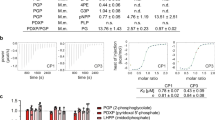
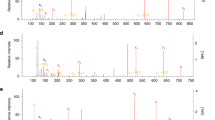
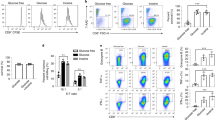
Abstract
Glucose is catabolized by two fundamental pathways, glycolysis to make ATP and the oxidative pentose phosphate pathway to make reduced nicotinamide adenine dinucleotide phosphate (NADPH). The first step of the oxidative pentose phosphate pathway is catalyzed by the enzyme glucose-6-phosphate dehydrogenase (G6PD). Here we develop metabolite reporter and deuterium tracer assays to monitor cellular G6PD activity. Using these, we show that the most widely cited G6PD antagonist, dehydroepiandosterone, does not robustly inhibit G6PD in cells. We then identify a small molecule (G6PDi-1) that more effectively inhibits G6PD. Across a range of cultured cells, G6PDi-1 depletes NADPH most strongly in lymphocytes. In T cells but not macrophages, G6PDi-1 markedly decreases inflammatory cytokine production. In neutrophils, it suppresses respiratory burst. Thus, we provide a cell-active small molecule tool for oxidative pentose phosphate pathway inhibition, and use it to identify G6PD as a pharmacological target for modulating immune response.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Voet, D., Voet, J. G. & Pratt, C. W. Fundamentals of Biochemistry: Life at the Molecular Level 5th edn (Wiley, 2016).
Stanton, R. C. Glucose-6-phosphate dehydrogenase, NADPH, and cell survival. IUBMB Life 64, 362–369 (2012).
Uhlen, M. et al. Proteomics. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Kowalik, M. A., Columbano, A. & Perra, A. Emerging role of the pentose phosphate pathway in hepatocellular carcinoma. Front. Oncol. 7, https://doi.org/10.3389/fonc.2017.00087 (2017).
Zhang, Q. et al. Overexpression of G6PD represents a potential prognostic factor in clear cell renal cell carcinoma. J. Cancer 8, 665–673 (2017).
Nagashio, R. et al. Prognostic significance of G6PD expression and localization in lung adenocarcinoma. Biochim. Biophys. Acta 1867, 38–46 (2019).
Pu, H. et al. Overexpression of G6PD is associated with high risks of recurrent metastasis and poor progression-free survival in primary breast carcinoma. World J. Surgical Oncol. 13, 323–323 (2015).
Longo, L. et al. Maternally transmitted severe glucose 6‐phosphate dehydrogenase deficiency is an embryonic lethal. EMBO J. 21, 4229–4239 (2002).
Cappellini, M. D. & Fiorelli, G. Glucose-6-phosphate dehydrogenase deficiency. Lancet 371, 64–74 (2008).
Chen, L. et al. NADPH production by the oxidative pentose-phosphate pathway supports folate metabolism. Nat. Metabolism 1, 404–415 (2019).
Hamilton, N. M. et al. Novel steroid inhibitors of glucose 6-phosphate dehydrogenase. J. Med. Chem. 55, 4431–4445 (2012).
Preuss, J. et al. Identification and characterization of novel human glucose-6-phosphate dehydrogenase inhibitors. J. Biomol. Screening 18, 286–297 (2012).
Mele, L. et al. A new inhibitor of glucose-6-phosphate dehydrogenase blocks pentose phosphate pathway and suppresses malignant proliferation and metastasis in vivo. Cell Death Dis. 9, 572 (2018).
Marks, P. A. & Banks, J. Inhibition of mammalian glucose-6-phosphate dehydrogenase by steroids. Proc. Natl Acad. Sci. USA 46, 447–452 (1960).
Di Monaco, M. et al. Role of glucose-6-phosphate dehydrogenase inhibition in the antiproliferative effects of dehydroepiandrosterone on human breast cancer cells. Brit. J. Cancer 75, 589–592 (1997).
Pashko, L. L., Lewbart, M. L. & Schwartz, A. G. Inhibition of 12-O-tetradecanoylphorbol-13-acetate-promoted skin tumor formation in mice by 16α-fluoro-5-androsten-17-one and its reversal by deoxyribonucleosides. Carcinogenesis 12, 2189–2192 (1991).
Girón, R. A., Montaño, L. F., Escobar, M. L. & López-Marure, R. Dehydroepiandrosterone inhibits the proliferation and induces the death of HPV-positive and HPV-negative cervical cancer cells through an androgen- and estrogen-receptor independent mechanism. FEBS J. 276, 5598–5609 (2009).
Ho, H. Y., Cheng, M. L., Chiu, H. Y., Weng, S. F. & Chiu, D. T. Dehydroepiandrosterone induces growth arrest of hepatoma cells via alteration of mitochondrial gene expression and function. Int. J. Oncol. 33, 969–977 (2008).
Pacold, M. E. et al. A PHGDH inhibitor reveals coordination of serine synthesis and one-carbon unit fate. Nat. Chem. Biol. 12, 452 (2016).
