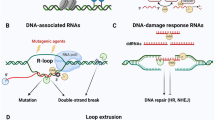
Abstract
Noncoding RNAs are exquisitely titrated by the cellular RNA surveillance machinery for regulating diverse biological processes. The RNA exosome, the predominant 3′ RNA exoribonuclease in mammalian cells, is composed of nine core and two catalytic subunits. Here, we developed a mouse model with a conditional allele to study the RNA exosome catalytic subunit DIS3. In DIS3-deficient B cells, integrity of the immunoglobulin heavy chain (Igh) locus in its topologically associating domain is affected, with accumulation of DNA-associated RNAs flanking CTCF-binding elements, decreased CTCF binding to CTCF-binding elements and disorganized cohesin localization. DIS3-deficient B cells also accumulate activation-induced cytidine deaminase–mediated asymmetric nicks, altering somatic hypermutation patterns and increasing microhomology-mediated end-joining DNA repair. Altered mutation patterns and Igh architectural defects in DIS3-deficient B cells lead to decreased class-switch recombination but increased chromosomal translocations. Our observations of DIS3-mediated architectural regulation at the Igh locus are reflected genome wide, thus providing evidence that noncoding RNA processing is an important mechanism for controlling genome organization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data are available via the NCBI under accession number PRJNA544488. Source data are provided with this paper.
Code availability
Code is available at https://github.com/basulab-cu/Dis3-project.
References
Laffleur, B. & Basu, U. Biology of RNA surveillance in development and disease. Trends Cell Biol. 29, 428–445 (2019).
Nair, L., Chung, H. & Basu, U. Regulation of long noncoding RNAs and genome dynamics by the RNA surveillance machinery. Nat. Rev. Mol. Cell Biol. 21, 123–136 (2020).
Kilchert, C., Wittmann, S. & Vasiljeva, L. The regulation and functions of the nuclear RNA exosome complex. Nat. Rev. Mol. Cell Biol. 17, 227–239 (2016).
Pefanis, E. et al. RNA exosome-regulated long noncoding RNA transcription controls super-enhancer activity. Cell 161, 774–789 (2015).
Preker, P. et al. PROMoter uPstream Transcripts share characteristics with mRNAs and are produced upstream of all three major types of mammalian promoters. Nucleic Acids Res. 39, 7179–7193 (2011).
Almada, A. E., Wu, X., Kriz, A. J., Burge, C. B. & Sharp, P. A. Promoter directionality is controlled by U1 snRNP and polyadenylation signals. Nature 499, 360–363 (2013).
Pefanis, E. et al. Noncoding RNA transcription targets AID to divergently transcribed loci in B cells. Nature 514, 389–393 (2014).
Rothschild, G. et al. Noncoding RNA transcription alters chromosomal topology to promote isotype-specific class-switch recombination. Sci. Immunol. 5, eaay5864 (2020).
Liu, J. et al. N6-methyladenosine of chromosome-associated regulatory RNA regulates chromatin state and transcription. Science 367, 580–586 (2020).
Singh, I. et al. MiCEE is a ncRNA–protein complex that mediates epigenetic silencing and nucleolar organization. Nat. Genet. 50, 990–1001 (2018).
Alt, F. W., Zhang, Y., Meng, F.-L., Guo, C. & Schwer, B. Mechanisms of programmed DNA lesions and genomic instability in the immune system. Cell 152, 417–429 (2013).
Methot, S. P. & Di Noia, J. M. Molecular mechanisms of somatic hypermutation and class-switch recombination. Adv. Immunol. 133, 37–87 (2017).
Zhang, X. et al. Fundamental roles of chromatin loop extrusion in antibody class switching. Nature 575, 385–389 (2019).
Vian, L. et al. The energetics and physiological impact of cohesin extrusion. Cell 173, 1165–1178 (2018).
Schoenfelder, S. & Fraser, P. Long-range enhancer–promoter contacts in gene expression control. Nat. Rev. Genet. 20, 437–455 (2019).
Nora, E. P. et al. Targeted degradation of CTCF decouples local insulation of chromosome domains from genomic compartmentalization. Cell 169, 930–944 (2017).
Rao, S. S. P. et al. Cohesin loss eliminates all loop domains. Cell 171, 305–320 (2017).
