Abstract
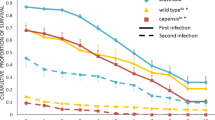
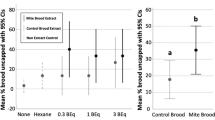
Invasive species events related to globalization are increasing, resulting in parasitic outbreaks. Understanding of host defense mechanisms is needed to predict and mitigate against the consequences of parasite invasion. Using the honey bee Apis mellifera and the mite Varroa destructor, as a host–parasite model, we provide a comprehensive study of a mechanism of parasite detection that triggers a behavioral defense associated with social immunity. Six Varroa-parasitization-specific (VPS) compounds are identified that (1) trigger Varroa-sensitive hygiene (VSH, bees’ key defense against Varroa sp.), (2) enable the selective recognition of a parasitized brood and (3) induce responses that mimic intrinsic VSH activity in bee colonies. We also show that individuals engaged in VSH exhibit a unique ability to discriminate VPS compounds from healthy brood signals. These findings enhance our understanding of a critical mechanism of host defense against parasites, and have the potential to apply the integration of pest management in the beekeeping sector.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated for the present study are available upon request to the corresponding author.
Code availability
All computer codes generated for the present study are available upon request to the corresponding author.
References
Bertelsmeier, C., Ollier, S., Liebhold, A. & Keller, L. Recent human history governs global ant invasion dynamics. Nat. Ecol. Evol. 1, 0184 (2017).
Ugelvig, L. V. & Cremer, S. Effects of social immunity and unicoloniality on host–parasite interactions in invasive insect societies. Funct. Ecol. 26, 1300–1312 (2012).
Mack, R. et al. Biotic invasions: causes, epidemiology, global consequences, and control. Ecol. Appl. 10, 689–710 (2000).
Essl, F. et al. Socioeconomic legacy yields an invasion debt. Proc. Natl Acad. Sci. USA 108, 203–207 (2011).
Boomsma, J. J., Schmid-Hempel, P. & Hughes, W. O. H. in Insect Evolutionary Ecology (eds Fellowes, M. et al.) 139–175 (CABI, 2005).
Cremer, S., Armitage, S. A. & Schmid-Hempel, P. Social immunity. Curr. Biol. 17, 693–702 (2007).
Parker, B. J., Barribeau, S. M., Laughton, A. M., de Roode, J. C. & Gerardo, N. M. Non-immunological defense in an evolutionary framework. Trends Ecol. Evol. 26, 242–248 (2011).
De Roode, J. C. & Lefèvre, T. Behavioral immunity in insects. Insects 3, 789–820 (2012).
Wyatt, T. D. Pheromones and Animal Behaviour: Communication by Smell and Taste (Cambridge Univ. Press, 2003).
Penn, D. & Potts, W. K. Chemical signals and parasite-mediated sexual selection. Trends Ecol. Evol. 13, 391–396 (1998).
Bagnères, A. G. & Lorenzi, M. C. in Insect Hydrocarbons: Biology, Biochemistry and Chemical Ecology (eds Blomquist G. J. & Bagnères A. G.) 282–324 (Cambridge Univ. Press, 2010).
Baracchi, D., Fadda, A. & Turillazzi, S. Evidence for antiseptic behaviour towards sick adult bees in honey bee colonies. J. Insect Physiol. 58, 1589–1596 (2012).
Schöning, C. et al. Evidence for damage-dependent hygienic behaviour towards Varroa destructor-parasitised brood in the western honey bee, Apis mellifera. J. Exp. Biol. 215, 264–271 (2012).
Csata, E. et al. Lock-picks: fungal infection facilitates the intrusion of strangers into ant colonies. Sci. Rep. 7, 46323 (2017).
Pull, C. D., Metzler, S., Naderlinger, E. & Cremer, S. Protection against the lethal side effects of social immunity in ants. Curr. Biol. 28, 1139–1140 (2018).
Nazzi, F., Della Vedova, G. & d’Agaro, M. A semiochemical from brood cells infested by Varroa destructor triggers hygienic behaviour in Apis mellifera. Apidologie 35, 65–70 (2004).
Swanson, J. A. et al. Odorants that induce hygienic behavior in honeybees: identification of volatile compounds in chalkbrood-infected honeybee larvae. J. Chem. Ecol. 35, 1108–1116 (2009).
Wagoner, K., Spivak, M., Hefetz, A., Reams, T. & Rueppell, O. Stock-specific chemical brood signals are induced by Varroa and deformed wing virus, and elicit hygienic response in the honey bee. Sci. Rep. 9, 8753 (2019).
Sun, Q., Haynes, K. F. & Zhou, X. Dynamic changes in death cues modulate risks and rewards of corpse management in a social insect. Funct. Ecol. 31, 697–706 (2017).
