Abstract
Mitochondrial DNA (mtDNA) mutations are the major cause of mitochondrial diseases. Cells harboring disease-related mtDNA mutations exhibit various phenotypic abnormalities, such as reduced respiration and elevated lactic acid production. Induced pluripotent stem cell (iPSC) lines derived from patients with mitochondrial disease, with high proportions of mutated mtDNA, exhibit defects in maturation into neurons or cardiomyocytes. In this study, we have discovered a small-molecule compound, which we name tryptolinamide (TLAM), that activates mitochondrial respiration in cybrids generated from patient-derived mitochondria and fibroblasts from patient-derived iPSCs. We found that TLAM inhibits phosphofructokinase-1 (PFK1), which in turn activates AMPK-mediated fatty-acid oxidation to promote oxidative phosphorylation, and redirects carbon flow from glycolysis toward the pentose phosphate pathway to reinforce anti-oxidative potential. Finally, we found that TLAM rescued the defect in neuronal differentiation of iPSCs carrying a high ratio of mutant mtDNA, suggesting that PFK1 represents a potential therapeutic target for mitochondrial diseases.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article and its Supplementary Information file. Source data are provided with this paper.
References
Schon, E. A., DiMauro, S. & Hirano, M. Human mitochondrial DNA: roles of inherited and somatic mutations. Nat. Rev. Genet. 13, 878–890 (2012).
Taylor, R. W. & Turnbull, D. M. Mitochondrial DNA mutations in human disease. Nat. Rev. Genet. 6, 389–402 (2005).
Goto, Y., Nonaka, I. & Horai, S. A mutation in the tRNALeu(UUR) gene associated with the MELAS subgroup of mitochondrial encephalomyopathies. Nature 348, 651–653 (1990).
Avula, S., Parikh, S., Demarest, S., Kurz, J. & Gropman, A. Treatment of mitochondrial disorders. Curr. Treat. Options Neurol. 16, 292 (2014).
Yun, J. & Finkel, T. Mitohormesis. Cell Metab. 19, 757–766 (2014).
Katayama, Y. et al. Accumulation of oxidative stress around the stroke-like lesions of MELAS patients. Mitochondrion 9, 306–313 (2009).
Ikawa, M. et al. Evaluation of systemic redox states in patients carrying the MELAS A3243G mutation in mitochondrial DNA. Eur. Neurol. 67, 232–237 (2012).
Whitehouse, S., Cooper, R. H. & Randle, P. J. Mechanism of activation of pyruvate dehydrogenase by dichloroacetate and other halogenated carboxylic acids. Biochem. J. 141, 761–774 (1974).
Kato, M., Li, J., Chuang, J. L. & Chuang, D. T. Distinct structural mechanisms for inhibition of pyruvate dehydrogenase kinase isoforms by AZD7545, dichloroacetate and radicicol. Structure 15, 992–1004 (2007).
Kaufmann, P. et al. Dichloroacetate causes toxic neuropathy in MELAS: a randomized, controlled clinical trial. Neurology 66, 324–330 (2006).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Chen, W. & Guéron, M. The inhibition of bovine heart hexokinase by 2-deoxy-d-glucose-6-phosphate: characterization by 31P NMR and metabolic implications. Biochimie 74, 867–873 (1992).
Chang, J. C. et al. Peptide-mediated delivery of donor mitochondria improves mitochondrial function and cell viability in human cybrid cells with the MELAS A3243G mutation. Sci. Rep. 7, 10710 (2017).
Seo, K. S. et al. KL1333, a Novel NAD+ modulator, improves energy metabolism and mitochondrial dysfunction in MELAS fibroblasts. Front. Neurol. 9, 552 (2018).
Ma, H. et al. Metabolic rescue in pluripotent cells from patients with mtDNA disease. Nature 524, 234–238 (2015).
Goto, Y. et al. Mitochondrial myopathy, encephalopathy, lactic acidosis and stroke-like episodes (MELAS): a correlative study of the clinical features and mitochondrial DNA mutation. Neurology 42, 545–550 (1992).
Koga, Y., Nonaka, I., Kobayashi, M., Tojyo, M. & Nihei, K. Findings in muscle in complex I (NADH coenzyme Q reductase) deficiency. Ann. Neurol. 24, 749–756 (1988).
Kirino, Y. et al. Codon-specific translational defect caused by a wobble modification deficiency in mutant tRNA from a human mitochondrial disease. Proc. Natl Acad. Sci. USA 101, 15070–15075 (2004).
