Abstract
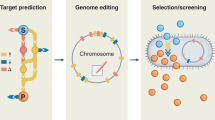
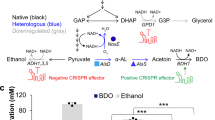
The design and optimization of biosynthetic pathways for industrially relevant, non-model organisms is challenging due to transformation idiosyncrasies, reduced numbers of validated genetic parts and a lack of high-throughput workflows. Here we describe a platform for in vitro prototyping and rapid optimization of biosynthetic enzymes (iPROBE) to accelerate this process. In iPROBE, cell lysates are enriched with biosynthetic enzymes by cell-free protein synthesis and then metabolic pathways are assembled in a mix-and-match fashion to assess pathway performance. We demonstrate iPROBE by screening 54 different cell-free pathways for 3-hydroxybutyrate production and optimizing a six-step butanol pathway across 205 permutations using data-driven design. Observing a strong correlation (r = 0.79) between cell-free and cellular performance, we then scaled up our highest-performing pathway, which improved in vivo 3-HB production in Clostridium by 20-fold to 14.63 ± 0.48 g l−1. We expect iPROBE to accelerate design–build–test cycles for industrial biotechnology.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All cell-free data generated and shown in this manuscript are provided in Supplementary Table 2 and Supplementary Datasets 1 and 2 (.xlsx). Any additional data or unique materials presented in the manuscript may be available from the authors upon reasonable request and through a materials transfer agreement.
References
Nielsen, J. et al. Engineering synergy in biotechnology. Nat. Chem. Biol. 10, 319–322 (2014).
Nielsen, J. & Keasling, J. D. Engineering cellular metabolism. Cell 164, 1185–1197 (2016).
Green, E. M. Fermentative production of butanol–the industrial perspective. Curr. Opin. Biotechnol. 22, 337–343 (2011).
Jones, D. T. & Woods, D. R. Acetone-butanol fermentation revisited. Microbiol. Rev. 50, 484–524 (1986).
Tracy, B. P., Jones, S. W., Fast, A. G., Indurthi, D. C. & Papoutsakis, E. T. Clostridia: the importance of their exceptional substrate and metabolite diversity for biofuel and biorefinery applications. Curr. Opin. Biotechnol. 23, 364–381 (2012).
Takors, R. et al. Using gas mixtures of CO, CO2 and H2 as microbial substrates: the do’s and don’ts of successful technology transfer from laboratory to production scale. Microb. Biotechnol. 11, 606–625 (2018).
Charubin, K., Bennett, R. K., Fast, A. G. & Papoutsakis, E. T. Engineering Clostridium organisms as microbial cell-factories: challenges & opportunities. Metab. Eng. 50, 173–191 (2018).
Connelly Jr., T. M. et al. Industrialization of Biology: A Roadmap to Accelerate the Advanced Manufacturing of Chemicals (The National Academies Press, 2015).
Dudley, Q. M., Karim, A. S. & Jewett, M. C. Cell-free metabolic engineering: biomanufacturing beyond the cell. Biotechnol. J. 10, 69–82 (2015).
Morgado, G., Gerngross, D., Roberts, T. M. & Panke, S. Synthetic biology for cell-free biosynthesis: fundamentals of designing novel in vitro multi-enzyme reaction networks. Adv. Biochem Eng. Biotechnol. 162, 117–146 (2018).
Silverman, A. D., Karim, A. S. & Jewett, M. C. Cell-free gene expression: an expanded repertoire of applications. Nat. Rev. Genet. 21, 151–170 (2020).
Bogorad, I. W., Lin, T. S. & Liao, J. C. Synthetic non-oxidative glycolysis enables complete carbon conservation. Nature 502, 693–697 (2013).
Zhu, F. et al. In vitro reconstitution of mevalonate pathway and targeted engineering of farnesene overproduction in Escherichia coli. Biotechnol. Bioeng. 111, 1396–1405 (2014).
Karim, A. S. & Jewett, M. C. A cell-free framework for rapid biosynthetic pathway prototyping and enzyme discovery. Metab. Eng. 36, 116–126 (2016).
