yalcin, S.E., O’Brien, J.P., Gu, Y. et al. Electric field stimulates production of highly conductive microbial OmcZ nanowires. Nat Chem Biol 16, 1136–1142 (2020). https://doi.org/10.1038/s41589-020-0623-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-020-0623-9
This article is cited by
-
Widespread extracellular electron transfer pathways for charging microbial cytochrome OmcS nanowires via periplasmic cytochromes PpcABCDE
Nature Communications (2024)
-
Characterization and environmental applications of soil biofilms: a review
Environmental Chemistry Letters (2024)
-
Electroactivity of the magnetotactic bacteria Magnetospirillum magneticum AMB-1 and Magnetospirillum gryphiswaldense MSR-1
Frontiers of Environmental Science & Engineering (2024)
-
Multi-heme cytochrome-mediated extracellular electron transfer by the anaerobic methanotroph ‘Candidatus Methanoperedens nitroreducens’
Nature Communications (2023)
-
Structure of Geobacter cytochrome OmcZ identifies mechanism of nanowire assembly and conductivity
Nature Microbiology (2023)