Kirino, Y., Goto, Y., Campos, Y., Arenas, J. & Suzuki, T. Specific correlation between the wobble modification deficiency in mutant tRNAs and the clinical features of a human mitochondrial disease. Proc. Natl Acad. Sci. USA 102, 7127–7132 (2005).
Wong, A. et al. Differentiation-specific effects of LHON mutations introduced into neuronal NT2 cells. Hum. Mol. Genet. 11, 431–438 (2002).
Mattiazzi, M. et al. The mtDNA T8993G (NARP) mutation results in an impairment of oxidative phosphorylation that can be improved by antioxidants. Hum. Mol. Genet. 13, 869–879 (2004).
Wojewoda, M., Duszyński, J. & Szczepanowska, J. Antioxidant defence systems and generation of reactive oxygen species in osteosarcoma cells with defective mitochondria: effect of selenium. Biochim. Biophys. Acta 1797, 890–896 (2010).
Pang, C. Y., Lee, H. C. & Wei, Y. H. Enhanced oxidative damage in human cells harboring A3243G mutation of mitochondrial DNA: implication of oxidative stress in the pathogenesis of mitochondrial diabetes. Diabetes Res. Clin. Pract. 54, S45–S56 (2001).
Hardie, D. G. & Pan, D. A. Regulation of fatty acid synthesis and oxidation by the AMP-activated protein kinase. Biochem. Soc. Trans. 30, 1064–1070 (2002).
Ducommun, S. et al. Enhanced activation of cellular AMPK by dual-small molecule treatment: AICAR and A769662. Am. J. Physiol. Endocrinol. Metab. 306, E688–E696 (2014).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001).
Lopaschuk, G. D., Wall, S. R., Olley, P. M. & Davies, N. J. Etomoxir, a carnitine palmitoyltransferase I inhibitor, protects hearts from fatty acid-induced ischemic injury independent of changes in long chain acylcarnitine. Circ. Res. 63, 1036–1043 (1988).
Yamamoto, T. et al. Reduced methylation of PFKFB3 in cancer cells shunts glucose towards the pentose phosphate pathway. Nat. Commun. 5, 3480 (2014).
Moreno-Sánchez, R. et al. Phosphofructokinase type 1 kinetics, isoform expression and gene polymorphisms in cancer cells. J. Cell. Biochem. 113, 1692–1703 (2012).
Yokota, M., Hatakeyama, H., Ono, Y., Kanazawa, M. & Goto, Y. I. Mitochondrial respiratory dysfunction disturbs neuronal and cardiac lineage commitment of human iPSCs. Cell Death Dis. 8, e2551 (2017).
Zancan, P., Rosas, A. O., Marcondes, M. C., Marinho-Carvalho, M. M. & Sola-Penna, M. Clotrimazole inhibits and modulates heterologous association of the key glycolytic enzyme 6-phosphofructo-1-kinase. Biochem. Pharmacol. 73, 1520–1527 (2007).
Marcondes, M. C., Sola-Penna, M. & Zancan, P. Clotrimazole potentiates the inhibitory effects of ATP on the key glycolytic enzyme 6-phosphofructo-1-kinase. Arch. Biochem. Biophys. 497, 62–67 (2010).
Kim, S. G., Manes, N. P., El-Maghrabi, M. R. & Lee, Y. H. Crystal structure of the hypoxia-inducible form of 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase (PFKFB3): a possible new target for cancer therapy. J. Biol. Chem. 281, 2939–2944 (2006).
Webb, B. A. et al. Structures of human phosphofructokinase-1 and atomic basis of cancer-associated mutations. Nature 523, 111–114 (2015).
Mor, I., Cheung, E. C. & Vousden, K. H. Control of glycolysis through regulation of PFK1: old friends and recent additions. Cold Spring Harb. Symp. Quant. Biol. 76, 211–216 (2011).
Wang, M. D. et al. Acetyl-coenzyme A carboxylase alpha promotion of glucose-mediated fatty acid synthesis enhances survival of hepatocellular carcinoma in mice and patients. Hepatology 63, 1272–1286 (2016).
Tufi, R. et al. Enhancing nucleotide metabolism protects against mitochondrial dysfunction and neurodegeneration in a PINK1 model of Parkinson’s disease. Nat. Cell Biol. 16, 157–166 (2014).
