Abstract
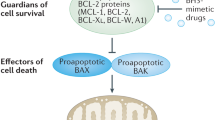
BCL-2 family protein interactions regulate apoptosis, a critical process that maintains tissue homeostasis but can cause a host of human diseases when deregulated. Venetoclax is the first FDA-approved drug to reactivate apoptosis in cancer by selectively targeting an anti-apoptotic BCL-2 family member. The drug’s activity relies on an ‘inhibit the inhibitor’ mechanism, whereby blockade of a key surface groove on BCL-2 disables its capacity to neutralize pro-apoptotic effectors, such as BAX, a chief executioner protein of the apoptotic pathway. A series of physiologic and pharmacologic regulatory sites that mediate the activation or inhibition of BAX have recently been identified, providing blueprints for the development of alternative apoptosis modulators to block pathologic cell survival or avert unwanted cell death by drugging BAX directly.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Souers, A. J. et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat. Med. 19, 202–208 (2013).
Muchmore, S. W. et al. X-ray and NMR structure of human Bcl-xL, an inhibitor of programmed cell death. Nature 381, 335–341 (1996).
Sattler, M. et al. Structure of Bcl-xL-Bak peptide complex: recognition between regulators of apoptosis. Science 275, 983–986 (1997). This paper was the first to reveal the structure of the canonical BH3-in-groove interaction that both established the paradigm for heterodimerization between pro- and anti-apoptotic BCL-2 family members and provided a blueprint for developing anti-apoptotic inhibitors to reactivate apoptosis in cancer.
Green, D. R. & Kroemer, G. The pathophysiology of mitochondrial cell death. Science 305, 626–629 (2004).
Korsmeyer, S. J., Shutter, J. R., Veis, D. J., Merry, D. E. & Oltvai, Z. N. Bcl-2/Bax: a rheostat that regulates an anti-oxidant pathway and cell death. Semin. Cancer Biol. 4, 327–332 (1993).
Wei, M. C. et al. Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292, 727–730 (2001).
Wang, K., Gross, A., Waksman, G. & Korsmeyer, S. J. Mutagenesis of the BH3 domain of BAX identifies residues critical for dimerization and killing. Mol. Cell. Biol. 18, 6083–6089 (1998).
Chen, L. et al. Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol. Cell 17, 393–403 (2005).
Willis, S. N. et al. Apoptosis initiated when BH3 ligands engage multiple Bcl-2 homologs, not Bax or Bak. Science 315, 856–859 (2007).
Suzuki, M., Youle, R. J. & Tjandra, N. Structure of Bax: coregulation of dimer formation and intracellular localization. Cell 103, 645–654 (2000). This paper reported the first high-resolution structure of pro-apoptotic BAX, revealing the conformational organization of key functional regions implicated in the BAX activation pathway.
Wang, K., Yin, X. M., Chao, D. T., Milliman, C. L. & Korsmeyer, S. J. BID: a novel BH3 domain-only death agonist. Genes Dev. 10, 2859–2869 (1996).
Cheng, E. H. et al. BCL-2, BCL-XL sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol. Cell 8, 705–711 (2001).
Letai, A. et al. Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics. Cancer Cell 2, 183–192 (2002).
Wei, M. C. et al. tBID, a membrane-targeted death ligand, oligomerizes BAK to release cytochrome c. Genes Dev. 14, 2060–2071 (2000).
Walensky, L. D. et al. Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science 305, 1466–1470 (2004).
Walensky, L. D. et al. A stapled BID BH3 helix directly binds and activates BAX. Mol. Cell 24, 199–210 (2006).
Gavathiotis, E. et al. BAX activation is initiated at a novel interaction site. Nature 455, 1076–1081 (2008). This paper applied a stapled BIM BH3 helix to uncover the location of a trigger site at the N-terminal surface (α1/α6) of BAX that mediates its direct activation by select BH3-only proteins.
Czabotar, P. E., Colman, P. M. & Huang, D. C. Bax activation by Bim? Cell Death Differ. 16, 1187–1191 (2009).