Schwarzer, W. et al. Two independent modes of chromatin organization revealed by cohesin removal. Nature 551, 51–56 (2017).
Zhang, Y. et al. The fundamental role of chromatin loop extrusion in physiological V(D)J recombination. Nature 573, 600–604 (2019).
Ba, Z. et al. CTCF orchestrates long-range cohesin-driven V(D)J recombinational scanning. Nature 586, 305–310 (2020).
Shen, H. M., Peters, A., Baron, B., Zhu, X. & Storb, U. Mutation of BCL-6 gene in normal B cells by the process of somatic hypermutation of Ig genes. Science 280, 1750–1752 (1998).
Mlynarczyk, C., Fontán, L. & Melnick, A. Germinal center-derived lymphomas: the darkest side of humoral immunity. Immunol. Rev. 288, 214–239 (2019).
Chiarle, R. et al. Genome-wide translocation sequencing reveals mechanisms of chromosome breaks and rearrangements in B cells. Cell 147, 107–119 (2011).
Klein, I. A. et al. Translocation-capture sequencing reveals the extent and nature of chromosomal rearrangements in B lymphocytes. Cell 147, 95–106 (2011).
Meng, F.-L. et al. Convergent transcription at intragenic super-enhancers targets AID-initiated genomic instability. Cell 159, 1538–1548 (2014).
Birshtein, B. K. Epigenetic regulation of individual modules of the immunoglobulin heavy chain locus 3′ regulatory region. Front. Immunol. 5, 163 (2014).
Nojima, T. et al. Deregulated expression of mammalian lncRNA through loss of SPT6 induces R-loop formation, replication stress and cellular senescence. Mol. Cell 72, 970–984 (2018).
Wahba, L., Amon, J. D., Koshland, D. & Vuica-Ross, M. RNase H and multiple RNA biogenesis factors cooperate to prevent RNA:DNA hybrids from generating genome instability. Mol. Cell 44, 978–988 (2011).
Domingo-Prim, J. et al. EXOSC10 is required for RPA assembly and controlled DNA end resection at DNA double-strand breaks. Nat. Commun. 10, 2135 (2019).
Merkenschlager, M. & Nora, E. P. CTCF and cohesin in genome folding and transcriptional gene regulation. Annu. Rev. Genomics Hum. Genet. 17, 17–43 (2016).
Dekker, J. & Mirny, L. The 3D genome as moderator of chromosomal communication. Cell 164, 1110–1121 (2016).
Saintamand, A. et al. Elucidation of IgH 3′ region regulatory role during class switch recombination via germline deletion. Nat. Commun. 6, 7084 (2015).
Fitz, J. et al. Spt5-mediated enhancer transcription directly couples enhancer activation with physical promoter interaction. Nat. Genet. 52, 505–515 (2020).
Wuerffel, R. et al. S-S synapsis during class switch recombination is promoted by distantly located transcriptional elements and activation-induced deaminase. Immunity 27, 711–722 (2007).
Denizot, Y. et al. Polymorphism of the human α1 immunoglobulin gene 3′ enhancer hs1,2 and its relation to gene expression. Immunology 103, 35–40 (2001).
Le Noir, S. et al. The IgH locus 3′ cis-regulatory super-enhancer co-opts AID for allelic transvection. Oncotarget 8, 12929–12940 (2017).
Pavri, R. et al. Activation-induced cytidine deaminase targets DNA at sites of RNA polymerase II stalling by interaction with Spt5. Cell 143, 122–133 (2010).
Steensel, Bvan & Furlong, E. E. M. The role of transcription in shaping the spatial organization of the genome. Nat. Rev. Mol. Cell Biol. 20, 327–337 (2019).
Hu, J. et al. Detecting DNA double-stranded breaks in mammalian genomes by linear amplification-mediated high-throughput genome-wide translocation sequencing. Nat. Protoc. 11, 853–871 (2016).
Matthews, A. J., Zheng, S., DiMenna, L. J. & Chaudhuri, J. Regulation of immunoglobulin class-switch recombination: choreography of noncoding transcription, targeted DNA deamination and long-range DNA repair. Adv. Immunol. 122, 1–57 (2014).
Gearhart, P. J., Johnson, N. D., Douglas, R. & Hood, L. IgG antibodies to phosphorylcholine exhibit more diversity than their IgM counterparts. Nature 291, 29–34 (1981).
