Abstract
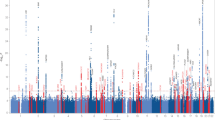
The genetic basis of Lewy body dementia (LBD) is not well understood. Here, we performed whole-genome sequencing in large cohorts of LBD cases and neurologically healthy controls to study the genetic architecture of this understudied form of dementia, and to generate a resource for the scientific community. Genome-wide association analysis identified five independent risk loci, whereas genome-wide gene-aggregation tests implicated mutations in the gene GBA. Genetic risk scores demonstrate that LBD shares risk profiles and pathways with Alzheimer’s disease and Parkinson’s disease, providing a deeper molecular understanding of the complex genetic architecture of this age-related neurodegenerative condition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Individual-level sequence data for the resource genomes have been deposited at dbGaP (accession no. phs001963.v1.p1 NIA DementiaSeq). The GWAS summary statistics have been deposited in the GWAS catalog: https://www.ebi.ac.uk/gwas/home. eQTLGen data are available at https://www.eqtlgen.org/cis-eqtls.html. PsychENCODE QTL data are available at http://resource.psychencode.org/. Bulk-tissue RNA-seq data (GTEx v.8) are available at the Genotype-Tissue Expression consortium portal (https://www.gtexportal.org/home/). Human single-nucleus RNA-seq data are available at the Allen Institute for Brain Science portal (portal.brain-map.org/atlases-and-data/rnaseq/human-mtg/smart-seq). Specificity values for the Allen Institute for Brain Science and GTEx data are available at: https://github.com.RHReynolds/MarkerGenes.
Code availability
Analyses were performed using open-source tools, and code for analysis is available at the associated website of each software package. Genome sequence alignment and variant calling followed the implementation of the GATK Best Practices pipeline (v.2016-June) (https://github.com/gatk-workflows/broad-prod-wgs-germline-snps-indels). Contamination rates were assessed using VerifyBamID (v.1.1.3) (https://genome.sph.umich.edu/wiki/VerifyBamID). Quality control checks, association analyses and conditional analyses were performed in PLINK2 (v.2.0-dev-20191128) (https://www.cog-genomics.org/plink/2.0/). Data formatting and visualizations were performed in R (v.3.5.2; https://www.r-project.org) using the following packages: MASS (v.7.3-51.4), tidyverse (v.1.2.1), stringr (v.1.4.0), ggrepel (v.0.8.1), data.table (v.1.12), viridis (v.0.5.1), ggplot2 (v.3.3.2), gridExtra (v.2.3) and grid (v.3.5.2). Imputation was performed using Minimac4 on data phased by Eagle (v.2.4) (https://github.com/poruloh/Eagle). Meta-analysis was performed using METAL (v.2018-08-28) (https://genome.sph.umich.edu/wiki/METAL). Heritability analysis was performed using GREML-LDMS in GCTA (v.1.26.0) (https://cnsgenomics.com/software/gcta). Rare variant analysis was performed using RVTESTS (v.2.1.0) (http://zhanxw.github.io/rvtests/) after annotation of variant files in ANNOVAR (v.2018-04/16) (https://annovar.openbioinformatics.org/en/latest/). Genetic risk score analyses were performed in PLINK 1.9 (v.1.9.0-beta4.4) (https://www.cog-genomics.org/plink). LBD summary statistics were converted from hg38 to hg19 using the R implementation of the LiftOver tool, which is available from the rtracklayer package (v.1.42.2) (genome.sph.umich.edu/wiki/LiftOver). Colocalization analyses were performed in R-3.2 using the package coloc (v.4.0.1) (https://github.com/chr1swallace/coloc). Specificity values for the AIBS-derived dataset were generated using gene-level exonic reads and the ‘generate.celltype.data’ function of the EWCE package (v.0.99.2) (https://github.com/NathanSkene/EWCE). Polygenic risk scores were constructed using PRSice-2 (v.2.1.1) (https://www.prsice.info). Pathway enrichment analysis was performed using the R package gprofiler2 (v.0.2.0) (https://cran.r-project.org/web/packages/gprofiler2/vignettes/gprofiler2.html).