Qiu, H. L. et al. Differential necrophoric behaviour of the ant Solenopsis invicta towards fungal-infected corpses of workers and pupae. Bull. Entomol. Res. 105, 607–614 (2015).
Wagoner, K. M., Millar, J. G., Schal, C. & Rueppell, O. Cuticular pheromones stimulate hygienic behavior in the honey bee (Apis mellifera). Sci. Rep. 10, 7132(2020).
Traynor, K. et al. Varroa destructor: a complex parasite, crippling honeybees worldwide. Trends Parasitol. 36, 592–606 (2020).
Mondet, F. et al. Honey bee survival mechanisms against the parasite Varroa destructor: a systematic review of phenotypic and genomic research efforts. Int. J. Parasitol. 50, 433–447 (2020).
Harbo, J. R. & Harris, J. W. Suppressed mite reproduction explained by the behaviour of adult bees. J. Apic. Res. 44, 21–23 (2005).
Nazzi, F. & Le Conte, Y. Ecology of Varroa destructor, the major ectoparasite of the western honey bee, Apis mellifera. Annu. Rev. Entomol. 61, 417–432 (2016).
Rosenkranz, P., Tewarson, N., Singh, A. & Engels, W. Differential hygienic behaviour towards Varroa jacobsoni in capped worker brood of Apis cerana depends on alien scent adhering to the mites. J. Apic. Res. 32, 89–93 (1993).
Mondet, F. et al. Antennae hold a key to Varroa-sensitive hygiene behaviour in honey bees. Sci. Rep. 5, 10454 (2015).
Martin, C. et al. Variations in chemical mimicry by the ectoparasitic mite Varroa jacobsoni according to the developmental stage of the host honey-bee Apis mellifera. Insect Biochem. Mol. Biol. 31, 365–379 (2001).
Martin, C. et al. Potential mechanism for detection by Apis mellifera of the parasitic mite Varroa destructor inside sealed brood cells. Physiol. Entomol. 27, 175–188 (2002).
Ziegelmann, B. & Rosenkranz, P. Mating disruption of the honeybee mite Varroa destructor under laboratory and field conditions. Chemoecology 24, 137–144 (2014).
Le Conte, Y., Arnold, G., Trouiller, J., Masson, C. & Chappe, B. Identification of a brood pheromone in honeybees. Naturwissenschaften 77, 334–336 (1990).
Mondet, F. et al. Specific cues associated with honey bee social defence against Varroa destructor infested brood. Sci. Rep. 6, 25444 (2016).
Harris, J. W. Bees with varroa sensitive hygiene preferentially remove mite infested pupae aged ≤five days post capping. J. Apic. Res. 46, 134–139 (2007).
Harbo, J. R. & Harris, J. W. Responses to Varroa by honey bees with different levels of varroa sensitive hygiene. J. Apic. Res. 48, 156–161 (2009).
Infantidis, M. D. Ontogenesis of the mite Varroa jacobsoni in worker and done honeybee brood cells. J. Agric. Res. 22, 200–206 (1983).
Blomquist, G. J., A. J. Chu, A. J. & Remaley, S. Biosynthesis of wax in the honeybee, Apis mellifera L. Insect Biochem. 10, 313–321 (1980).
Donzé, G. et al. Aliphatic alcohols and aldehydes of the honey bee cocoon induce arrestment behavior in Varroa jacobsoni (Acari: Mesostigmata), an ectoparasite of Apis mellifera. Arch. Insect Biochem. Physiol. 37, 129–145 (1998).
Shearer, D. A. & Boch, R. 2-Heptanone in the mandibular gland secretion of the honey-bee. Nature 206, 530–530 (1965).
Salvy, M. et al. Modifications of the cuticular hydrocarbon profile of Apis mellifera worker bees in the presence of the ectoparasitic mite Varroa jacobsoni in brood cells. Parasitology 122, 145–159 (2001).
Harris, J. W., Danka, R. G. & Villa, J. D. Honey bees (Hymenoptera: Apidae) with the trait of Varroa sensitive hygiene remove brood with all reproductive stages of Varroa mites (Mesostigmata: Varroidae). Ann. Entomol. Soc. Am. 103, 146–152 (2010).
McAfee, A. et al. A death pheromone, oleic acid, triggers hygienic behavior in honey bees (Apis mellifera L.). Sci. Rep. 8, 5719 (2018).
Slessor, K. N., Winston, M. L. & Le Conte, Y. Pheromone communication in the honeybee (Apis mellifera L.). J. Chem. Ecol. 31, 2731–2745 (2005).
Kim, S. H., Mondet, F., Hervé, M. & Mercer, A. Honey bees performing varroa sensitive hygiene remove the most mite-compromised bees from highly infested patches of brood. Apidologie 49, 335–345 (2018).