Vincent-Fabert, C. et al. Genomic deletion of the whole IgH 3′ regulatory region (hs3a, hs1,2, hs3b and hs4) dramatically affects class-switch recombination and Ig secretion to all isotypes. Blood 116, 1895–1898 (2010).
Volpi, S. A. et al. Germline deletion of IgH 3′ regulatory region elements hs 5, 6, 7 (hs5-7) affects B cell-specific regulation, rearrangement and insulation of the Igh locus. J. Immunol. 188, 2556–2566 (2012).
Panchakshari, R. A. et al. DNA double-strand break response factors influence end-joining features of IgH class switch and general translocation junctions. Proc. Natl Acad. Sci. USA 115, 762–767 (2018).
Mesin, L., Ersching, J. & Victora, G. D. Germinal center B-cell dynamics. Immunity 45, 471–482 (2016).
Mayer, C. T. et al. The microanatomic segregation of selection by apoptosis in the germinal center. Science 358, eaao2602 (2017).
Shih, T.-A. Y., Meffre, E., Roederer, M. & Nussenzweig, M. C. Role of BCR affinity in T-cell dependent antibody responses in vivo. Nat. Immunol. 3, 570–575 (2002).
Rouaud, P. et al. The IgH 3′ regulatory region controls somatic hypermutation in germinal center B cells. J. Exp. Med. 210, 1501–1507 (2013).
Morvan, C. L., Pinaud, E., Decourt, C., Cuvillier, A. & Cogné, M. The immunoglobulin heavy-chain locus hs3b and hs4 3′ enhancers are dispensable for VDJ assembly and somatic hypermutation. Blood 102, 1421–1427 (2003).
Hansen, A. S. et al. Distinct classes of chromatin loops revealed by deletion of an RNA-binding region in CTCF. Mol. Cell 76, 395–411 (2019).
Saldaña-Meyer, R. et al. RNA interactions are essential for CTCF-mediated genome organization. Mol. Cell 76, 412–422.e5 (2019).
Garland, W. et al. A functional link between nuclear RNA decay and transcriptional control mediated by the polycomb repressive complex 2. Cell Rep. 29, 1800–1811 (2019).
Hansen, A. S., Amitai, A., Cattoglio, C., Tjian, R. & Darzacq, X. Guided nuclear exploration increases CTCF target search efficiency. Nat. Chem. Biol. 16, 257–266 (2020).
Rao, S. S. P. et al. A 3D map of the human genome at kilobase resolution reveals principles of chromatin looping. Cell 159, 1665–1680 (2014).
Davidson, I. F. et al. Rapid movement and transcriptional re‐localization of human cohesin on DNA. EMBO J. 35, 2671–2685 (2016).
Niehrs, C. & Luke, B. Regulatory R-loops as facilitators of gene expression and genome stability. Nat. Rev. Mol. Cell Biol. 21, 167–178 (2020).
Rowley, M. J. & Corces, V. G. Organizational principles of 3D genome architecture. Nat. Rev. Genet. 19, 789–800 (2018).
Stik, G. et al. CTCF is dispensable for immune cell transdifferentiation but facilitates an acute inflammatory response. Nat. Genet. 52, 655–661 (2020).
Senigl, F. et al. Topologically associated domains delineate susceptibility to somatic hypermutation. Cell Rep. 29, 3902–3915 (2019).
Lim, J. et al. Nuclear proximity of Mtr4 to RNA exosome restricts DNA mutational asymmetry. Cell 169, 523–537 (2017).
Yan, C. T. et al. IgH class switching and translocations use a robust non-classical end-joining pathway. Nature 449, 478–482 (2007).
Boboila, C., Alt, F. W. & Schwer, B. Classical and alternative end-joining pathways for repair of lymphocyte-specific and general DNA double-strand breaks. Adv. Immunol. 116, 1–49 (2012).
Daniel, J. A. & Nussenzweig, A. The AID-induced DNA damage response in chromatin. Mol. Cell 50, 309–321 (2013).
Lionetti, M. et al. A compendium of DIS3 mutations and associated transcriptional signatures in plasma cell dyscrasias. Oncotarget 6, 26129–26141 (2015).
Walker, B. A. et al. Identification of novel mutational drivers reveals oncogene dependencies in multiple myeloma. Blood 132, 587–597 (2018).