References
Walker, Z., Possin, K. L., Boeve, B. F. & Aarsland, D. Lewy body dementias. Lancet 386, 1683–1697 (2015).
McKeith, I. G. et al. Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology 89, 88–100 (2017).
Meeus, B., Theuns, J. & Van Broeckhoven, C. The genetics of dementia with Lewy bodies: what are we missing? Arch. Neurol. 69, 1113–1118 (2012).
10 things you should know about LBD. Lewy Body Dementia Association https://www.lbda.org/10-things-you-should-know-about-lbd/
Guerreiro, R. et al. Investigating the genetic architecture of dementia with Lewy bodies: a two-stage genome-wide association study. Lancet Neurol. 17, 64–74 (2018).
Sabir, M. S. et al. Assessment of APOE in atypical parkinsonism syndromes. Neurobiol. Dis. 127, 142–146 (2019).
Nalls, M. A. et al. A multicenter study of glucocerebrosidase mutations in dementia with Lewy bodies. JAMA Neurol. 70, 727–735 (2013).
Singleton, A. B. et al. alpha-Synuclein locus triplication causes Parkinson’s disease. Science 302, 841 (2003).
Tsuang, D. et al. APOE epsilon4 increases risk for dementia in pure synucleinopathies. JAMA Neurol. 70, 223–228 (2013).
Pickering-Brown, S. M. et al. Apolipoprotein E4 and Alzheimer’s disease pathology in Lewy body disease and in other beta-amyloid-forming diseases. Lancet 343, 1155 (1994).
Seshadri, S. et al. Genome-wide analysis of genetic loci associated with Alzheimer disease. JAMA 303, 1832–1840 (2010).
Pankratz, N. et al. Genomewide association study for susceptibility genes contributing to familial Parkinson disease. Hum. Genet. 124, 593–605 (2009).
Lee, S. et al. Optimal unified approach for rare-variant association testing with application to small-sample case-control whole-exome sequencing studies. Am. J. Hum. Genet. 91, 224–237 (2012).
Geiger, J. T. et al. Next-generation sequencing reveals substantial genetic contribution to dementia with Lewy bodies. Neurobiol. Dis. 94, 55–62 (2016).
Singleton, A. & Hardy, J. A generalizable hypothesis for the genetic architecture of disease: pleomorphic risk loci. Hum. Mol. Genet. 20, R158–R162 (2011).
Nicolae, D. L. et al. Trait-associated SNPs are more likely to be eQTLs: annotation to enhance discovery from GWAS. PLoS Genet. 6, e1000888 (2010).
Li, Y. I. et al. RNA splicing is a primary link between genetic variation and disease. Science 352, 600–604 (2016).
Võsa, U. et al. Unraveling the polygenic architecture of complex traits using blood eQTL metaanalysis. Preprint at bioRxiv https://doi.org/10.1101/447367 (2018).
Wang, D. et al. Comprehensive functional genomic resource and integrative model for the human brain. Science 362, eaat8464 (2018).
Hawrylycz, M. J. et al. An anatomically comprehensive atlas of the adult human brain transcriptome. Nature 489, 391–399 (2012).
GTEx Consortium. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Kunkle, B. W. et al. Genetic meta-analysis of diagnosed Alzheimer’s disease identifies new risk loci and implicates Abeta, tau, immunity and lipid processing. Nat. Genet. 51, 414–430 (2019).
Nalls, M. A. et al. Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: a meta-analysis of genome-wide association studies. Lancet Neurol. 18, 1091–1102 (2019).
Kramarz, B. et al. Improving the gene ontology resource to facilitate more informative analysis and interpretation of Alzheimer’s disease data. Genes (Basel) 9, 593 (2018).
