Abstract
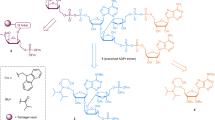
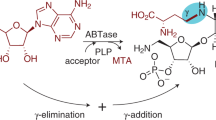
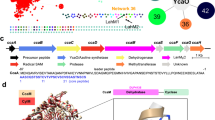
Interrupted adenylation domains are enigmatic fusions, in which one enzyme is inserted into another to form a highly unusual bifunctional enzyme. We present the first crystal structure of an interrupted adenylation domain that reveals a unique embedded methyltransferase. The structure and functional data provide insight into how these enzymes N-methylate amino acid precursors en route to nonribosomal peptides.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Park, H. B. et al. J. Nat. Prod. 74, 2309–2312 (2011).
Chatterjee, J., Rechenmacher, F. & Kessler, H. Angew. Chem. Int. Ed. Engl. 52, 254–269 (2013).
Chatterjee, J., Gilon, C., Hoffman, A. & Kessler, H. Acc. Chem. Res. 41, 1331–1342 (2008).
van der Velden, N.S. et al. Nat. Chem. Biol. 13, 833–835 (2017).
Ramm, S. et al. Angew. Chem. Int. Ed. Engl. 56, 9994–9997 (2017).
Labby, K. J., Watsula, S. G. & Garneau-Tsodikova, S. Nat. Prod. Rep. 32, 641–653 (2015).
Lombo, F. et al. ChemBioChem 7, 366–376 (2006).
Watanabe, K. et al. Nat. Chem. Biol. 2, 423–428 (2006).
Praseuth, A. P. et al. Biotechnol. Prog. 24, 1226–1231 (2008).
Watanabe, K. et al. J. Am. Chem. Soc. 131, 9347–9353 (2009).
Reimer, J. M., Aloise, M. N., Harrison, P. M. & Schmeing, T. M. Nature 529, 239–242 (2016).
Zhang, W., Heemstra, J. R. Jr., Walsh, C. T. & Imker, H. J. Biochemistry 49, 9946–9947 (2010).
Felnagle, E. A. et al. Biochemistry 49, 8815–8817 (2010).
Herbst, D. A., Boll, B., Zocher, G., Stehle, T. & Heide, L. J. Biol. Chem. 288, 1991–2003 (2013).
Miller, B. R., Drake, E. J., Shi, C., Aldrich, C. C. & Gulick, A. M. J. Biol. Chem. 291, 22559–22571 (2016).
Tarry, M. J., Haque, A. S., Bui, K. H. & Schmeing, T. M. Structure 25, 783–793 (2017).
Shi, R. et al. PLoS Biol. 8, e1000354 (2010).
Al-Mestarihi, A. H. et al. J. Am. Chem. Soc. 136, 17350–17354 (2014).
Mori, S., Garzan, A., Tsodikov, O. V. & Garneau-Tsodikova, S. Biochemistry 56, 6087–6097 (2017).
Ansari, M. Z., Sharma, J., Gokhale, R. S. & Mohanty, D. BMC Bioinformatics 9, 454 (2008).
Nakaya, M. et al. Biosci. Biotechnol. Biochem. 71, 2969–2976 (2007).
Hercouet, A., Bessieres, B. & Le Corre, M. Tetrahedron 7, 283–284 (1996).
Zolova, O. E. & Garneau-Tsodikova, S. Med. Chem. Commun. 3, 950–955 (2012).
Mady, A. S. et al. Mol. BioSyst. 7, 1999–2011 (2011).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 (1997).
McCoy, A. J. et al. J. Appl. Crystallogr. 40, 658–674 (2007).
Murshudov, G. N. et al. Acta Crystallogr. D Biol. Crystallogr. 67, 355–367 (2011).
Emsley, P. & Cowtan, K. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Acknowledgements
This work was supported by a NSF CAREER Award MCB-1149427 (to S.G.-T.) and by startup funds from the College of Pharmacy at the University of Kentucky (to S.G.-T. and O.V.T.).
Author information
Authors and Affiliations
Contributions
S.M., A.H.P., T.A.L., O.V.T., and S.G.-T. designed the study and wrote the manuscript. S.M. and T.A.L. performed biochemical experiments. A.H.P. performed crystallization and structure determination. A.G. performed chemical synthesis. S.M. and A.H.P. contributed equally.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–4 and Supplementary Figure 1–21
Rights and permissions
About this article
Cite this article
Mori, S., Pang, A.H., Lundy, T.A. et al. Structural basis for backbone N-methylation by an interrupted adenylation domain. Nat Chem Biol 14, 428–430 (2018). https://doi.org/10.1038/s41589-018-0014-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-018-0014-7
This article is cited by
-
Catalytic trajectory of a dimeric nonribosomal peptide synthetase subunit with an inserted epimerase domain
Nature Communications (2022)
-
Structures and function of a tailoring oxidase in complex with a nonribosomal peptide synthetase module
Nature Communications (2022)
-
Structural basis of keto acid utilization in nonribosomal depsipeptide synthesis
Nature Chemical Biology (2020)
-
Does the Future of Antibiotics Lie in Secondary Metabolites Produced by Xenorhabdus spp.? A Review
Probiotics and Antimicrobial Proteins (2020)
-
Modified substrate specificity of a methyltransferase domain by protein insertion into an adenylation domain of the bassianolide synthetase
Journal of Biological Engineering (2019)