Yi, W. et al. Phosphofructokinase 1 glycosylation regulates cell growth and metabolism. Science 337, 975–980 (2012).
Kim, N. H. et al. Snail reprograms glucose metabolism by repressing phosphofructokinase PFKP allowing cancer cell survival under metabolic stress. Nat. Commun. 8, 14374 (2017).
Miyazawa, H. et al. Rewiring of embryonic glucose metabolism via suppression of PFK-1 and aldolase during mouse chorioallantoic branching. Development 144, 63–73 (2017).
Naito, Y., Hino, K., Bono, H. & Ui-Tei, K. CRISPRdirect: software for designing CRISPR/Cas guide RNA with reduced off-target sites. Bioinformatics 31, 1120–1123 (2015).
Okita, K. et al. A more efficient method to generate integration-free human iPS cells. Nat. Methods 8, 409–412 (2011).
Hatakeyama, H., Katayama, A., Komaki, H., Nishino, I. & Goto, Y. Molecular pathomechanisms and cell-type-specific disease phenotypes of MELAS caused by mutant mitochondrial tRNATrp. Acta Neuropathol. Commun. 3, 52 (2015).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Pool, M., Thiemann, J., Bar-Or, A. & Fournier, A. E. NeuriteTracer: a novel ImageJ plugin for automated quantification of neurite outgrowth. J. Neurosci. Methods 168, 134–139 (2008).
Arganda-Carreras, I., Fernández-González, R., Muñoz-Barrutia, A. & Ortiz-De-Solorzano, C. 3D reconstruction of histological sections: application to mammary gland tissue. Microsc. Res. Tech. 73, 1019–1029 (2010).
King, M. P. & Attardi, G. Human cells lacking mtDNA: repopulation with exogenous mitochondria by complementation. Science 246, 500–503 (1989).
Mimaki, M. et al. Reversible infantile respiratory chain deficiency: a clinical and molecular study. Ann. Neurol. 68, 845–854 (2010).
Acknowledgements
We are grateful to the RIKEN CBS RRD for DNA sequencing analysis. We thank K. Ohgane, G. Hirai, K. Dodo and M. Sodeoka (RIKEN, Japan) for confirmation of the structure and purity of TLAM. We also thank Y. Ono, S. Okabe and M. Kanazawa (NCNP, Japan) for their assistance. This work was supported in part by the RIKEN Aging Project (to M.S. and M. Yoshida); a Grant-in-Aid for Scientific Research (S) (JP19H05640; M. Yoshida) and Grants-in-Aid for Young Scientist (JP16K18432 and JP19K16728; H.K.) from the Japan Society for the Promotion of Science (JSPS); a Grant-in-Aid for Scientific Research on Innovative Areas (JP18H05503; M. Yoshida) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT); Grants-in-Aid of the Research on Intractable Diseases (Mitochondrial Disorder; Y.G.) from the Ministry of Health, Labour and Welfare of Japan (MHLW); and the Intramural Research Grant for Neurological and Psychiatric Disorders of NCNP (24-8, 27-6 and 30-7; Y.G.). This work was inspired by the JSPS Asian Chemical Biology Initiative.
Author information
Authors and Affiliations
Contributions
H.K. designed and carried out the experiments (with technical assistance from H.N.) and drafted the manuscript. H.H. and M. Yokota established and characterized patient-derived iPSCs and performed the differentiation experiments. S.S. prepared samples for the karyotyping. H.H. helped write the manuscript. Y.T. and M.S. prepared protein samples for the ITC experiments. H.O. provided NPDepo compounds. M.M. generated cybrids. M. Yoshida and Y.G. coordinated the study and helped draft the manuscript. All authors gave final approval for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Screening for compounds that attenuate the toxicity of 2-deoxyglucose (2DG).
a, Effect of dichloroacetate (DCA), a positive control for screening, on cell number in HeLa cells incubated with 10 mM 2DG for 5 days. Data represent means + s.d. of three independent experiments. b, Results of a screen of 17,568 compounds from the NPDepo library. Lower panel represents raw values of high control, low control, and Z’ factor of each assay plate. c, Effect of TLAM on cell number in BxPC3 cells cultured for 5 days in the presence of 3 mM 2DG. Data represent means + s.d. of three independent experiments. d, Chemical structures of compounds tested in the SAR study. e, Effects of compounds in d on cell number in HeLa cells incubated with 10 mM 2DG for 5 days. Data represent means + s.d. of three independent experiments. f, Change in OCR (upper) and ECAR (lower) in HeLa cells after treatment with compound 2–6. Data represent means ± s.d. of three independent experiments. P values were calculated using the Holm comparison test (two-tailed) for a, c, and e. ***, P < 1.0 × 10−5.