Bandres-Ciga, S. et al. The endocytic membrane trafficking pathway plays a major role in the risk of Parkinson’s disease. Mov. Disord. 34, 460–468 (2019).
Morris, J. C. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology 43, 2412–2414 (1993).
Miyagawa, T. et al. BIN1 regulates BACE1 intracellular trafficking and amyloid-beta production. Hum. Mol. Genet. 25, 2948–2958 (2016).
Calafate, S., Flavin, W., Verstreken, P. & Moechars, D. Loss of Bin1 promotes the propagation of Tau pathology. Cell Rep. 17, 931–940 (2016).
Jinn, S. et al. TMEM175 deficiency impairs lysosomal and mitochondrial function and increases alpha-synuclein aggregation. Proc. Natl Acad. Sci. USA 114, 2389–2394 (2017).
Corder, E. H. et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 261, 921–923 (1993).
Sidransky, E. et al. Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N. Engl. J. Med. 361, 1651–1661 (2009).
Simon-Sanchez, J. et al. Genome-wide association study reveals genetic risk underlying Parkinson’s disease. Nat. Genet. 41, 1308–1312 (2009).
Jansen, I. E. et al. Genome-wide meta-analysis identifies new loci and functional pathways influencing Alzheimer’s disease risk. Nat. Genet. 51, 404–413 (2019).
Ross, O. A. et al. Genomic investigation of alpha-synuclein multiplication and parkinsonism. Ann. Neurol. 63, 743–750 (2008).
Pollard, M. O., Gurdasani, D., Mentzer, A. J., Porter, T. & Sandhu, M. S. Long reads: their purpose and place. Hum. Mol. Genet. 27, R234–R241 (2018).
Erikson, G. A. et al. Whole-genome sequencing of a healthy aging cohort. Cell 165, 1002–1011 (2016).
Emre, M. et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov. Disord. 22, 1689–1707 (2007).
Savica, R. et al. Incidence of dementia with Lewy bodies and Parkinson disease dementia. JAMA Neurol. 70, 1396–1402 (2013).
Regier, A. A. et al. Functional equivalence of genome sequencing analysis pipelines enables harmonized variant calling across human genetics projects. Nat. Commun. 9, 4038 (2018).
Van der Auwera, G. A. et al. From FastQ data to high confidence variant calls: the Genome Analysis Toolkit best practices pipeline. Curr. Protoc. Bioinformatics 43, 11.10.1–11.10.33 (2013).
Jun, G. et al. Detecting and estimating contamination of human DNA samples in sequencing and array-based genotype data. Am. J. Hum. Genet. 91, 839–848 (2012).
International HapMap Consortiumet al. Integrating common and rare genetic variation in diverse human populations. Nature 467, 52–58 (2010).
Schneider, V. A. et al. Evaluation of GRCh38 and de novo haploid genome assemblies demonstrates the enduring quality of the reference assembly. Genome Res. 27, 849–864 (2017).
Karczewski, K. J. et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581, 434–443 (2020).
Chang, C. C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4, 7 (2015).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics with S (Springer, 2002).
Das, S. et al. Next-generation genotype imputation service and methods. Nat. Genet. 48, 1284–1287 (2016).
Fuchsberger, C., Abecasis, G. R. & Hinds, D. A. minimac2: faster genotype imputation. Bioinformatics 31, 782–784 (2015).
Taliun, D. et al. Sequencing of 53,831 diverse genomes from the NHLBI TOPMed Program. Preprint at bioRxiv https://doi.org/10.1101/563866 (2019).
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
Mirra, S. S. et al. The consortium to establish a registry for Alzheimer’s Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer’s disease. Neurology 41, 479–486 (1991).
Braak, H., Alafuzoff, I., Arzberger, T., Kretzschmar, H. & Del Tredici, K. Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol. 112, 389–404 (2006).
Giambartolomei, C. et al. Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet. 10, e1004383 (2014).
Wallace, C. Eliciting priors and relaxing the single causal variant assumption in colocalisation analyses. PLoS Genet. 16, e1008720 (2020).