Brouwer, J. M. et al. Conversion of Bim-BH3 from activator to inhibitor of Bak through structure-based design. Mol. Cell 68, 659–672.e659 (2017).
Czabotar, P. E. et al. Bax crystal structures reveal how BH3 domains activate Bax and nucleate its oligomerization to induce apoptosis. Cell 152, 519–531 (2013). This paper reported crystal structures of activating BH3 interactions at the canonical groove of a BAX construct lacking its C-terminal helix and a symmetric homodimer of a BAX truncate (α2–α5) believed to represent a nucleating unit for BAX homo-oligomerization.
Dengler, M. A. et al. BAX activation: mutations near its proposed non-canonical BH3 binding site reveal allosteric changes controlling mitochondrial association. Cell Rep. 27, 359–373.e356 (2019).
Mérino, D. et al. The role of BH3-only protein Bim extends beyond inhibiting Bcl-2-like prosurvival proteins. J. Cell Biol. 186, 355–362 (2009).
Llambi, F. et al. A unified model of mammalian BCL-2 protein family interactions at the mitochondria. Mol. Cell 44, 517–531 (2011).
Gavathiotis, E., Reyna, D. E., Davis, M. L., Bird, G. H. & Walensky, L. D. BH3-triggered structural reorganization drives the activation of proapoptotic BAX. Mol. Cell 40, 481–492 (2010).
Garner, T. P. et al. An autoinhibited dimeric Form of BAX regulates the BAX activation pathway. Mol. Cell 64, 431 (2016).
Reyna, D. E. et al. Direct activation of BAX by BTSA1 overcomes apoptosis resistance in acute myeloid leukemia. Cancer Cell 32, 490–505.e410 (2017). Building on the identification of the first direct and selective BAX activator molecule (ref. 27), this paper advanced a more potent analog from mechanistic validation to in vivo proof of concept, demonstrating that direct pharmacologic activation of BAX could effectively treat a mouse model of human acute myeloid leukemia.
Gavathiotis, E., Reyna, D. E., Bellairs, J. A., Leshchiner, E. S. & Walensky, L. D. Direct and selective small-molecule activation of proapoptotic BAX. Nat. Chem. Biol. 8, 639–645 (2012).
Mason, K. D. et al. Programmed anuclear cell death delimits platelet life span. Cell 128, 1173–1186 (2007).
Certo, M. et al. Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell 9, 351–365 (2006).
Davids, M. S. et al. Comprehensive safety analysis of venetoclax monotherapy for patients with relapsed/refractory chronic lymphocytic leukemia. Clin. Cancer Res. 24, 4371–4379 (2018).
Oltersdorf, T. et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature 435, 677–681 (2005).
Shuker, S. B., Hajduk, P. J., Meadows, R. P. & Fesik, S. W. Discovering high-affinity ligands for proteins: SAR by NMR. Science 274, 1531–1534 (1996).
Pritz, J. R. et al. Allosteric sensitization of proapoptotic BAX. Nat. Chem. Biol. 13, 961–967 (2017).
Arnoult, D. et al. Cytomegalovirus cell death suppressor vMIA blocks Bax- but not Bak-mediated apoptosis by binding and sequestering Bax at mitochondria. Proc. Natl Acad. Sci. USA 101, 7988–7993 (2004).
Brahmbhatt, H., Uehling, D., Al-Awar, R., Leber, B. & Andrews, D. Small molecules reveal an alternative mechanism of Bax activation. Biochem. J. 473, 1073–1083 (2016).
Kale, J. et al. Phosphorylation switches Bax from promoting to inhibiting apoptosis thereby increasing drug resistance. EMBO Rep. 19, e45235 (2018).
Xin, M. et al. Small-molecule Bax agonists for cancer therapy. Nat. Commun. 5, 4935 (2014).
Gahl, R. F., He, Y., Yu, S. & Tjandra, N. Conformational rearrangements in the pro-apoptotic protein, Bax, as it inserts into mitochondria: a cellular death switch. J. Biol. Chem. 289, 32871–32882 (2014).