Mullarky, E. et al. Identification of a small molecule inhibitor of 3-phosphoglycerate dehydrogenase to target serine biosynthesis in cancers. Proc. Natl Acad. Sci. USA 113, 1778 (2016).
Ducker, G. S. et al. Human SHMT inhibitors reveal defective glycine import as a targetable metabolic vulnerability of diffuse large B-cell lymphoma. Proc. Natl Acad. Sci. USA 114, 11404–11409 (2017).
Lu, W., Wang, L., Chen, L., Hui, S. & Rabinowitz, J. D. Extraction and quantitation of nicotinamide adenine dinucleotide redox cofactors. Antioxidant. Redox Signal. 28, 167–179 (2018).
Gordon, G., Mackow, M. C. & Levy, H. R. On the mechanism of interaction of steroids with human glucose 6-phosphate dehydrogenase. Arch. Biochem. Biophys. 318, 25–29 (1995).
Zhang, Z., Chen, L., Liu, L., Su, X. & Rabinowitz, J. D. Chemical basis for deuterium labeling of fat and NADPH. J. Am. Chem. Soc. 139, 14368–14371 (2017).
Mercaldi, G. F., Ranzani, A. T. & Cordeiro, A. T. Discovery of new uncompetitive inhibitors of glucose-6-phosphate dehydrogenase. J. Biomol. Screening 19, 1362–1371 (2014).
Schafer, Z. T. et al. Antioxidant and oncogene rescue of metabolic defects caused by loss of matrix attachment. Nature 461, 109–113 (2009).
Buck, M. D., O’Sullivan, D. & Pearce, E. L. T cell metabolism drives immunity. J. Experimental Med. 212, 1345–1360 (2015).
Nobrega-Pereira, S. et al. G6PD protects from oxidative damage and improves healthspan in mice. Nat. Commun. 7, 10894 (2016).
Ron-Harel, N. et al. Mitochondrial biogenesis and proteome remodeling promote one-carbon metabolism for T cell activation. Cell Metab. 24, 104–117 (2016).
Geiger, R. et al. l-Arginine modulates T cell metabolism and enhances survival and anti-tumor activity. Cell 167, 829–842.e813 (2016).
Sena, L. A. et al. Mitochondria are required for antigen-specific T cell activation through reactive oxygen species signaling. Immunity 38, 225–236 (2013).
Padgett, L. E. & Tse, H. M. NADPH oxidase-derived superoxide provides a third signal for CD4 T cell effector responses. J. Immunol. 197, 1733–1742 (2016).
Mak, T. W. et al. Glutathione primes T cell metabolism for inflammation. Immunity 46, 675–689 (2017).
Nguyen, G. T., Green, E. R. & Mecsas, J. Neutrophils to the ROScue: mechanisms of NADPH oxidase activation and bacterial resistance. Front. Cell Infect. Microbiol. 7, 373 (2017).
Goldfarb, A. H., McIntosh, M. K. & Boyer, B. T. Vitamin E attenuates myocardial oxidative stress induced by DHEA in rested and exercised rats. J. Appl. Physiol. 80, 486–490 (1996).
Fan, J. et al. Quantitative flux analysis reveals folate-dependent NADPH production. Nature 510, 298–302 (2014).
Morelli, A., Benatti, U., Gaetani, G. F. & De Flora, A. Biochemical mechanisms of glucose-6-phosphate dehydrogenase deficiency. Proc. Natl Acad. Sci. USA 75, 1979–1983 (1978).
Marks, P. A., Johnson, A. B. & Hirschberg, E. Effect of age on the enzyme activity in erythrocytes. Proc. Natl Acad. Sci. USA 44, 529–536 (1958).
Cunningham, A. D., Colavin, A., Huang, K. C. & Mochly-Rosen, D. Coupling between protein stability and catalytic activity determines pathogenicity of G6PD variants. Cell Rep. 18, 2592–2599 (2017).
Morellini, M., Colonna-Romano, S., Meloni, T., Battistuzzi, G. & Gandini, E. Glucose-6-phosphate dehydrogenase of leukocyte subpopulations in normal and enzyme deficient individuals. Haematologica 70, 390–395 (1985).
Ardati, K. O., Bajakian, K. M. & Tabbara, K. S. Effect of glucose-6-phosphate dehydrogenase deficiency on neutrophil function. Acta Haematologica 97, 211–215 (1997).
Vives Corrons, J. L. et al. Severe-glucose-6-phosphate dehydrogenase (G6PD) deficiency associated with chronic hemolytic anemia, granulocyte dysfunction, and increased susceptibility to infections: description of a new molecular variant (G6PD Barcelona). Blood 59, 428–434 (1982).
Macintyre, A. N. et al. The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab. 20, 61–72 (2014).
Ho, P. C. et al. phosphoenolpyruvate is a metabolic checkpoint of anti-tumor T cell responses. Cell 162, 1217–1228 (2015).
Cham, C. M. & Gajewski, T. F. Glucose availability regulates IFN-gamma production and p70S6 kinase activation in CD8+ effector T cells. J. Immunol. 174, 4670–4677 (2005).