Bryois, J. et al. Genetic identification of cell types underlying brain complex traits yields insights into the etiology of Parkinson’s disease. Nat. Genet. 52, 482–493 (2020).
Skene, N. G. & Grant, S. G. Identification of vulnerable cell types in major brain disorders using single cell transcriptomes and expression weighted cell type enrichment. Front. Neurosci. 10, 16 (2016).
Yang, J. et al. Genetic variance estimation with imputed variants finds negligible missing heritability for human height and body mass index. Nat. Genet. 47, 1114–1120 (2015).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
Zhan, X., Hu, Y., Li, B., Abecasis, G. R. & Liu, D. J. RVTESTS: an efficient and comprehensive tool for rare variant association analysis using sequence data. Bioinformatics 32, 1423–1426 (2016).
Bohler, A. et al. Reactome from a WikiPathways perspective. PLoS Comput. Biol. 12, e1004941 (2016).
Kanehisa, M., Sato, Y., Kawashima, M., Furumichi, M. & Tanabe, M. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res. 44, D457–D462 (2016).
Acknowledgements
We thank contributors who collected samples used in this study, as well as patients and families whose help and participation made this work possible. This research was supported in part by the Intramural Research Program of the National Institutes of Health (National Institute on Aging and National Institute of Neurological Disorders and Stroke: (project nos. 1ZIAAG000935 (B.J.T.), 1ZIANS003154 (S.W.S.), 1ZIANS0030033 and 1ZIANS003034 (D.S.G.)). This work used the computational resources of the NIH HPC Biowulf cluster. (http://hpc.nih.gov). A complete list of acknowledgments is given in the Supplementary Note.
Author information
Authors and Affiliations
Consortia
Contributions
C.L.D., B.J.T. and S.W.S. conceptualized and supervised the study. M.S.S., S.A., R.L.W., J.T.G. and Y.A. performed sample preparations. C.V. performed library preparations and genome sequencing. J.D., A.M., J.R.G. and C.L.D. performed genome sequence alignment, variant calling and initial quality control checks. R.C., S.W.S. and B.J.T. curated the data. R.C. performed quality control checks and genome-wide association analysis, and Z.S. contributed to this analysis. R.C. also led the genome-wide, gene-based rare variant analysis, with contributions from M.B.M., M.D.-F. and C.B. M.S.S. performed the heritability analysis. S.B.-C. performed the genetic risk score analysis. S.S.-A. computed polygenic risk scores and performed enrichment analyses. R.H.R., E.G. and M.R. performed eQTL analyses. M.A.N. consulted on the statistical analysis. M.K.P. performed validation experiments. R.H.R., M.R., M.G., A. Calvo, G.M., A. Canosa, G.F., R.C.B., F.B., Z.G.-O., P.M., R.K., D.S.G., G.L., N.T., E.S., L.N.-K., J.-A.P., H.K., V.G.S., M.P., K.L.N., E.M., R.C.K., C.A.C., E.S.M., M.B., T.M.D., L.S.R., M.S.A., O.P., J.C.T., M.E.F., Q.M., E.H.B., E.R.-R., J.I., C.L., I.G.-A., P.S.-J., B.G., J.K., S.E.B., M.M., E.R., C.D., A.B., S.L., G.X., M.J.B., B.S.T., S.G., G.L., G.E.S., T.G.B., I.G.M., A.J.T., J.A., C.M.M., L.P., S.L., C.T., S.A.-S., A.K.H., D. Aarsland, G.K., S.M.K., R.W., P.P., L.M. Bekris, J.B.L., L.M. Besser, A.K., A.E.R., A.G., D.A.B., C.R.S., H.R.M., R.F., D. Albani, S.P.-B., K.F., W.A.K., E.M.-R., A.L., J.F., D. Alcolea, J.C., L.F., S.M.R., T.T., T.M.F., N.R.G.-R., Z.K.W., T.F., B.F.B., J.A.H., D.W.D., A.B.S., A. Chiò, O.A.R., B.J.T. and S.W.S. provided biospecimens and clinical data. D.J.S., J.E., L.P., A.B.S., J.A.H., O.A., L.C., L.S.H., K. Marder, A.L., P. St.G.-H., E.L., K. Morgan, T.L., T.T.W., Z.J., D.G., I.S., P.J.T., L.M., M.O., N.J.C., J.C.M., G.M.H., V.M.V.D., J.Q.T., T.G., C.S., F.L., E.S., D.C., A. Chiò, B.J.T. and S.W.S. provided replication data. A.T., E.J.T., D.G.H., J.R.G. and A.B.S. provided convenience control genomes. S.W.S. wrote the initial manuscript. All authors critically reviewed and edited the article.
