Abstract
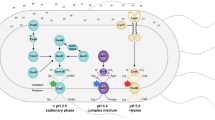
To successfully colonize the intestine, bacteria must survive passage through the stomach. The permeability of the outer membrane renders the periplasm of Gram-negative bacteria vulnerable to stomach acid, which inactivates proteins. Here we report that the semipermeable nature of the outer membrane allows the development of a strong Donnan equilibrium across this barrier at low pH. As a result, when bacteria are exposed to conditions that mimic gastric juice, periplasmic chloride concentrations rise to levels that exceed 0.6 M. At these chloride concentrations, proteins readily aggregate in vitro. The acid sensitivity of strains lacking acid-protective chaperones is enhanced by chloride, suggesting that these chaperones protect periplasmic proteins both from acidification and from the accompanying accumulation of chloride. These results illustrate how organisms have evolved chaperones to respond to the substantial chemical threat imposed by otherwise innocuous chloride concentrations that are amplified to proteotoxic levels by low-pH-induced Donnan equilibrium effects.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data used in this study are included in this published article (and its supplementary information files) and are available from the corresponding authors upon reasonable request.
References
Dressman, J. B. et al. Upper gastrointestinal (GI) pH in young, healthy men and women. Pharm. Res. 7, 756–761 (1990).
Russell, T. L. et al. Upper gastrointestinal pH in seventy-nine healthy, elderly, North American men and women. Pharm. Res. 10, 187–196 (1993).
Smith, J. L. The role of gastric acid in preventing foodborne disease and how bacteria overcome acid conditions. J. Food. Prot. 66, 1292–1303 (2003).
Kanjee, U. & Houry, W. A. Mechanisms of acid resistance in Escherichia coli. Annu. Rev. Microbiol. 67, 65–81 (2013).
Nikaido, H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol. Mol. Biol. Rev. 67, 593–656 (2003).
Malki, A. et al. Solubilization of protein aggregates by the acid stress chaperones HdeA and HdeB. J. Biol. Chem. 283, 13679–13687 (2008).
Hong, W. et al. Periplasmic protein HdeA exhibits chaperone-like activity exclusively within stomach pH range by transforming into disordered conformation. J. Biol. Chem. 280, 27029–27034 (2005).
Kern, R., Malki, A., Abdallah, J., Tagourti, J. & Richarme, G. Escherichia coli HdeB is an acid stress chaperone. J. Bacteriol. 189, 603–610 (2007).
Tapley, T. L. et al. Structural plasticity of an acid-activated chaperone allows promiscuous substrate binding. Proc. Natl. Acad. Sci. USA 106, 5557–5562 (2009).
Zhang, M. et al. A genetically incorporated crosslinker reveals chaperone cooperation in acid resistance. Nat. Chem. Biol. 7, 671–677 (2011).
Zhang, S. et al. Comparative proteomics reveal distinct chaperone-client interactions in supporting bacterial acid resistance. Proc. Natl. Acad. Sci. USA 113, 10872–10877 (2016).
Gajiwala, K. S. & Burley, S. K. HDEA, a periplasmic protein that supports acid resistance in pathogenic enteric bacteria. J. Mol. Biol. 295, 605–612 (2000).
Tapley, T. L., Franzmann, T. M., Chakraborty, S., Jakob, U. & Bardwell, J. C. A. Protein refolding by pH-triggered chaperone binding and release. Proc. Natl. Acad. Sci. USA 107, 1071–1076 (2010).
Dahl, J.-U. et al. HdeB functions as an acid-protective chaperone in bacteria. J. Biol. Chem. 290, 65–75 (2015).
Ding, J., Yang, C., Niu, X., Hu, Y. & Jin, C. HdeB chaperone activity is coupled to its intrinsic dynamic properties. Sci. Rep. 5, 16856 (2015).
Meeroff, J. C., Rofrano, J. A. & Meeroff, M. Electrolytes of the gastric juice in health and gastroduodenal diseases. Am. J. Dig. Dis. 18, 865–872 (1973).
Goto, Y. & Fink, A. L. Conformational states of beta-lactamase: molten-globule states at acidic and alkaline pH with high salt. Biochemistry 28, 945–952 (1989).
Goto, Y., Takahashi, N. & Fink, A. L. Mechanism of acid-induced folding of proteins. Biochemistry 29, 3480–3488 (1990).
Fink, A. L., Calciano, L. J., Goto, Y., Kurotsu, T. & Palleros, D. R. Classification of acid denaturation of proteins: intermediates and unfolded states. Biochemistry 33, 12504–12511 (1994).
Goto, Y. & Fink, A. L. Phase diagram for acidic conformational states of apomyoglobin. J. Mol. Biol. 214, 803–805 (1990).
Goto, Y., Calciano, L. J. & Fink, A. L. Acid-induced folding of proteins. Proc. Natl. Acad. Sci. USA 87, 573–577 (1990).
Lin, S. et al. Site-specific incorporation of photo-cross-linker and bioorthogonal amino acids into enteric bacterial pathogens. J. Am. Chem. Soc. 133, 20581–20587 (2011).
Yang, Y. et al. Genetically encoded protein photocrosslinker with a transferable mass spectrometry-identifiable label. Nat. Commun. 7, 12299 (2016).
Kararli, T. T. Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm. Drug. Dispos. 16, 351–380 (1995).
