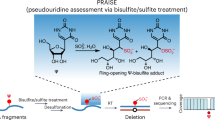
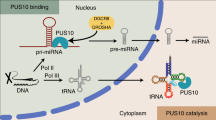
Abstract
Pseudouridine (Ψ) is a post-transcriptional RNA modification that alters RNA–RNA and RNA–protein interactions that affect gene expression. Messenger RNA pseudouridylation was recently discovered as a widespread and conserved phenomenon, but the mechanisms responsible for selective, regulated pseudouridylation of specific sequences within mRNAs were unknown. Here, we have revealed mRNA targets for five pseudouridine synthases and probed the determinants of mRNA target recognition by the predominant mRNA pseudouridylating enzyme, Pus1, by developing high-throughput kinetic analysis of pseudouridylation in vitro. Combining computational prediction and rational mutational analysis revealed an RNA structural motif that is both necessary and sufficient for mRNA pseudouridylation. Applying this structural context information predicted hundreds of additional mRNA targets that were pseudouridylated in vivo. These results demonstrate a structure-dependent mode of mRNA target recognition by a conserved pseudouridine synthase and implicate modulation of RNA structure as the probable mechanism to regulate mRNA pseudouridylation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Yeast strains and plasmids are available upon request. All sequencing data and oligonucleotide pool sequences have been deposited in GEO, accession GSE99487.
Code availability
Custom Bash and Python codes used for analysis are available on request.
References
Boccaletto, P. et al. MODOMICS: a database of RNA modification pathways. Nucleic Acids Res. 46, D303–D307 (2018).
Gilbert, W. V., Bell, T. A. & Schaening, C. Messenger RNA modifications: form, distribution, and function. Science 352, 1408–1412 (2016).
Carlile, T. M. et al. Pseudouridine profiling reveals regulated mRNA pseudouridylation in yeast and human cells. Nature 515, 143–146 (2014).
Schwartz, S. et al. Transcriptome-wide mapping reveals widespread dynamic-regulated pseudouridylation of ncRNA and mRNA. Cell 159, 148–162 (2014).
Lovejoy, A. F., Riordan, D. P. & Brown, P. O. Transcriptome-wide mapping of pseudouridines: pseudouridine synthases modify specific mRNAs in S. cerevisiae. PLoS ONE 9, e110799 (2014).
Li, X. et al. Chemical pulldown reveals dynamic pseudouridylation of the mammalian transcriptome. Nat. Chem. Biol. 11, 592–597 (2015).
Safra, M., Nir, R., Farouq, D., Slutzkin, I. V. & Schwartz, S. TRUB1 is the predominant pseudouridine synthase acting on mammalian mRNA via a predictable and conserved code. Genome Res. 27, 393–406 (2017).
Hamma, T. & Ferre-D’Amare, A. R. Pseudouridine synthases. Chem. Biol. 13, 1125–1135 (2006).
Motorin, Y. et al. The yeast tRNA: pseudouridine synthase Pus1p displays a multisite substrate specificity. RNA 4, 856–869 (1998).
Behm-Ansmant, I. et al. A previously unidentified activity of yeast and mouse RNA: pseudouridine synthases 1 (Pus1p) on tRNAs. RNA 12, 1583–1593 (2006).
Massenet, S. et al. Pseudouridine mapping in the Saccharomyces cerevisiae spliceosomal U small nuclear RNAs (snRNAs) reveals that pseudouridine synthase Pus1p exhibits a dual substrate specificity for U2 snRNA and tRNA. Mol. Cell Biol. 19, 2142–2154 (1999).
Basak, A. & Query, C. C. A pseudouridine residue in the spliceosome core is part of the filamentous growth program in yeast. Cell Rep. 8, P966–P973 (2014).
Bakin, A. & Ofengand, J. Four newly located pseudouridylate residues in Escherichia coli 23S ribosomal RNA are all at the peptidyltransferase center: analysis by the application of a new sequencing technique. Biochemistry 32, 9754–9762 (1993).
Carlile, T. M., Rojas-Duran, M. F. & Gilbert, W. V. Pseudo-seq: genome-wide detection of pseudouridine modifications in RNA. Methods Enzymol. 560, 219–245 (2015).
Lorenz, R. et al. ViennaRNA package 2.0. Algorithms Mol. Biol. 6, 26 (2011).
Kertesz, M. et al. Genome-wide measurement of RNA secondary structure in yeast. Nature 467, 103–107 (2010).
Taoka, M. et al. The complete chemical structure of Saccharomyces cerevisiae rRNA: partial pseudouridylation of U2345 in 25S rRNA by snoRNA snR9. Nucleic Acids Res. 44, 8951–8961 (2016).
Xia, T. et al. Thermodynamic parameters for an expanded nearest-neighbor model for formation of RNA duplexes with Watson–Crick base pairs. Biochemistry 37, 14719–14735 (1998).