Corresponding author
Ethics declarations
Competing interests
T.G.B. is a consultant for Prothena Biosciences, Vivid Genomics and Avid Radiopharmaceutical, and is a scientific advisory board member for Vivid Genomics. J.A.H., H.R.M., S.P.-B., P.J.T. and B.J.T. hold US, EU and Canadian patents on the clinical testing and therapeutic intervention for the hexanucleotide repeat expansion of C9orf72. H.R.M. reports paid consultancy from Biogen, Biohaven, Lundbeck, UCB and Denali and lecture fees/honoraria from the Wellcome Trust and Movement Disorders Society. H.R.M. received research grants from Parkinson’s UK, Cure Parkinson’s Trust, PSP Association, CBD Solutions, Drake Foundation and the Medical Research Council. H.R.M. is a coapplicant on a patent application related to C9orf72—Method for diagnosing a neurodegenerative disease (no. PCT/GB2012/052140). J.E. was an employee of a for-profit company (Merck) at the time of the collaboration. M.A.N.‘s participation is supported by a consulting contract between Data Tecnica International and the National Institute on Aging, NIH, Bethesda, MD, USA; as a possible conflict of interest, M.A.N. also consults for Neuron23 Inc., Lysosomal Therapeutics Inc., Illumina Inc., the Michael J. Fox Foundation and Vivid Genomics among others. A.B.S. is an associate editor for the journals Brain, Movement Disorders and npj Parkinson’s Disease. H.K. is Editor-in-Chief of Clinical Autonomic Research, serves as PI of a clinical trial sponsored by Biogen MA, Inc. (TRACK MSA, no. S19-01846) and received consultancy fees from Lilly USA LLC, Biohaven Pharmaceuticals Inc., Takeda Pharmaceutical Company Ltd, Ono Pharma UK Ltd, Lundbeck LLC and Theravance Biopharma US Inc. J.-A.P. is an editorial board member of Movement Disorders, Parkinsonism & Related Disorders, BMC Neurology and Clinical Autonomic Research. B.F.B., J.B.L. and S.W.S. serve on the Scientific Advisory Council of the Lewy Body Dementia Association. S.W.S. is an editorial board member for the Journal of Parkinson’s Disease and JAMA Neurology. B.J.T. is an editorial board member for JAMA Neurology, Journal of Neurology, Neurosurgery and Psychiatry, Brain and Neurobiology of Aging. D.A. is associate editor of the Journal of Alzheimer’s Disease. R.K. is Coordinator of the National Centre for Excellence in Research on Parkinson’s disease and received speaker’s honoraria and/or travel grants from Abbvie, Zambon and Medtronic, and he participated as PI or site-PI for industry-sponsored clinical trials without receiving honoraria. Z.K.W. serves as a principal investigator or coprincipal investigator on Biogen, Inc. (no. 228PD201), Biohaven Pharmaceuticals, Inc. (nos. BHV4157-206 and BHV3241-301) and Neuraly, Inc. (no. NLY01-PD-1) grants. Z.K.W. also serves as the coprincipal investigator of the Mayo Clinic Florida American Parkinson Disease Association Center for Advanced Research. Z.G.-O. received consultancy fees from Lysosomal Therapeutics Inc., Idorsia, Prevail Therapeutics, Inceptions Sciences (now Ventus), Ono Therapeutics, Denali, Deerfield, Neuron23 and Handle Therapeutics. Z.G.-O. is an Associate Editor of the Journal of Parkinson’s Disease and editorial board member in Parkinsonism and Related Disorders. A.T. serves on the scientific advisory board for Vivid Genomics. All other authors report no competing interests.