Sanz, L. A. & Chédin, F. High-resolution, strand-specific R-loop mapping via S9.6-based DNA–RNA immunoprecipitation and high-throughput sequencing. Nat. Protoc. 14, 1734–1755 (2019).
Jain, S., Ba, Z., Zhang, Y., Dai, H.-Q. & Alt, F. W. CTCF-binding elements mediate accessibility of RAG substrates during chromatin scanning. Cell 174, 102–116 (2018).
Bredemeyer, A. L. et al. DNA double-strand breaks activate a multi-functional genetic program in developing lymphocytes. Nature 456, 819–823 (2008).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Pertea, M. et al. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 33, 290–295 (2015).
Robinson, J. T. et al. Integrative Genomics Viewer. Nat. Biotechnol. 29, 24–26 (2011).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B-cell identities. Mol. Cell 38, 576–589 (2010).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Zhang, Y. et al. Model-based analysis of ChIP–seq (MACS). Genome Biol. 9, R137 (2008).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Durand, N. C. et al. Juicer provides a one-click system for analyzing loop-resolution Hi-C experiments. Cell Syst. 3, 95–98 (2016).
Durand, N. C. et al. Juicebox provides a visualization system for Hi-C contact maps with unlimited zoom. Cell Syst. 3, 99–101 (2016).
Lieberman-Aiden, E. et al. Comprehensive mapping of long-range interactions reveals folding principles of the human genome. Science 326, 289–293 (2009).
Crane, E. et al. Condensin-driven remodelling of X chromosome topology during dosage compensation. Nature 523, 240–244 (2015).
Schirmer, M. et al. Insight into biases and sequencing errors for amplicon sequencing with the Illumina MiSeq platform. Nucleic Acids Res. 43, e37 (2015).
Krzywinski, M. et al. Circos: an information aesthetic for comparative genomics. Genome Res. 19, 1639–1645 (2009).
Acknowledgements
We thank M. Nussenzweig (Rockefeller University) for providing VHB1-8 mice, T. Honjo (Kyoto University) for AID−/− mice, F. Alt (Harvard University) for the LAM-HTGTS protocol, A. Schooley and J. Dekker for help in analyses of insulation scores, B. Sleckman (University of Alabama) for sharing the Abelson cell line system and R. Pavri (IMP, Vienna) for discussions regarding this study. Research in the Basu laboratory is supported by grants to B.L. (EMBO fellowship, ALTF 906-2015) and U.B. (NIAID 1R01AI099195, RO1AI134988 and RO1AI143897), Leukemia & Lymphoma Society, and the Pershing Square Sohn Cancer Research Alliance. This study utilized facilities at Columbia University Irving Medical Center flow cytometry core facility and genome center (P30CA013696).
Author information
Authors and Affiliations
Contributions
U.B., B.L. and J.L. designed the experiments and interpreted the data. B.L. and J.L. performed the experiments. W.Z., Y.C. and J.W. analyzed the bioinformatic data. E.P. and A.N.E. participated in the creation of the Dis3 COIN allele. J.B., C.R.B. and L.W. participated in the experiments during the revisions. U.B. and B.L. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Genetics thanks Yves Denizot, Ralph Stadhouders and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Generation of Dis3COIN mouse model and efficient Dis3 deletion in activated B cells.
a, Dis3 targeted allele includes mCherry and Neo resistance cassettes, inverted Dis3 exon 3, GFP gene, and loxP/FRT sites (top). After FLP-mediated removal of mCherry/Neo cassettes and exon 3 reversion, Dis3COIN allele is functional (middle). Dis3 KO allele after cre-mediated inversion (bottom). b, ESC screening by Southern blot with 3′ or mCherry probes. Clones G3 and F12 were selected for microinjection. Screening was performed one time, each clone tested with different probes. c, Mouse genotyping. Dis3 wt allele generates a band of 369 bp, Dis3COIN allele 489 bp. This gel is representative of our genotyping, performed at least 10 times. d, Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C B cells were stimulated in the presence (+) or in the absence (-) of tamoxifen, and CSR to IgG1 was quantified (3 independent experiments, mean is shown +/- s.e.m., two-tailed paired t-test). e, qPCR quantification of Dis3 allelic inversion on genomic DNA, normalized to the GFP gene. This qPCR is specific of the Dis3 COIN allele before inversion, and shows loss of the Dis3 functional allele after inversion. f, g, and h, RT-qPCR quantification of Dis3, Aicda (AID) mRNAs, and γ1 germline transcript expression on total RNA, normalized to Gapdh. Kinetic in panels e to h was performed one time in triplicate, mean is shown +/- s.e.m., two-tailed unpaired t-test). i, Resting B splenocytes were collected and stimulated with LPS and tamoxifen to inactivate Dis3COIN alleles, IL4 was added at day 1. ChIP, Hi-C, and 3C experiments were performed at early time points. RNA-seq were performed 2 days after IL4 treatment, while cells were harvested at late time-point to study the accumulated DNA junctions from CSR or translocations by LAM-HTGTS.