Dudley, Q. M., Anderson, K. C. & Jewett, M. C. Cell-free mixing of Escherichia coli crude extracts to prototype and rationally engineer high-titer mevalonate synthesis. ACS Synth. Biol. 5, 1578–1588 (2016).
Hold, C., Billerbeck, S. & Panke, S. Forward design of a complex enzyme cascade reaction. Nat. Commun. 7, 12971 (2016).
Kelwick, R. et al. Cell-free prototyping strategies for enhancing the sustainable production of polyhydroxyalkanoates bioplastics. Synth. Biol. 3, ysy016 (2018).
Kay, J. E. & Jewett, M. C. Lysate of engineered Escherichia coli supports high-level conversion of glucose to 2,3-butanediol. Metab. Eng. 32, 133–142 (2015).
Karim, A. S., Heggestad, J. T., Crowe, S. A. & Jewett, M. C. Controlling cell-free metabolism through physiochemical perturbations. Metab. Eng. 45, 86–94 (2018).
Dudley, Q. M., Nash, C. J. & Jewett, M. C. Cell-free biosynthesis of limonene using enzyme-enriched Escherichia coli lysates. Synth. Biol. 4, ysz003 (2019).
Grubbe, W. S., Rasor, B. J., Krüger, A., Jewett, M. C. & Karim, A. S. Cell-free styrene biosynthesis at high titers. Preprint at bioRxiv https://doi.org/10.1101/2020.03.05.979302 (2020).
Karim, A. S. & Jewett, M. C. Cell-free synthetic biology for pathway prototyping. Methods Enzymol. 608, 31–57 (2018).
Clomburg, J. M., Crumbley, A. M. & Gonzalez, R. Industrial biomanufacturing: the future of chemical production. Science 355, 6320 (2017).
Tseng, H. C., Martin, C. H., Nielsen, D. R. & Prather, K. L. Metabolic engineering of Escherichia coli for enhanced production of (R)- and (S)-3-hydroxybutyrate. Appl. Environ. Microbiol. 75, 3137–3145 (2009).
McMahon, M. D. & Prather, K. L. Functional screening and in vitro analysis reveal thioesterases with enhanced substrate specificity profiles that improve short-chain fatty acid production in Escherichia coli. Appl. Environ. Microbiol. 80, 1042–1050 (2014).
Jewett, M. C. & Swartz, J. R. Mimicking the Escherichia coli cytoplasmic environment activates long-lived and efficient cell-free protein synthesis. Biotechnol. Bioeng. 86, 19–26 (2004).
Jewett, M. C., Calhoun, K. A., Voloshin, A., Wuu, J. J. & Swartz, J. R. An integrated cell-free metabolic platform for protein production and synthetic biology. Mol. Syst. Biol. 4, 220 (2008).
Woolston, B. M., Emerson, D. F., Currie, D. H. & Stephanopoulos, G. Rediverting carbon flux in Clostridium ljungdahlii using CRISPR interference (CRISPRi). Metab. Eng. 48, 243–253 (2018).
Fluchter, S. et al. Anaerobic production of poly(3-hydroxybutyrate) and its precursor 3-hydroxybutyrate from synthesis gas by autotrophic Clostridia. Biomacromolecules 20, 3271–3282 (2019).
Atsumi, S. et al. Metabolic engineering of Escherichia coli for 1-butanol production. Metab. Eng. 10, 305–311 (2008).
Inui, M. et al. Expression of Clostridium acetobutylicum butanol synthetic genes in Escherichia coli. Appl. Microbiol. Biotechnol. 77, 1305–1316 (2008).
Shen, C. R. et al. Driving forces enable high-titer anaerobic 1-butanol synthesis in Escherichia coli. Appl. Environ. Microbiol. 77, 2905–2915 (2011).
Nguyen, N. P., Raynaud, C., Meynial-Salles, I. & Soucaille, P. Reviving the Weizmann process for commercial n-butanol production. Nat. Commun. 9, 3682 (2018).
Li, F. et al. Coupled ferredoxin and crotonyl coenzyme A (CoA) reduction with NADH catalyzed by the butyryl-CoA dehydrogenase/Etf complex from Clostridium kluyveri. J. Bacteriol. 190, 843–850 (2008).