Additional information
Peer review information Nature Genetics thanks Sudha Seshadri and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 BIN1 and TMEM175 genotype-phenotype analysis.
Relationship between BIN1 and TMEM175 genotypes and the presence of Alzheimer’s disease co-pathology in definite LBD cases. The color gradation refers to semi-quantitative pathological measures of neuritic plaques (assessed by CERAD method) and neurofibrillary tangles (assessed by Braak stage). Darker colors refer to higher burden of pathology. Homozygous BIN1 risk allele carriers (TT) were found to have significantly increased neurofibrillary tangle pathology compared to homozygous major allele carriers (CC; Fisher’s exact test P-value on Braak staging = 0.0002). Although the proportion of LBD cases that had high neuritic plaque burden was higher in homozygous risk allele carries compared to homozygous major allele carries, the difference between these groups was not statistically significant (P = 0.23). There was no association of TMEM175 risk allele dosage and Alzheimer’s disease co-pathology, though a trend toward lower Alzheimer’s disease co-pathology was observed among homozygous TMEM175 risk allele carriers.
Extended Data Fig. 2 Regional association plots.
a-g, Regional association plots, local linkage disequilibrium, and recombination rates at the significantly associated LBD GWAS risk signals. Regional associations are plotted as a function of their genomic position, denoting the index variant by a red diamond. Single nucleotide variants or indels surrounding the index variant are color-coded to reflect the strength of linkage disequilibrium with the index variant based on pairwise r2-values in the study cohort (red, 1.0 ≥ r2 ≥ 0.8; orange, 0.8 > r2 ≥ 0.6; green 0.6 > r2 ≥ 0.4; light blue, 0.4 > r2 ≥ 0.2; dark blue, 0.2 > r2 ≥ 0; gray, no r2 value available). Transcript annotations according to the University of California Santa Cruz genome browser are depicted under each association plot.
Extended Data Fig. 3 Conditional analysis.
a-f, Conditional analyses for all genome-wide significant GWAS signals are depicted. For each panel, the x-axis denotes the chromosomal position in build 38, and the y-axis indicates the association P-values on a -log10 scale. The unconditioned GWAS signal is shown in the upper pane of each panel, while the lower pane illustrates the association results after correction for the index variant(s) at each respective signal. This analysis demonstrated two signals at the APOE locus (e, f). The locus name is based on the closest gene to the index variant.
Extended Data Fig. 4 Sensitivity analyses.
a,b, Sensitivity analyses of colocalization between eQTLs regulating TMEM175 expression and LBD GWAS signals (a) and SNCA-AS1 expression and LBD GWAS signals (b). eQTLs for TMEM175 were derived from eQTL-Gen, while eQTLs for SNCA-AS1 were derived from PsychENCODE. Plots of prior (left) and posterior (right) probabilities for H0-H4 hypotheses across varying p12 priors are shown. A dashed vertical line indicates the value of p12 used in the initial analysis (p12 = 5 x 10−6). The green shaded areas in these plots show the regions for which the posterior probability of H4 ≥ 0.90 would still be supported. Abbreviations: H0, hypothesis 0 (no association with either trait); H1, hypothesis 1 (association with trait 1, not with trait 2); H2, hypothesis 2 (association with trait 2, not with trait 1); H3, hypothesis 3 (association with trait 1 and trait 2, two independent SNPs); H4, hypothesis 4 (association with trait 1 and trait 2, one shared SNP).