Extended Data Fig. 2 Accumulation of DIS3-sensitive ncRNAs in B cells isolated from the germinal center in vivo and stimulated in vitro.
a, mRNA expression in vitro and in vivo. RNA-sequencing from Rosacre/+ Dis3C/+ versus Rosacre/+ Dis3C/C in vitro activated B cells (n = 2), and in vivo AIDcre/+ Dis3C/+ versus AIDcre/+ Dis3C/C (n = 2) were analyzed. b, Differential gene expression in DIS3-deficient B cells. RNA-sequencing from Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C activated B cells (n = 2) were analyzed using a hierarchical unsupervised clustering method and results are shown as heat maps. Up-regulated genes are shown in green and down-regulated genes are shown in blue. A total of 160 genes showed differential RNA expression at this early time-point. c to e, Examples of DIS3-sensitive aTSS and antisense RNAs at various genes, including Myc, Cd19, and Cd79a, genes frequently translocated in B cells. f, eRNAs expressed from the Igh 3′RR super-enhancer. g, Example of DIS3-sensitive enhancer RNAs (eRNAs) at super-enhancer, with the active enhancer mark H3K27ac. h, Example of DIS3-sensitive eRNAs at intronic enhancer, from Pvt1 locus, with the active enhancer mark H3K27ac. i, Example of DIS3-sensitive CBE-overlapping RNAs at Fh1 locus. j, Example of DIS3-sensitive CBE-overlapping RNAs downstream the Igk super-enchancer.
Extended Data Fig. 3 Accumulation of DNA-associated RNAs in the absence of DIS3.
a DNA:RNA immunoprecipitation (DRIP). DRIP was performed on Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C activated B cells. DNA was extracted, digested by restriction enzymes, and treated with RNase H for the negative controls before immunoprecipitation using S9.6 antibody. qPCR quantification of DNA:RNA hybrids at Sµ region. DRIP products were quantified directly by qPCR, relative to the input. One experiment performed in duplicate, each dot represents one qPCR value. b to j, DRIP-sequencing: these DRIP products were prepared for deep sequencing. 3 deep-sequencings were performed with similar results. Coding and non-coding transcription determined by RNA-sequencing is shown (‘RNA’ tracks). b, DNA:RNA hybrids accumulation at Sµ region, relative to a. c. Example of DNA:RNA hybrids accumulation at the Bcl6 gene, correlating with the accumulation of intronic ncRNA in DIS3-deficient cells. d, Example of DNA:RNA hybrids accumulation at the Jak2 gene. e, Peak calling was applied to quantify the different peaks corresponding to DNA:RNA hybrids in DRIP sequencing experiments. Quantification is shown in Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C activated B cells (bar graphs show the number of peaks, χ2 two-tailed proportions test). f, Read numbers from the different spike-in controls used in DRIP experiments are shown. g, Distribution of DNA:RNA hybrids per TAD in Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C activated B cells, means are indicated by dashed lines. h to k, Multiple examples of accumulation of DNA:RNA hybrids overlapping CBEs and decreasing CTCF binding in the absence of DIS3.