Czudnochowski, N., Wang, A. L., Finer-Moore, J. & Stroud, R. M. In human pseudouridine synthase 1 (hPus1), a C-terminal helical insert blocks tRNA from binding in the same orientation as in the Pus1 bacterial homologue TruA, consistent with their different target selectivities. J. Mol. Biol. 425, 3875–3887 (2013).
Kwok, C. K., Tang, Y., Assmann, S. M. & Bevilacqua, P. C. The RNA structurome: transcriptome-wide structure probing with next-generation sequencing. Trends Biochem. Sci. 40, 221–232 (2015).
Breker, M., Gymrek, M., Moldavski, O. & Schuldiner, M. LoQAtE—Localization and Quantitation ATlas of the yeast proteomE. A new tool for multiparametric dissection of single-protein behavior in response to biological perturbations in yeast. Nucleic Acids Res. 42, D726–D730 (2014).
Urban, A., Behm-Ansmant, I., Branlant, C., Motorin, Y. & Motorlin, Y. RNA sequence and two-dimensional structure features required for efficient substrate modification by the Saccharomyces cerevisiae RNA:Ψ-synthase Pus7p. J. Biol. Chem. 284, 5845–5858 (2009).
Winzeler, E. A. et al. Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285, 901–906 (1999).
Jiang, H. Q., Motorin, Y., Jin, Y. X. & Grosjean, H. Pleiotropic effects of intron removal on base modification pattern of yeast tRNA(Phe): an in vitro study. Nucleic Acids Res. 25, 2694–2701 (1997).
Byun, Y. & Han, K. PseudoViewer3: generating planar drawings of large-scale RNA structures with pseudoknots. Bioinformatics 25, 1435–1437 (2009).
Kerpedjiev, P., Höner Zu Siederdissen, C. & Hofacker, I. L. Predicting RNA 3D structure using a coarse-grain helix-centered model. RNA 21, 1110–1121 (2015).
Carlile, T. M., Rojas-Duran, M. F. & Gilbert, W. V. Transcriptome-wide identification of pseudouridine modifications using Pseudo-seq. Curr. Protoc. Mol. Biol. 112, 4.25.1–4.25.24 (2015).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620 (2014).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Crooks, G. E., Hon, G., Chandonia, J. M. & Brenner, S. E. WebLogo: a sequence logo generator. Genome Res. 14, 1188–1190 (2004).
Waterhouse, A. et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Acknowledgements
We thank Y. Motorin (Université de Lorraine) for the yeast Pus1 expression plasmid and R. Stroud (University of California, San Francisco) for human Pus1 expression plasmids. We also thank G. Mawla and C. Mason for technical assistance and members of the Gilbert lab for critical reading of the manuscript. This work was supported by the National Institutes of Health (grant no. GM101316) (W.V.G.), American Cancer Society (grant no. RSG-13-396-01-RMC) (W.V.G.), American Cancer Society postdoctoral fellowship (T.M.C.), Jane Coffin Childs postdoctoral fellowship (N.M.M.), NSF graduate research fellowship (C.S.) and National Institutes of Health predoctoral training grant (no. T32GM007287) (T.A.B.).
Author information
Authors and Affiliations
Contributions
T.M.C., B.Z., T.A.B. and W.V.G. conceived and designed the experiments. T.M.C., N.M.M., A.S., T.A.B. and W.V.G. performed the experiments. T.M.C., N.M.M. and C.S. performed the bioinformatic analyses. T.M.C. and W.V.G. interpreted the results and wrote the paper with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–2 and Supplementary Figures 1–7
Supplementary Dataset 1
Pseduo-seq signal for S. cerevisiae RNA pools treated with S100 extracts.
Supplementary Dataset 2
Pseudo-seq signal for H. sapiens RNA pools pseudouridylated with recombinant human PUS proteins.
Supplementary Dataset 3
Structural characteristics of S. cerevisiae Pus1 substrates.
Supplementary Dataset 4
Kinetic analysis of pseudouridylation of wild-type and mutant sequences by recombinant Pus1.
Supplementary Dataset 5
Kinetic analysis of pseudouridylation of wild-type and stem extension mutant sequences by recombinant Pus1.
Supplementary Dataset 6
Random forest classifier predicted Pus1 mRNA Ψs.
Supplementary Dataset 7
Summary of libraries contained in this study.
Rights and permissions
About this article
Cite this article
Carlile, T.M., Martinez, N.M., Schaening, C. et al. mRNA structure determines modification by pseudouridine synthase 1. Nat Chem Biol 15, 966–974 (2019). https://doi.org/10.1038/s41589-019-0353-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-019-0353-z
This article is cited by
-
RNA modifications in cancer
British Journal of Cancer (2023)
-
Semi-quantitative detection of pseudouridine modifications and type I/II hypermodifications in human mRNAs using direct long-read sequencing
Nature Communications (2023)
-
RNA modification in cardiovascular disease: implications for therapeutic interventions
Signal Transduction and Targeted Therapy (2023)
-
A selective and atom-economic rearrangement of uridine by cascade biocatalysis for production of pseudouridine
Nature Communications (2023)
-
The importance of pseudouridylation: human disorders related to the fifth nucleoside
Biologia Futura (2023)