Extended Data Fig. 2 TLAM activates mitochondrial function in cells without remarkable toxicity.
a, Mitochondrial functions in fibro-3243[0] and fibro-3243[100] in the absence of TLAM. b, Effect of TLAM (60 µM) on mitochondrial functions in fibro-3243[0] and fibro-3243[100]. Values represent fold increase relative to DMSO-treated fibro-3243[0]. c, d, Effect of TLAM on mitochondrial function in cells treated with rotenone (RTN). c, OCR in HeLa cells pretreated with 10 nM RTN in response to DMSO / 60 µM TLAM, 0.3 µM OMA, 0.25 µM FCCP, 1 µM RTN, and 1 µM AMA. d, Effect of TLAM on mitochondrial functions in RTN-pretreated HeLa cells. e, Effect of TLAM (60 µM) on mitochondrial ROS in 143B and cybrid[97]. f, ROS levels in 143B or cybrid[97], treated with vehicle, TLAM (60 µM), or tBHP (25–100 µM). g, Effect of TLAM on cell proliferation in 143B and cybrid[97]. 143B and cybrid[97] cells were treated with DMSO or TLAM, and then imaged over a period of 4 days. Confluence (left) and doubling time (right) were calculated. Throughout, data represent means + (±) s.d. of three independent experiments. P values were calculated using Student’s two-tailed t-test for a and d; Welch’s two-tailed t-test for b; the Holm comparison test (two-tailed) for e and f; and two-way ANOVA with Tukey’s comparison test for g. ***, P < 1.0 × 10-5.
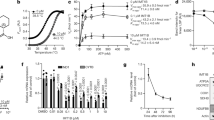
Extended Data Fig. 3 TLAM induces AMPK activation.
a–c, Effect of TLAM on levels of PDH phosphorylation (a), oxidative phosphorylation (OXPHOS) proteins (b), and ACC phosphorylation (c). HeLa cells were treated with DCA, TLAM, or AMPK activators (2 mM ACAR and 30 µM A769662) for 24 h (PDH phosphorylation and OXPHOS proteins) or 1 h (ACC phosphorylation), and then subjected to western blotting with the indicated antibodies. d, Effect of compound C (10 µM) on the levels of phosphorylation of ACC in HeLa cells treated with TLAM or AMPK activators. Throughout, data are from single experiments that are representative of at least three experiments.
Extended Data Fig. 4 TLAM modulates cellular metabolism.
a, Effect of TLAM on lactate secretion in HeLa cells. Data represent means + s.d. of three independent experiments. b, Design of the pilot metabolomic analysis. c, d, Metabolomic analysis of TLAM-treated HeLa and BxPC3 cells. Data show amounts of metabolites of G6P, F6P, and F1,6-BP (c), and intracellular/extracellular lactate (d). The list of metabolites measured in the experiment is available in Supplementary Table 1. Data are from a single experiment (c and d). P values were calculated using the Holm comparison test (two-tailed) for a.
Extended Data Fig. 5 TLAM redirects carbon flow from glycolysis to the pentose phosphate pathway.
a, Stacked bar charts of amounts of metabolites in the tracer experiments using [1,2-13C]glucose, related to Fig. 4. Data represent means of technical triplicates from a single experiment. b, ECAR in HeLa cells in response to glucose addition in the presence of TLAM. Data represent means ± s.d. of three independent experiments. c, Metabolites in cells treated with TLAM in response to [U-13C]glucose stimulation. The design of the experiment is available in Supplementary Fig. 2b. Data show amounts of each isotopologue of metabolites (means of technical triplicates from a single experiment). The list of metabolites measured in the experiment, including results of statistical tests, is available in Supplementary Table 3. d, AMP/ATP ratio, calculated using the data from the 13C-labeled metabolomic analysis (Supplementary Table 3). Data represent means + s.d. of technical triplicate from a single experiment. e, Total levels of 13C-labeled glycolytic metabolites (G6P, F6P, F1,6-BP, GAP, DHAP, 3-PG, and PEP) and PPP metabolites (G6P, 6-PG, Ru-5-P, and PRPP) in each experimental condition (steady-state: from Fig. 4 and Supplementary Table 2; glucose stimulation: from Extended Data Fig. 5c and Supplementary Table 3). Data represent means + s.d. of technical triplicate from a single experiment. f, Effect of TLAM on 2DG uptake. Incorporation of 2DG was measured by luminescence with Glucose Uptake-Glo (Promega) under the same experimental conditions as the metabolomic studies, using 2DG in place of 13C-glucose (steady-state, left; glucose stimulation, right). Data represent means + (±) s.d. of three independent experiments. P values were calculated using Welch’s two-tailed t-test for d, e, and f (right plot); and repeated measures two-way ANOVA for f (left plot).