Extended Data Fig. 4 Genome-wide decrease of CTCF and RAD21 binding in the absence of DIS3.
a and b. CTCF and RAD21 ChIP–seq from Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C activated B cells (biological replicates number two). Common peaks (n = 19,042 and n = 6,873 respectively) between two replicates were used in the heat map, two-tailed Wilcoxon rank-sum test. c. IgG negative control. d and e. CTCF binding and RAD21 localization to CBEs overlapped by RNA are more affected than other CBEs of the genome in the absence of DIS3. f and g. CTCF binding and RAD21 localization to TAD anchor CBEs are more affected than other part of the genome in the absence of DIS3. Panels d to g: box and whiskers plots represent ChIP values (fold changes), bottom and top whiskers represent LQ – 1.5*(UQ-LQ) and UQ + 1.5*(UQ-LQ) where LQ and UQ are lower and upper quartiles, outside interquartile range. Box plots show lower quartile, median and upper quartile, two-tailed Wilcoxon rank-sum test. h. Percentages of CTCF and RAD21 ChIP reads found inside peaks. Total number of reads was similar between Dis3C/+ and Dis3C/C (top), while reads found in peaks were decreased for CTCF and RAD21. Bar graphs show the percentage of reads inside the peaks, χ2 two-tailed proportions test. i. Analysis of ncRNAs and CBE peaks overlap. CTCF occupancy in the B cell genome was determined from control B cells (Dis3C/+) and overlapped with all ncRNAs found in Dis3C/C B cells (1 kb window). The Venn diagram shows 73 CBEs located at the Igκ and 122 CBEs at the Igh loci have overlap ncRNAs. A simulated random distribution demonstrates that the numbers are superior to what would be observed from random genomic overlaps. j. Decreased CTCF binding at Igh V CBEs. IGV tracks show some example of decreased CTCF-binding to VH CBEs where RNAs are accumulated in the absence of DIS3.
Extended Data Fig. 5 Co-transcriptional DNA:RNA hybrids accumulation in DIS3-deficient cells impairs optimal RAD21 localization.
a. Model suggesting impaired cohesin scanning in the presence of DNA:RNA hybrids in Dis3C/C cells. While DIS3-proficient cells resolve DNA:RNA hybrids using DIS3 activity, DIS3-deficient cells accumulate these structures (and associated proteins) which impair cohesin scanning (and/or decreases the processing speed of the cohesin complex) and ultimately perturb loop extrusion. Transcription inhibitors decrease the amount of co-transcriptional DNA:RNA hybrids and potentially restore cohesin positioning, even in the absence of DIS3. RAD21 ChIP–sequencing in activated B cells using transcription inhibitors. Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C cells were treated with IL4 for 24 h and with transcription (TC) inhibitors or DMSO as control for 4 h before fixation (one experiment). b. Number and percentage of RAD21 peaks decreased in the absence of DIS3 and rescued after transcription inhibitors treatments. All peaks with a fold increase >1 after TC inhibitors treatment were considered. c. Genome-wide analysis of RAD21 binding with or without transcription inhibitors in Dis3C/C. Here we focused on RAD21 peaks which were decreased by 2-fold compare to the control Dis3C/+ without treatment. In these conditions 1,012 peaks show efficient rescue. The box and whiskers plots represent these values, the bottom and top whiskers represent the minimum and maximum scores, respectively, outside the interquartile range. The box plots show the lower quartile, median and upper quartile. This experiment was performed one time, two-tailed Wilcoxon rank-sum test. d to i. Multiple examples of RAD21 restoration after transcription inhibitor treatment. IGV tracks show the defect of RAD21 positioning in the absence of DIS3 (Dis3C/+ versus Dis3C/C, grey tracks) while transcription inhibitors restore RAD21 localization even in the absence of DIS3 (blue tracks). Black arrows indicate the RAD21 peaks which are strongly affected by DIS3-deficiency and rescued after transcription inhibitors treatments.
Extended Data Fig. 6 G-rich sequences directly inhibit CTCF/RAD21 binding in the absence of RNA exosome activity.