Stock, J. B., Rauch, B. & Roseman, S. Periplasmic space in Salmonella typhimurium and Escherichia coli. J. Biol. Chem. 252, 7850–7861 (1977).
Sen, K., Hellman, J. & Nikaido, H. Porin channels in intact cells of Escherichia coli are not affected by Donnan potentials across the outer membrane. J. Biol. Chem. 263, 1182–1187 (1988).
Cayley, S., Lewis, B. A., Guttman, H. J. & Record, M. T. Jr. Characterization of the cytoplasm of Escherichia coli K-12 as a function of external osmolarity. Implications for protein-DNA interactions in vivo. J. Mol. Biol. 222, 281–300 (1991).
Cayley, S., Lewis, B. A. & Record, M. T. Jr. Origins of the osmoprotective properties of betaine and proline in Escherichia coli K-12. J. Bacteriol. 174, 1586–1595 (1992).
Cayley, D. S., Guttman, H. J. & Record, M. T. Jr. Biophysical characterization of changes in amounts and activity of Escherichia coli cell and compartment water and turgor pressure in response to osmotic stress. Biophys. J. 78, 1748–1764 (2000).
Epstein, W. The roles and regulation of potassium in bacteria. Prog. Nucleic. Acid. Res. Mol. Biol. 75, 293–320 (2003).
Quan, S., Hiniker, A., Collet, J.-F. & Bardwell, J. C. A. Isolation of bacteria envelope proteins. Methods Mol. Biol. 966, 359–366 (2013).
Iyer, R., Iverson, T. M., Accardi, A. & Miller, C. A biological role for prokaryotic ClC chloride channels. Nature 419, 715–718 (2002).
Accardi, A. & Miller, C. Secondary active transport mediated by a prokaryotic homologue of ClC Cl- channels. Nature 427, 803–807 (2004).
Richard, H. & Foster, J. W. Escherichia coli glutamate- and arginine-dependent acid resistance systems increase internal pH and reverse transmembrane potential. J. Bacteriol. 186, 6032–6041 (2004).
Yang, M. et al. Biocompatible click chemistry enabled compartment-specific pH measurement inside E. coli. Nat. Commun. 5, 4981 (2014).
Bloomfield, V. A., Crothers, D. M. & Tinoco, I. Jr. Nucleic Acids: Structures, Properties and Functions (University Science Books, Sausalito, CA, 2000).
Gray, M. J. et al. Polyphosphate is a primordial chaperone. Mol. Cell 53, 689–699 (2014).
Docter, B. E., Horowitz, S., Gray, M. J., Jakob, U. & Bardwell, J. C. A. Do nucleic acids moonlight as molecular chaperones? Nucleic Acids Res. 44, 4835–4845 (2016).
Lennon, C. W. et al. Folding optimization in vivo uncovers new chaperones. J. Mol. Biol. 427, 2983–2994 (2015).
Schuck, P. Size-distribution analysis of macromolecules by sedimentation velocity ultracentrifugation and lamm equation modeling. Biophys. J. 78, 1606–1619 (2000).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 116, 71–76 (1996).
Acknowledgements
We thank P. Koldewey (University of Michigan) for providing technical expertise on the analytical ultracentrifugation experiments, R. Mitra (University Michigan) for preliminary assistance with the CD experiments and C. Miller (Brandeis U.) for the E. coli strain with both Cl–/H+ antiporters knocked out. The primary antibodies for detecting DsbC and AppA were a gift from M. Berkmen (New England BioLabs); the antibodies for detecting Skp, SurA and DegP were a gift from T. Silhavy (Princeton University); the antibody for detecting RBP was a gift from M. Ehrmann (University of Duisburg-Essen). We also thank the Purdue Cryo-EM facility, especially S. Mattoo (Purdue University) for arranging access and T. Klose (Purdue University) for help with data collection. This work was funded by the National Institutes of Health (R01-GM102829 to J.C.A.B. and R00-GM111767 to R.B.S.) and an Alfred P. Sloan Research Fellowship (to R.B.S.). J.C.A.B is a Howard Hughes Medical Investigator.
Author information
Authors and Affiliations
Contributions
F.S. and J.C.A.B. conceived the project. F.S. and H.H. performed the experiments. R.B.S. provided technical expertise on the solute distribution measurements. All authors analyzed the data. F.S. wrote the manuscript with contributions from R.B.S. and J.C.A.B.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–2, Supplementary Figures 1–6
Supplementary Video 1
Cryo-electron tomography tilt of E. coli at pH 2
Supplementary Video 2
Cryo-electron tomography tilt of E. coli at pH 7
Rights and permissions
About this article
Cite this article
Stull, F., Hipp, H., Stockbridge, R.B. et al. In vivo chloride concentrations surge to proteotoxic levels during acid stress. Nat Chem Biol 14, 1051–1058 (2018). https://doi.org/10.1038/s41589-018-0143-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-018-0143-z
This article is cited by
-
The Donnan potential revealed
Nature Communications (2022)
-
The role of bacterial cell envelope structures in acid stress resistance in E. coli
Applied Microbiology and Biotechnology (2020)
-
Protein quality control: from mechanism to disease
Cell Stress and Chaperones (2019)
-
Compartmentalizing acid stress in bacteria
Nature Chemical Biology (2018)