Shi, L. Z. et al. HIF1α–dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J. Experimental Med. 208, 1367–1376 (2011).
Chang, C. H. et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153, 1239–1251 (2013).
Martinez Molina, D. et al. Monitoring drug target engagement in cells and tissues using the cellular thermal shift assay. Science 341, 84–87 (2013).
Ron-Harel, N. et al. T cell activation depends on extracellular alanine. Cell Rep. 28, 3011–3021.e3014 (2019).
Xiao, H. et al. HDAC5 controls the functions of Foxp3(+) T-regulatory and CD8(+) T cells. Internat. J. Cancer 138, 2477–2486 (2016).
Wang, R. et al. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity 35, 871–882 (2011).
Katz, J. & Wood, H. G. The use of C14O2 yields from glucose-1- and -6-C14 for the evaluation of the pathways of glucose metabolism. J. Biol. Chem. 238, 517–523 (1963).
Lu, W. et al. Metabolomic analysis via reversed-phase ion-pairing liquid chromatography coupled to a stand alone orbitrap mass spectrometer. Anal. Chem. 82, 3212–3221 (2010).
Seim, G. L. et al. Two-stage metabolic remodelling in macrophages in response to lipopolysaccharide and interferon-γ stimulation. Nat. Metabolism 1, 731–742 (2019).
Su, X., Lu, W. & Rabinowitz, J. D. Metabolite spectral accuracy on orbitraps. Anal. Chem. 89, 5940–5948 (2017).
Acknowledgements
We thank C. DeCoste of the Princeton University flow cytometry resource facility and the Cytomics Unit of the IIS-La Fe for experimental set-up and design; R.S. O’Connor of University of Pennsylvania for assistance in setting up T cell experiments and for comments and suggestions on the figures; J. Jiao of the Children’s Hospital of Philadelphia for technical assistance with the Treg experiments; I. Babic of Nerdbio for assistance with the CETSA experiments; Y. Huang of Peking University for helpful suggestions pertaining to structure–activity relationship analysis and C. Bartman and the rest of members of the Rabinowitz laboratory for comments and suggestions. This work was supported by National Institutes of Health grant nos. 1DP1DK113643 and R01 CA163591. J.C.G.C. is supported by funding from the European Union’s Horizon 2020 research and innovation program under the Marie Sklodowska-Curie grant agreement no. 751423.
Author information
Authors and Affiliations
Contributions
J.D.R., J.M.G., J.C.G.-C. and H.K. conceived the study and designed the experiments. J.M.G. developed the in vitro and cell-based assays, conducted the biochemical characterization of G6PDi-1 and characterized the metabolic effects of G6PDi-1 HCT116, HepG2 and other adherent cell lines. J.C.G.C. characterized the metabolic effects of G6PDi-1 in suspension cell lines and the functional effects of G6PDi-1 in T cells and macrophages. J.W., E.S. and H.P. conducted protein expression and purification, in vitro activity assays and western blotting. L.C. isolated mPGD HCT116 cells. Z.Z. and T.T. isolated and cultured primary murine hepatocytes. U.H.B. designed and conducted the Treg experiments. E.C.B. and J.F. designed and conducted the neutrophil experiments. M.C.G.-C. provided the G6PD transgenic mice. J.C.G.C., M.P.-N., M.C.G.-C. and A.L. designed and conducted the experiments with G6PD transgenic mice. H.K. and J.M.G. conducted the structure–activity relationship analysis. H.K. designed and oversaw the chemical synthetic strategy. J.D.R., J.M.G. and J.C.G.-C. wrote the paper. All authors edited and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
Princeton University has filed a patent relating to the new G6PD inhibitors and their uses. J.D.R. is a cofounder of Raze Therapeutics, advisor and stock owner in Kadmon, Agios, CRP, LEAF and Bantam Pharmaceuticals and consultant to Pfizer. No competing interests were disclosed by the other authors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–3, Figs. 1–16 and Note.
Rights and permissions
About this article
Cite this article
Ghergurovich, J.M., García-Cañaveras, J.C., Wang, J. et al. A small molecule G6PD inhibitor reveals immune dependence on pentose phosphate pathway. Nat Chem Biol 16, 731–739 (2020). https://doi.org/10.1038/s41589-020-0533-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-020-0533-x
This article is cited by
-
RORα inhibits gastric cancer proliferation through attenuating G6PD and PFKFB3 induced glycolytic activity
Cancer Cell International (2024)
-
Mitochondrial ATP generation is more proteome efficient than glycolysis
Nature Chemical Biology (2024)
-
Metabolic reprogramming in colorectal cancer: regulatory networks and therapy
Cell & Bioscience (2023)
-
Cyclosporine A alleviates colitis by inhibiting the formation of neutrophil extracellular traps via the regulating pentose phosphate pathway
Molecular Medicine (2023)
-
Neutrophil metabolomics in severe COVID-19 reveal GAPDH as a suppressor of neutrophil extracellular trap formation
Nature Communications (2023)