a. CBEs were cloned into an expression vector containing G-rich regions corresponding to Sµ. Expected phenotypes in the presence (control) or in the absence of RNA exosome (KO) activity are shown. b. We generated Abelson cell lines. After tamoxifen treatment C/C cells inverse Exosc3COIN alleles, inactivating RNA exosome activity and expressing GFP. c. Abelson cell lines were treated with tamoxifen during 4 days, GFP expression was quantified by flow cytometry. Grey histograms show untreated cells and green histograms show tamoxifen-treated cells. d and e. Kinetic of GFP expression and viability in Abelson cell lines, performed 2 times in triplicates with similar results (mean is shown +/- s.e.m.). f. RNA-seq and DRIP-seq tracks showing ncRNAs and DNA:RNA hybrids accumulation at Sµ region in activated B cells (see Fig. 4e and Extended Data Fig. 3b). The G-rich sequence (dotted line region) was cloned into our construct, flanking CBEs on both sides. g. Abelson cell lines (C/C) were transfected and selected for the presence of the construct containing ectopic CBEs. Cells were split in two, as control and tamoxifen-treated, and CTCF and RAD21 ChIP were performed. h. Reproducible defect in RAD21 recruitment at ectopic CBEs. We isolated independent Abelson cell lines (C/C) clones to perform ChIP in the presence (ctrl) or in the absence (KO) of RNA exosome and evaluated RAD21 DNA binding directly by qPCR in these biological replicates (2 independent experiments performed on 3 clones, qPCR were done in triplicates, two-tailed unpaired t-test). The box and whiskers plots represent these values, the bottom and top whiskers represent the minimum and maximum scores, respectively, outside the interquartile range. The box plots show the lower quartile, median and upper quartile. i. Primary B cells were stimulated to perform RNA polymerase II S5 phosphorylated ChIP.
Extended Data Fig. 7 Genome-wide alteration in chromosome organization in DIS3-deficient B cells.
Combined data from 4 independent were used for these analyses. Hi-C data visualization using ‘Juicebox’ and DNA interaction changes on all chromosomes (1 to 19 and chromosome X). Interaction frequencies are calculated by ‘control value divided by observed value (observed/control)’, the relative gained interactions are shown in red and the relative lost interactions are shown in blue. Each square represents a full chromosome, beginning on the top left corner and finishing on the bottom right corner. a. Control KO/wt interactions are shown. An irrelevant KO (located on chromosome 12) was used to evaluate the global impact on genomic organization. Random patterns were obtained implying random gain and loss of interaction equivalent to no change. b. Dis3C/C/Dis3C/+ interactions are shown, with an obvious specific pattern of altered genomic interactions.
Extended Data Fig. 8 Quantification of chromosome alterations in DIS3-deficient B cells.
a and b. ‘Interaction score’: the average log2 fold-change of interactions along each diagonal of Hi-C maps is shown for each chromosome to evaluate the distance-dependent changes in interactions. Each colored curve represents one chromosome, and the black curve shows the average curve. These data are related to Extended Data Fig. 7. a. No differences were detected when dividing control KO by wt interactions. b. DIS3-deficiency induces a clear shift in this score, reflecting the altered patterns shown in Extended Data Fig. 7. c. Insulation scores at TAD boundaries are shown for control KO and wt activated B cells. S: slope of the fitted linear model. 1,224 insulation scores are shown. d. Insulation scores at TAD boundaries are shown for Dis3C/C and Dis3C/+ activated B cells. S: slope of the fitted linear model. 1,224 insulation scores are shown. The Igh 5’ and 3’ ends are shown as red dots. e. APA analysis showing the genome-wide loop accumulation, in control Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C cells. A total of 800 loops were analyzed. f. APA analysis showing the loop accumulation in the top 25% differential loops in control Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C cells. Loop interactions are defective genome-wide in the absence of DIS3, and much pronounced for the top 200 differential loops, as previously described.
Extended Data Fig. 9 Decrease physiological DNA double strand breaks at Igh switch regions, and increase chromosomal translocations at the Myc TAD in the absence of DIS3.
a. LAM-HTGTS from AID−/−, Rosacre/+ Dis3C/+, and Rosacre/+ Dis3C/C activated B cells at Sµ, Sγ3, Sγ1, Sγ2b, and Sε regions with corresponding RNA-seq (combined data from 3 independent experiments). b to f. CRISPR/Cas9-induced DNA breaks and processing at the Myc locus Primary B cells (Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C) were infected during in vitro stimulation with CRISPR/Cas9 retrovirus to induce DNA breaks at the Myc locus. DNA junctions were analyzed by LAM-HTGTS using Myc bait (2 independent experiments with 2 or 3 biological replicates and 3 independent libraries used for sequencing). b. DNA breaks induced by CRISPR/Cas9 at Myc intron 1 and captured using Myc bait. Black arrow indicates the position of CRISPR/Cas9-induced DNA breaks. Myc TAD is outlined by dashed lines. c. Circos plots showing global DNA translocations. d. 2 fold decrease in intra-TAD recombination in the absence of DIS3. The proportion of intra-Myc TAD recombination and inter-TADs translocations is shown (total DNA junctions of 8,122 in Dis3C/+ and 5,529 in Dis3C/C), χ2 two-tailed proportions test. e. NcRNAs and DNA:RNA hybrids accumulation at the Myc locus decrease CTCF/RAD21 binding. RNA-seq data show aTSS and intronic ncRNAs accumulation in Dis3C/C cells, DRIP displays increased DNA:RNA hybrids, while CTCF and RAD21 ChIP-seq reveal decreased CTCF/RAD21 binding. Black arrow indicates CRISPR/Cas9-induced DNA breaks. f. Global analysis of the DNA junctions from Myc LAM-HTGTS, showing length and frequencies of microhomology overlapping bait and prey sequences. All DNA junctions from 3 experiments were combined and the distributions of insertions were evaluated by χ2 two-tailed proportions test (comparing DNA junctions with microhomology length >4 to the total junctions between Rosacre/+ Dis3C/+ and Rosacre/+ Dis3C/C cells).