Qi, F. et al. Improvement of butanol production in Clostridium acetobutylicum through enhancement of NAD(P)H availability. J. Ind. Microbiol. Biotechnol. 45, 993–1002 (2018).
Köpke, M. et al. Clostridium ljungdahlii represents a microbial production platform based on syngas. Proc. Natl Acad. Sci. USA 107, 13087–13092 (2010).
Chowdhury, N. P., Kahnt, J. & Buckel, W. Reduction of ferredoxin or oxygen by flavin-based electron bifurcation in Megasphaera elsdenii. FEBS J. 282, 3149–3160 (2015).
Gao, H. J., Wu, Q. & Chen, G. Q. Enhanced production of D-(−)-3-hydroxybutyric acid by recombinant Escherichia coli. FEMS Microbiol. Lett. 213, 59–65 (2002).
Yun, E. J. et al. Production of (S)-3-hydroxybutyrate by metabolically engineered Saccharomyces cerevisiae. J. Biotechnol. 209, 23–30 (2015).
Liew, F. et al. Metabolic engineering of Clostridium autoethanogenum for selective alcohol production. Metab. Eng. 40, 104–114 (2017).
Perez, J. M., Richter, H., Loftus, S. E. & Angenent, L. T. Biocatalytic reduction of short-chain carboxylic acids into their corresponding alcohols with syngas fermentation. Biotechnol. Bioeng. 110, 1066–1077 (2013).
Kataoka, N. et al. Enhancement of (R)-1,3-butanediol production by engineered Escherichia coli using a bioreactor system with strict regulation of overall oxygen transfer coefficient and pH. Biosci. Biotechnol., Biochem. 78, 695–700 (2014).
Nemr, K. et al. Engineering a short, aldolase-based pathway for (R)-1,3-butanediol production in Escherichia coli. Metab. Eng. 48, 13–24 (2018).
Jing, F. et al. Direct dehydration of 1,3-butanediol into butadiene over aluminosilicate catalysts. Catal. Sci. Technol. 6, 5830–5840 (2016).
Heijstra, B. D., Kern, E., Koepke, M., Segovia, S. & Liew, F. M. Novel bacteria and methods of use thereof. US patent 20130217096A1 (2013).
Liew, F. et al. Gas fermentation: a flexible platform for commercial scale production of low-carbon fuels and chemicals from waste and renewable feedstocks. Front. Microbiol. 7, 694 (2016).
Heap, J. T., Pennington, O. J., Cartman, S. T. & Minton, N. P. A modular system for Clostridium shuttle plasmids. J. Microbiol. Methods 78, 79–85 (2009).
Nagaraju, S., Davies, N. K., Walker, D. J., Kopke, M. & Simpson, S. D. Genome editing of Clostridium autoethanogenum using CRISPR/Cas9. Biotechnol. Biofuels 9, 219 (2016).
Köpke, M. et al. 2,3-Butanediol production by acetogenic bacteria, an alternative route to chemical synthesis, using industrial waste gas. Appl. Environ. Microbiol. 77, 5467–5475 (2011).
Kwon, Y. C. & Jewett, M. C. High-throughput preparation methods of crude extract for robust cell-free protein synthesis. Sci. Rep. 5, 8663 (2015).
Jewett, M. C. & Swartz, J. R. Substrate replenishment extends protein synthesis with an in vitro translation system designed to mimic the cytoplasm. Biotechnol. Bioeng. 87, 465–472 (2004).
Koepke, M., Jensen, R. O., Behrendorff, J. B. Y. H. & Hill, R. E. Genetically engineered bacterium comprising energy-generating fermentation pathway. US patent 9,738,875 (2017).
Valgepea, K. et al. Maintenance of ATP homeostasis triggers metabolic shifts in gas-fermenting acetogens. Cell Syst. 4, 505–515 (2017).
Haykin, S. Neural Networks: A Comprehensive Foundation (Prentice Hall PTR, 1994).
Whitley, D. A genetic algorithm tutorial. Stat. Comput. 4, 65–85 (1994).
Nelder, J. A. & Mead, R. A simplex method for function minimization. Computer J. 7, 308–313 (1965).