Extended Data Fig. 5 GWAS variants correlate with increased SNCA-AS1 expression.
Shown here are genome-wide significant SNPs that decrease risk for LBD and their correlation with increased SNCA-AS1 expression. a, Scatterplot of beta coefficients and association P-values (on a -log10 scale) for SNPs shared between the LBD GWAS (left) and PsychENCODE (right). The SNPs represented in this plot are those that are eQTLs regulating SNCA-AS1 expression. The top SNP in the LBD GWAS (as determined by the lowest association test P-value) is indicated in both scatterplots by a red point. The dashed line represents the cut-off for genome-wide significance (5 x 10-8). b, Scatterplot of SNPs shared between the LBD GWAS and PsychENCODE, which pass genome-wide significance in the LBD GWAS. Spearman’s rho (R) and associated P-value are displayed.
Extended Data Fig. 6 Tissue and cell-type specificity of SNCA-AS1 and TMEM175.
a,b, Plot of SNCA-AS1 and TMEM175 specificity in 35 human tissues (GTEx dataset) (a) and seven broad categories of cell types derived from human middle temporal gyrus (Allen Institute for Brain Science dataset) (b). Tissues are colored by whether they belong to the brain. In all plots, tissues and cell types have been ordered by specificity.
Extended Data Fig. 7 Tissue and cell-specificity of SNCA-AS1 and SNCA.
a,b, Plots of SNCA-AS1 and SNCA specificity in 35 human tissues (GTEx dataset) (a) and seven broad categories of cell types derived from human middle temporal gyrus (Allen Institute for Brain Science dataset) (b). Tissues are colored by whether they belong to the brain. In all plots, tissues and cell types have been ordered by specificity.
Extended Data Fig. 8 LBD polygenic risk score is associated with dementia severity.
Dementia severity score proportions (measured by the Clinical Dementia Rating scale) at baseline evaluation relative to LBD polygenic risk score quintiles. LBD patients in the highest quintile had significantly more severe cognitive impairment at baseline compared to cases in the lowest quintile (χ2 = 5.60, df = 1, test P-value = 0.009).
Extended Data Fig. 9 Principal components analysis and QQ plot.
Quality control metrics of GWAS data. a, Population structure is shown by plotting the first two principal components of the study cohorts (n = 2,591 LBD cases and n = 4,027 controls) compared to the HapMap3 Genome Reference panel. b, Quantile-quantile (QQ) plot of single-variant associations depicting observed (y-axis) versus expected P-values (x-axis). The sample size adjusted genomic inflation factor λ1000 was 1.004.
Extended Data Fig. 10 Quality control metrics.
This figure depicts quality control metrics of the genome data across study cohorts. a, Heterozygous-to-homozygous single nucleotide variant (SNV) ratios. b, Mean coverage across the study cohorts.
Supplementary information
Supplementary Information
Supplementary Note and Tables 3–5
Supplementary Tables
Supplementary Tables 1 and 2
Rights and permissions
About this article
Cite this article
Chia, R., Sabir, M.S., Bandres-Ciga, S. et al. Genome sequencing analysis identifies new loci associated with Lewy body dementia and provides insights into its genetic architecture. Nat Genet 53, 294–303 (2021). https://doi.org/10.1038/s41588-021-00785-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-021-00785-3
This article is cited by
-
Gut microbiota, circulating cytokines and dementia: a Mendelian randomization study
Journal of Neuroinflammation (2024)
-
Overlaps and divergences between tauopathies and synucleinopathies: a duet of neurodegeneration
Translational Neurodegeneration (2024)
-
Elevated serum LDL-C increases the risk of Lewy body dementia: a two-sample mendelian randomization study
Lipids in Health and Disease (2024)
-
Proxy-analysis of the genetics of cognitive decline in Parkinson’s disease through polygenic scores
npj Parkinson's Disease (2024)
-
Genetic analysis of the X chromosome in people with Lewy body dementia nominates new risk loci
npj Parkinson's Disease (2024)