Extended Data Fig. 10 In vivo somatic hypermutation exploration in Dis3C/C B cells.
a. B splenocytes from one pair of AIDcre/+ Dis3C/+ and AIDcre/+ Dis3C/C mice were stimulated. GFP expression was examined after 4 days. In ex vivo culture conditions AIDcre/+ Dis3C/C cells do not encounter selection and maintain numbers of GFP+ cells. b. RT-qPCR quantification of Dis3 mRNA expression in AIDcre/+ Dis3C/C Peyer’s patches B cells. B220+ GL7+ and B220+ GL7− cells were sorted to show the specific deletion of the Dis3COIN allele in activated B cells (one experiment performed in triplicate, mean is shown +/- s.e.m., two-tailed unpaired t-test). c. RNA-seq showing increased ncRNAs overlapping JH genes in the absence of DIS3 activity during B cell stimulation. Top. RNA-seq tracks encompassing the Eµ intronic enhancer and JH genes. Bottom. RNA-seq tracks showing increased sense transcription (red) and DNA:RNA hybrid accumulation (black) downstream of the JH4 gene in the absence of DIS3 activity (2 RNA-sequencing and 3 DRIP-sequencing) d. Top: Schematic of wt recombined VDJ genes in the context of CBEs. Bottom: VHB1-8KI alleles have different configuration, conserving the major CTCF-binding region IGCR1 (IGCR1 is deleted from the functional allele in physiological conditions). e. DNA mutation analyses of GC (B220+ GL7+) B cells from VHB1-8KI/KI AIDcre/+ Dis3C/+ (n = 3) mice. Tail DNA from the same animals was used as control (n = 2). AID hot-spots are indicated, and C or G mutations refer to the sense DNA strand. f. Quantification of C and G mutation frequencies in GC B cells. 3 pairs of VHB1-8KI/KI AIDcre/+ Dis3C/+ and VHB1-8KI/KI AIDcre/+ Dis3C/C mice were used, analyses were performed at the JH gene from the VHB1-8 allele. χ2 two-tailed proportions tests were used to compare the number of C to T mutations relatively to the total C sequenced, and the number of G to A mutations compared to the total G sequenced.
Supplementary information
Supplementary Information
Supplementary Figs. 1–3 and Supplementary Discussion
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 1
Blots and gels source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Source Data Extended Data Fig. 10
Statistical source data.
Rights and permissions
About this article
Cite this article
Laffleur, B., Lim, J., Zhang, W. et al. Noncoding RNA processing by DIS3 regulates chromosomal architecture and somatic hypermutation in B cells. Nat Genet 53, 230–242 (2021). https://doi.org/10.1038/s41588-020-00772-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-020-00772-0
This article is cited by
-
Primase promotes the competition between transcription and replication on the same template strand resulting in DNA damage
Nature Communications (2024)
-
RNA processing mechanisms contribute to genome organization and stability in B cells
Oncogene (2024)
-
Cohesin maintains replication timing to suppress DNA damage on cancer genes
Nature Genetics (2023)
-
Alternative splicing in multiple myeloma is associated with the non-homologous end joining pathway
Blood Cancer Journal (2023)
-
Noncoding mutations cause super-enhancer retargeting resulting in protein synthesis dysregulation during B cell lymphoma progression
Nature Genetics (2023)