Extended Data Fig. 6 TLAM physically interacts with PFKP.
A solution of PFKP (39 µM) was titrated with a solution of TLAM (400 µM). Data are from a single experiment that was representative of three determinations using the same protein preparation. The Kd value is the mean ± s.d. of those three trials.
Extended Data Fig. 7 Sensitivities to TLAM in PFK1-deficient cells.
a, Effects of 60 µM TLAM on OCR/ECAR ratio (upper) and ECAR in response to 1 mM glucose (lower) in PFK1-deficient #2. Data represent means ± s.d. of four (upper) or three (lower) independent experiments. b–d, Effect of TLAM on cell number in the presence or absence of 3 mM 2DG for 5 days in wild-type HeLa, PFK1-deficient #1, and PFK1-deficient #2 cells (b); in vector- or PFKP-reintroduced PFK1-deficient #1 cells (c); and in vector- or PFKP-reintroduced PFK1-deficient #2 cells (d). Data represent means + s.d. of three independent experiments. P values were calculated using Holm’s comparison test (two-tailed). ***, P < 1.0 × 10-5.
Extended Data Fig. 8 PFK1 regulates cellular AMPK activity and the PPP.
a, b, Effect of PFK1 expression on phosphorylated ACC. Protein levels of phosphorylated ACC and total ACC were detected in wild-type HeLa, PFK1-deficient #1, and PFK1-deficient #2 cells (a), or in vector- or PFKP-expressing PFK1-deficient #1 and #2 cells (b). Lower panels show densitometric data representing means + s.d. of three independent experiments. c, d, Effect of TLAM on cellular NADPH level (c) and activity of caspase-3/7 (d) in the presence of hydrogen peroxide (H2O2). c, HeLa cells were pretreated with DMSO or 50 µM TLAM for 30 min, exposed to 250 µM H2O2 for 90 min, and then subjected to NADPH assay. Data represent means + s.d. of three independent experiments. d, HeLa cells were pretreated with DMSO, TLAM, or zVAD-fmk (zVAD, a control for inhibition of caspase-3/7 activity), and then exposed to H2O2. The cells were further treated with active caspase-3/7 detection reagent and incubated. After 15 h of incubation, the cells were imaged (left) and quantified for the activity of caspase-3/7 (right). Green signals in the images represent caspase-3/7 activity. Scale bar, 300 µm. Data represent means + s.d. of three independent experiments. e, Involvement of the PPP in TLAM-induced respiratory activation. Protein levels of G6PD in HeLa cells transfected with siRNA against G6PD (upper). OCR in G6PD-silenced HeLa cells treated with TLAM for 80 min (lower). Data represent means + s.d. of four independent experiments. f, Effects of TLAM and N-acetyl-L-cysteine (NAC) on OCR in the presence of diamide. HeLa cells were pretreated with vehicle, TLAM (60 µM), or NAC (0.3 mM), and exposed to 1 mM diamide. After 7 min, OCR was analyzed. Data represent means + s.d. of three independent experiments. P values were calculated using the Holm comparison test (two-tailed) for a and c; Welch’s two-tailed t-test for b; and two-way ANOVA with Tukey’s comparison test for d, e, and f. ***, P < 1.0 × 10-5. Western blots are from single experiments that are representative of three independent experiments.