Acknowledgements
We thank A. M. Mueller, R. T. Tappel, W. Allen, L. Tran and S. D. Brown (LanzaTech) for conversations regarding this work. In addition, we thank C. Reynolds (Lockheed Martin) for conversations on the design of experiments using neural networks. This work is supported by the US Department of Energy, Office of Biological and Environmental Research in the Department of Environment Office of Science under award number DE-SC0018249. M.C.J. gratefully acknowledges the David and Lucile Packard Foundation and the Camille Dreyfus Teacher–Scholar Program. We also thank the following investors in LanzaTech’s technology: BASF, CICC Growth Capital Fund I, CITIC Capital, Indian Oil Company, K1W1, Khosla Ventures, the Malaysian Life Sciences, Capital Fund, L. P., Mitsui, the New Zealand Superannuation Fund, Petronas Technology Ventures, Primetals, Qiming Venture Partners, Softbank China and Suncor.
Author information
Authors and Affiliations
Contributions
A.S.K., S.D.S., M.K. and M.C.J. designed the study. A.S.K., Q.M.D. and M.C.J. developed the cell-free framework. A.S.K., S.A.C., J.T.H., W.S.G. and B.J.R. performed all cell-free experiments. A.S.K. and Q.M.D. analyzed cell-free data. A.J. performed Clostridium strain engineering for 3-HB and 1,3-BDO. T.A. performed C. autoethanogenum gas fermentation for 3-HB and 1,3-BDO. Y.Y., F.E.L., R.O.J., S.G. and M.K. performed C. autoethanogenum strain engineering and gas fermentation for butanol. A.J., Y.Y. and M.K.. analyzed C. autoethanogenum data. A.Q. developed analytical methods for 3-HB, 1,3-BDO and butanol. D.C., M.T., M.Kr. and J.S. performed all design of experiments using neural networks. A.S.K., M.K. and M.C.J. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
A.J., T.A., S.G., A.Q., Y.Y., F.E.L., R.O.J., S.D.S. and M.K. are employees of LanzaTech, which has commercial interest in gas fermentation with C. autoethanogenum. Production of 3-HB, 1,3-BDO and 1-butanol from C1 gases has been patented (US patents 9,738,875 and 9,359,611). A.S.K. and M.C.J. are co-inventors on the US provisional patent application 62/173,818 that incorporates discoveries described in this manuscript. All other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2, Figs. 1–11 and Notes 1 and 2.
Supplementary Dataset 1
The dataset contains five sheets related to the 3-HB data presented in this manuscript. Sheet 1 provides enzyme nomenclature, sheet 2 lists enzyme combinations, sheet 3 lists enzyme concentrations in final reactions, sheet 4 lists metabolite concentrations over time and sheet 5 lists all TREE scores and component parts.
Supplementary Dataset 2
The dataset contains five sheets related to the butanol data presented in this manuscript. Sheet 1 provides enzyme nomenclature, sheet 2 lists enzyme combinations, sheet 3 lists enzyme concentrations in final reactions, sheet 4 lists metabolite concentrations over time and sheet 5 lists all TREE scores and component parts.
Rights and permissions
About this article
Cite this article
Karim, A.S., Dudley, Q.M., Juminaga, A. et al. In vitro prototyping and rapid optimization of biosynthetic enzymes for cell design. Nat Chem Biol 16, 912–919 (2020). https://doi.org/10.1038/s41589-020-0559-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-020-0559-0
This article is cited by
-
Bioprocessing 4.0 in biomanufacturing: paving the way for sustainable bioeconomy
Systems Microbiology and Biomanufacturing (2024)
-
Modular bioengineering of whole-cell catalysis for sialo-oligosaccharide production: coordinated co-expression of CMP-sialic acid synthetase and sialyltransferase
Microbial Cell Factories (2023)
-
Machine learning-enabled retrobiosynthesis of molecules
Nature Catalysis (2023)
-
Enabling technology and core theory of synthetic biology
Science China Life Sciences (2023)
-
Metabolic engineering strategies for microbial utilization of C1 feedstocks
Systems Microbiology and Biomanufacturing (2023)