Galizia, C. G. & Sachse, S. Odor coding in insects. in The Neurobiology of Olfaction (ed. Menini, A.) 35–70 (CRC Press/Taylor & Francis, 2010).
McAfee, A., Collins, T. F., Madilao, L. L. & Foster, L. J. Odorant cues linked to social immunity induce lateralized antenna stimulation in honey bees (Apis mellifera L.). Sci. Rep. 7, 46171 (2017).
Villa, J. D., Danka, R. G. & Harris, J. W. Simplified methods of evaluating colonies for levels of varroa sensitive hygiene (VSH). J. Apic. Res. 48, 162–167 (2009).
Grob, R. L., Kaiser, M. A., Grob, R. L. & Barry, E. F. in Modern Practice of Gas Chromatography, 4th edn (ed. Grob, R. L.) 403–460 (Wiley, 2004).
Sparkman, O. D., Penton, Z. & Kitson, F. G. Gas Chromatography and Mass Spectrometry: A Practical Guide (Academic Press, 2011).
Arathi, H. S., Burns, I. & Spivak, M. Ethology of hygienic behaviour in the honey bee Apis mellifera L. (Hymenoptera: Apidae): behavioural repertoire of hygienic bees. Ethology 106, 365–379 (2000).
Dietemann, V. et al. Standard methods for varroa research. J. Apic. Res. 52, 1–54 (2013).
Matsumoto, Y., Menzel, R., Sandoz, J. C. & Giurfa, M. Revisiting olfactory classical conditioning of the proboscis extension response in honey bees: a step toward standardized procedures. J. Neurosci. Methods 211, 159–167 (2012).
Scheiner, R., Page, R. E. Jr. & Erber, J. Responsiveness to sucrose affects tactile and olfactory learning in pre-foraging honey bees of two genetic strains. Behav. Brain Res. 120, 67–73 (2001).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 57, 289–300 (1995).
Acknowledgements
We thank Y. Poquet for help in the collection of VSH bees and mites, F. Laas for providing mite-infested colonies, D. Crauser and J. Senechal for giving access to honey bee colonies, and GDR Mediatec for fruitful scientific discussions. This project was funded by grants provided by INRAE (SPE Department), the University of Otago and a Casdar fund from the French Ministry of Agriculture (Mosar). F.M. was supported by grants from the France–New Zealand Friendship Fund, by the French Ministry of Agriculture and a New Zealand international doctoral research scholarship. MS analyses were in part performed at the Plateforme d’Analyses Chimiques en Ecologie, technical facilities of the CeMEB.
Author information
Authors and Affiliations
Contributions
F.M., A.R.M. and Y.L.C. designed the project. F.M., S.B., D.B., N.B., B.L., C.B., S.H.K., B.B. and G.C. performed the experiments with contributions as follows. F.M., S.B., C.B., B.B. did the field assays. F.M., S.B. and D.B. did the GC analyses. F.M., N.B. and G.C. did the MS analyses. F.M. and S.H.K. performed the sucrose responsiveness assay. F.M. performed the learning assay. F.M., C.B. and B.L. did the EAGs. F.M. and M.H. analyzed the data. F.M., A.R.M. and Y.L.C. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 One-hour memory recall in VSH bees and NVS bees.
Percentages of VSH bees and NVS bees displaying conditioned proboscis extension responses (PER) 1 hour after the final (6th) conditioning trial. All bees were trained to discriminate between an odorant paired with a sugar reward (CS+, healthy brood extract + VPS compounds) and a non-reinforced odorant (CS-, healthy brood extract). The figure shows the percentages of bees responding to the CS+, the CS- and to a novel odour not presented during the learning phase (Diff, Nonanol), (GLMM, nVSH = 32, nNVS = 28). Different letters indicate significant results in the analyses.
Supplementary information
Rights and permissions
About this article
Cite this article
Mondet, F., Blanchard, S., Barthes, N. et al. Chemical detection triggers honey bee defense against a destructive parasitic threat. Nat Chem Biol 17, 524–530 (2021). https://doi.org/10.1038/s41589-020-00720-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-020-00720-3
This article is cited by
-
The impact of honey bee (Apis mellifera) group size on hygienic behavior performance
Behavioral Ecology and Sociobiology (2024)
-
Honey bees (Apis mellifera) preselected for Varroa sensitive hygiene discriminate between live and dead Varroa destructor and inanimate objects
Scientific Reports (2023)
-
Varroa resistance in Apis cerana: a review
Apidologie (2023)
-
Apis mellifera Worker Bees Selected for Varroa-sensitive Hygiene Show Higher Specific Sensitivity and Perception Speed Towards Low Concentrations of Chemical Cues Emitted by the Brood
Journal of Insect Behavior (2023)
-
Protecting pollinators and our food supply: understanding and managing threats to pollinator health
Insectes Sociaux (2023)