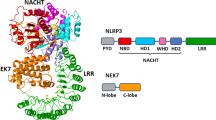
Abstract
NLRP3 (NOD-like receptor pyrin domain-containing protein 3) is an innate immune sensor that contributes to the development of different diseases, including monogenic autoinflammatory syndromes, gout, atherosclerosis, and Alzheimer’s disease. The molecule sulfonylurea MCC950 is a NLRP3 inflammasome inhibitor with potential clinical utility. However, the mechanism of action of MCC950 remains unknown. Here, we characterize the mechanism of action of MCC950 in both wild-type and autoinflammatory-related NLRP3 mutants, and demonstrate that MCC950 closes the ‘open’ conformation of active NLRP3.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Source data for Figs. 1, 2 and 3 are presented online. Supplementary Figs. 8 and 9 present data for Figs. 1a,f,g and 3a–c,e as dot-plots to shows data distribution. Supplementary Fig. 10 presents uncropped western blots presented in the main and supplementary figures. All other data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
McGettrick, A. F. et al. Diabetes, Obes. Metab. 15, 19–25 (2013).
Heneka, M. T. et al. Nat. Immunol. 16, 229–236 (2015).
Martinon, F. et al. Nature 440, 237–241 (2006).
Masters, S. L. et al. Sci. Transl. Med. 3, 81ps17 (2011).
de Torre-Minguela, C. et al. Front. Immunol. 8, 43 (2017).
Schroder, K. & Tschopp, J. Cell 140, 821–832 (2010).
Rubartelli, A. et al. Front. Immunol. 4, 398 (2013).
Hafner-Bratkovič, I. & Pelegrín, P. Curr. Opin. Immunol. 52, 8–17 (2018).
Boucher, D. et al. J. Exp. Med. 215, 827–840 (2018).
Laliberte, R. E. et al. J. Biol. Chem. 278, 16567–16578 (2003).
Coll, R. C. et al. Nat. Med. 21, 248–255 (2015).
Primiano, M. J. et al. J. Immunol. 197, 2421–2433 (2016).
Jiang, H. et al. J. Exp. Med. 214, 3219–3238 (2017).
Cocco, M. et al. J. Med. Chem. 60, 3656–3671 (2017).
Baldwin, A. G. et al. Cell Chem. Biol. 24, 1321–1335.e5 (2017).
Compan, V. et al. Immunity 37, 487–500 (2012).
Martín-Sánchez, F. et al. Methods Mol. Biol. 1417, 159–168 (2016).
Mensa-Vilaro, A. et al. Arthritis Rheumatol. 68, 3035–3041 (2016).
Duncan, J. A. et al. Proc. Natl Acad. Sci. 104, 8041–8046 (2007).
Hafner-Bratkovič, I. et al. Nat. Commun. 9, 5182 (2018).
He, Y. et al. Nature 530, 354–357 (2016).
Shi, H. et al. Nat. Immunol. 17, 250–258 (2016).
Coll, R. C. et al. Nat Chem Biol. https://doi.org/10.1038/s41589-019-0277-7 (2019).
Virginio, C. et al. J. Physiol. 519, 335–346 (1999).
Martín-Sánchez, F. et al. Cell Death Differ. 23, 1219–1231 (2016).
Compan, V. et al. J. Immunol. 194, 1261–1273 (2015).
Martín-Sánchez, F. et al. Cell Death Dis 8, e2984 (2017).
Sester, D. P. et al. J. Immunol. 194, 455–462 (2014).
de Torre-Minguela, C. et al. Sci. Rep. 6, 22586 (2016).
Young, M. T. et al. Br. J. Pharmacol. 149, 261–268 (2006).
Trott, O. & Olson, A. J. Comput. Chem. 31, 455–461 (2010).
Frisch, M. J. et al. Gaussian, Inc. (Wallingford, CT, 2016).
Sánchez-Linares, I. et al. BMC Bioinformatics 13, S13 (2012).
Acknowledgements
We thank M. C. Baños and A. I. Gómez for technical assistance with molecular biology and cell culture. We also wish to thank L. Martínez-Alarcón for help with healthy volunteer blood collection, I. Hafner-Bratkovič (National Institute of Chemistry, Ljubljana, Slovenia) for stable immortalized macrophages lines, and C. Vargas (Hospital Virgen de la Macarena, Sevilla, Spain), E. Ramos and S. Jimenez-Treviño (Hospital Central de Asturias, Oviedo, Spain), and M. Basagaña Torrento (Hospital Universitario Germans Trias i Pujol, Badalona, Spain) for samples from individuals with autoinflammatory syndromes. We also thank the patients and healthy volunteers enrolled in this study, and the Biobanco en Red de la Región de Murcia (PT13/0010/0018), which is integrated into the Spanish National Biobanks Network (B.000859), for its collaboration. This research was partially supported by the e-infrastructure program of the Research Council of Norway, and the supercomputer center of UiT at the Arctic University of Norway. The authors are also grateful for the computer resources at CTE-POWER and the technical support provided by Barcelona Supercomputing Center (RES-BCV-2018-3-0008). H.M.-B. was supported by a Rio Hortega fellowship from the Instituto Salud Carlos III (CM14/00008). D.A.-B. was supported by a Juan de la Cierva postdoctoral fellowship from the Ministerio de Economía y Competitividad (FJCI-2014-22041). This work was supported by grants from the Instituto Salud Carlos III-Fondo Europeo de Desarrollo Regional (PI13/00174 to P.P.), the Ministerio de Economia, Industria y Competitividad–Fondo Europeo de Desarrollo Regional (project nos. SAF2017-88276-R to P.P. and CTQ2017-87974-R to H.P.-S.), Fundación Séneca (20859/PI/18 to P.P.), and the European Research Council (ERC-2013-CoG 614578 to P.P.).
Author information
Authors and Affiliations
Contributions
A.T.-A., D.A.-B., H.M.-B. and C.dT.-M. conducted the experiments and interpreted data. J.P.C.-C. and H.P.-S. conducted BD and MD simulations. D.A.-B. performed structural modeling. J.I.A. coordinated human samples from autoinflammatory individuals. P.P. conceived, designed, and supervised this study, wrote the paper with feedback from all coauthors, and sourced funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–10
Rights and permissions
About this article
Cite this article
Tapia-Abellán, A., Angosto-Bazarra, D., Martínez-Banaclocha, H. et al. MCC950 closes the active conformation of NLRP3 to an inactive state. Nat Chem Biol 15, 560–564 (2019). https://doi.org/10.1038/s41589-019-0278-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-019-0278-6
This article is cited by
-
Drugging the NLRP3 inflammasome: from signalling mechanisms to therapeutic targets
Nature Reviews Drug Discovery (2024)
-
Pathogenic NLRP3 mutants form constitutively active inflammasomes resulting in immune-metabolic limitation of IL-1β production
Nature Communications (2024)
-
Pyroptotic cell death: an emerging therapeutic opportunity for radiotherapy
Cell Death Discovery (2024)
-
IP3R2-mediated Ca2+ release promotes LPS-induced cardiomyocyte pyroptosis via the activation of NLRP3/Caspase-1/GSDMD pathway
Cell Death Discovery (2024)
-
Pioglitazone ameliorates ischemia/reperfusion-induced acute kidney injury via oxidative stress attenuation and NLRP3 inflammasome
Human Cell (2024)