Edlich, F. et al. Bcl-xL retrotranslocates Bax from the mitochondria into the cytosol. Cell 145, 104–116 (2011).
Dai, H. et al. Transient binding of an activator BH3 domain to the Bak BH3-binding groove initiates Bak oligomerization. J. Cell Biol. 194, 39–48 (2011).
Leshchiner, E. S., Braun, C. R., Bird, G. H. & Walensky, L. D. Direct activation of full-length proapoptotic BAK. Proc. Natl Acad. Sci. USA 110, E986–E995 (2013).
Moldoveanu, T. et al. BID-induced structural changes in BAK promote apoptosis. Nat. Struct. Mol. Biol. 20, 589–597 (2013). This paper reported a solution structure of the activating interaction between a stapled BID BH3 helix and the canonical groove of BAK lacking its C-terminal helix, informing the ‘hit-and-run’ mechanism for direct BAK activation.
Edwards, A. L. et al. Multimodal interaction with BCL-2 family proteins underlies the proapoptotic activity of PUMA BH3. Chem. Biol. 20, 888–902 (2013).
Kotschy, A. et al. The MCL1 inhibitor S63845 is tolerable and effective in diverse cancer models. Nature 538, 477–482 (2016).
Cuconati, A. & White, E. Viral homologs of BCL-2: role of apoptosis in the regulation of virus infection. Genes Dev. 16, 2465–2478 (2002).
Ma, J. et al. Structural mechanism of Bax inhibition by cytomegalovirus protein vMIA. Proc. Natl Acad. Sci. USA 109, 20901–20906 (2012). This paper revealed a physiologic mechanism by which a viral protein can directly target BAX to suppress its conformational activation.
Barclay, L. A. et al. Inhibition of pro-apoptotic BAX by a noncanonical interaction mechanism. Mol. Cell 57, 873–886 (2015).
Niu, X. et al. A small-molecule inhibitor of Bax and Bak oligomerization prevents genotoxic cell death and promotes neuroprotection. Cell Chem. Biol. 24, 493–506.e495 (2017).
Garner, T. P. et al. Small-molecule allosteric inhibitors of BAX. Nat. Chem. Biol. 15, 322–330 (2019). This paper identified molecular inhibitors of BAX that operate by targeting a surface pocket and imposing allosteric restraint on key regions implicated in the conformational activation of BAX.
Acknowledgements
The author would like to thank E. Smith for figure preparation and graphics support. This work was supported by National Institutes of Health (NIH) grant R35CA197583 and a Leukemia and Lymphoma Society (LLS) Scholar Award. The author is also indebted to the Wolpoff Family Foundation, J. and L. LaTorre, the family of I. Coll, and the Todd J. Schwartz Memorial Fund for their steadfast financial contributions to our cancer chemical biology research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.D.W. is a scientific advisory board member and consultant for Aileron Therapeutics.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Walensky, L.D. Targeting BAX to drug death directly. Nat Chem Biol 15, 657–665 (2019). https://doi.org/10.1038/s41589-019-0306-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-019-0306-6
This article is cited by
-
Targeting PRSS23 with tipranavir induces gastric cancer stem cell apoptosis and inhibits growth of gastric cancer via the MKK3/p38 MAPK-IL24 pathway
Acta Pharmacologica Sinica (2024)
-
Covalent inhibition of pro-apoptotic BAX
Nature Chemical Biology (2024)
-
NIR/pH-triggered aptamer-functionalized DNA origami nanovehicle for imaging-guided chemo-phototherapy
Journal of Nanobiotechnology (2023)
-
Targeting a mitochondrial E3 ubiquitin ligase complex to overcome AML cell-intrinsic Venetoclax resistance
Leukemia (2023)
-
The effect of apigenin and chemotherapy combination treatments on apoptosis-related genes and proteins in acute leukaemia cell lines
Scientific Reports (2022)