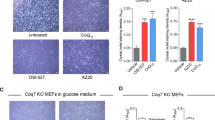
Extended Data Fig. 9 Effects of clotrimazole on glycolytic flux, ATP content, and the neuronal maturation defect of iPSCs carrying mutated mtDNA.
a, b, Effects of 10 µM clotrimazole on glycolytic flux and ATP content in 143B cells and cybrid[97]. a, 143B cells and cybrid[97] were treated with DMSO, TLAM, or clotrimazole. After 14 min, ECAR was analyzed. Data represent means + s.d. of three independent experiments. b, 143B cells and cybrid[97] were treated with DMSO, TLAM, clotrimazole, or 2DG. After 1 h of incubation, ATP content was analyzed. Data represent means + s.d. of three independent experiments. c, d, Effect of 10 µM clotrimazole on the neuronal maturation defect of iPSCs carrying mutated mtDNA. After neural stem cell specification of iPSC-3243[100] (c) and iPSC-3291[100] (d), clotrimazole was continuously administered during terminal differentiation into neuron-like cells. The cells were fixed, stained with anti–NF-H antibody (green) and Hoechst 33342 (blue), imaged (left), and quantified (right). Data represent means of two experimental replicates. Scale bar, 100 µm. P values were calculated using one-way ANOVA with Tukey’s multiple comparison test for a and the Holm comparison test (two-tailed) for b.
Extended Data Fig. 10 Schematic model of cellular metabolism altered by TLAM.
Left: in the absence of TLAM, PFK1 can promote glycolytic flux (corresponding to ECAR). Right: tryptolinamide (TLAM) inhibits PFK1, thereby decreasing glycolytic flux and redirecting G6P/F6P toward the pentose phosphate pathway (PPP). The reduction in glycolytic flux induces activation of AMPK, which phosphorylates ACC, which in turn promotes fatty-acid oxidation (FAO). In addition, the redirection enhances the PPP, which generates NADPH for anti-oxidant defense and increases OXPHOS (corresponding to OCR). The redirected flow can converge into the payoff phase of glycolysis through the non-oxidative PPP.
Supplementary information
Supplementary Information
Supplementary Figs. 1–11 and Tables 1–5.
Source data
Source Data Fig. 1
Statistical source data for Fig.1b,c.
Source Data Fig. 2
Statistical source data for Fig.2b–f.
Source Data Fig. 3
Statistical source data for Fig. 3a,b,d.
Source Data Fig. 3
Uncropped western blot images for Fig. 3c.
Source Data Fig. 4
Statistical source data for Fig. 4.
Source Data Fig. 5
Statistical source data for Fig. 5a,c,f.
Source Data Fig. 6
Statistical source data for Fig. 6a,b.
Source Data Extended Data Fig. 1
Statistical source data for Extended Data Fig. 1a,c,e.
Source Data Extended Data Fig. 2
Statistical source data for Extended Data Fig. 2a,b,d–g.
Source Data Extended Data Fig. 3
Uncropped western blot images for Extended Data Fig. 3a–d.
Source Data Extended Data Fig. 4
Statistical source data for Extended Data Fig. 4a.
Source Data Extended Data Fig. 5
Statistical source data for Extended Data Fig. 5d–f.
Source Data Extended Data Fig. 7
Statistical source data for Extended Data Fig. 7b–d.
Source Data Extended Data Fig. 8
Statistical source data for Extended Data Fig. 8a–f.
Source Data Extended Data Fig. 8
Uncropped western blot images for Extended Data Fig. 8a,b,e.
Source Data Extended Data Fig. 9
Statistical source data for Extended Data Fig. 9a,b.
Source Data Supplementary Fig. 3
Uncropped western blot images for Supplementary Fig. 3b,d.
Source Data Supplementary Fig. 6
Uncropped western blot images for Supplementary Fig. 6.
Source Data Supplementary Fig. 7
Uncropped gel images for Supplementary Fig. 7a–c.
Source Data Supplementary Table 2
Statistical source data for Supplementary Table 2.
Source Data Supplementary Table 3
Statistical source data for Supplementary Table 3.
Rights and permissions
About this article
Cite this article
Kobayashi, H., Hatakeyama, H., Nishimura, H. et al. Chemical reversal of abnormalities in cells carrying mitochondrial DNA mutations. Nat Chem Biol 17, 335–343 (2021). https://doi.org/10.1038/s41589-020-00676-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-020-00676-4
This article is cited by
-
Induced pluripotent stem cells: ex vivo models for human diseases due to mitochondrial DNA mutations
Journal of Biomedical Science (2023)
-
Deep exome sequencing identifies enrichment of deleterious mosaic variants in neurodevelopmental disorder genes and mitochondrial tRNA regions in bipolar disorder
Molecular Psychiatry (2023)