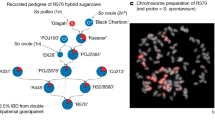
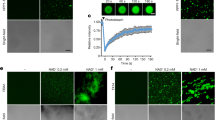
Abstract
Host resistance and fungicide treatments are cornerstones of plant-disease control. Here, we show that these treatments allow sex and modulate parenthood in the fungal wheat pathogen Zymoseptoria tritici. We demonstrate that the Z. tritici–wheat interaction complies with the gene-for-gene model by identifying the effector AvrStb6, which is recognized by the wheat resistance protein Stb6. Recognition triggers host resistance, thus implying removal of avirulent strains from pathogen populations. However, Z. tritici crosses on wheat show that sex occurs even with an avirulent parent, and avirulence alleles are thereby retained in subsequent populations. Crossing fungicide-sensitive and fungicide-resistant isolates under fungicide pressure results in a rapid increase in resistance-allele frequency. Isolates under selection always act as male donors, and thus disease control modulates parenthood. Modeling these observations for agricultural and natural environments reveals extended durability of host resistance and rapid emergence of fungicide resistance. Therefore, fungal sex has major implications for disease control.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barrett, L. G., Thrall, P. H., Burdon, J. J. & Linde, C. C. Life history determines genetic structure and evolutionary potential of host-parasite interactions. Trends Ecol. Evol. 23, 678–685 (2008).
Peterson, P. D., Leonard, K. J., Roelfs, A. P. & Sutton, T. B. Effect of barberry eradication on changes in populations of Puccinia graminis in Minnesota. Plant Dis. 89, 935–940 (2005).
Seidl, M. F. & Thomma, B. P. H. J. Sex or no sex: evolutionary adaptation occurs regardless. BioEssays 36, 335–345 (2014).
Goodwin, S. B. et al. Finished genome of the fungal wheat pathogen Mycosphaerella graminicola reveals dispensome structure, chromosome plasticity, and stealth pathogenesis. PLoS Genet. 7, e1002070 (2011).
Hunter, T., Coker, R. R. & Royle, D. J. The teleomorph stage, Mycosphaerella graminicola, in epidemics of septoria tritici blotch on winter wheat in the UK. Plant Pathol. 48, 51–57 (1999).
Kema, G. H. J., Verstappen, E. C. P., Todorova, M. & Waalwijk, C. Successful crosses and molecular tetrad and progeny analyses demonstrate heterothallism in Mycosphaerella graminicola. Curr. Genet. 30, 251–258 (1996).
Shaw, M. W. & Royle, D. J. Airborne inoculum as a major source of Septoria tritici (Mycosphaerella graminicola) infections in winter wheat crops in the UK. Plant Pathol. 38, 35–43 (1989).
Shaw, M. W. & Royle, D. J. Factors determining the severity of epidemics of Mycosphaerella graminicola (Septoria tritici) on winter wheat in the UK. Plant Pathol. 42, 882–899 (1993).
Chartrain, L., Brading, P. A. & Brown, J. K. M. Presence of the Stb6 gene for resistance to septoria tritici blotch (Mycosphaerella graminicola) in cultivars used in wheat-breeding programmes worldwide. Plant Pathol. 54, 134–143 (2005).
Saintenac, C. et al. Wheat receptor-kinase-like protein Stb6 controls gene-for-gene resistance to fungal pathogen Zymoseptoria tritici. Nat. Genet. https://doi.org/10.1038/s41588-018-0051-x (2018).
Linde, C. C., Zhan, J. & McDonald, B. A. Population structure of Mycosphaerella graminicola: from lesions to continents. Phytopathology 92, 946–955 (2002).
Wittenberg, A. H. J. et al. Meiosis drives extraordinary genome plasticity in the haploid fungal plant pathogen Mycosphaerella graminicola. PLoS One 4, e5863 (2009).
Mehrabi, R. et al. Gα and Gβ proteins regulate the cyclic AMP pathway that is required for development and pathogenicity of the phytopathogen Mycosphaerella graminicola. Eukaryot. Cell 8, 1001–1013 (2009).
Thrall, P. H., Barrett, L. G., Dodds, P. N. & Burdon, J. J. Epidemiological and evolutionary outcomes in gene-for-gene and matching allele models. Front. Plant Sci. 6, 1084 (2016).
Brading, P. A., Verstappen, E. C. P., Kema, G. H. J. & Brown, J. K. M. A gene-for-gene relationship between wheat and Mycosphaerella graminicola, the septoria tritici blotch pathogen. Phytopathology 92, 439–445 (2002).
Brown, J. K. M. & Tellier, A. Plant-parasite coevolution: bridging the gap between genetics and ecology. Annu. Rev. Phytopathol. 49, 345–367 (2011).
Kema, G. H. J. et al. A combined amplified fragment length polymorphism and randomly amplified polymorphism DNA genetic linkage map of Mycosphaerella graminicola, the septoria tritici leaf blotch pathogen of wheat. Genetics 161, 1497–1505 (2002).
Kema, G. H. J., Verstappen, E. C. P. & Waalwijk, C. Avirulence in the wheat septoria tritici leaf blotch fungus Mycosphaerella graminicola is controlled by a single locus. Mol. Plant Microbe Interact. 13, 1375–1379 (2000).
Mirzadi Gohari, A. et al. Effector discovery in the fungal wheat pathogen Zymoseptoria tritici. Mol. Plant Pathol. 16, 931–945 (2015).
Waalwijk, C., Mendes, O., Verstappen, E. C. P., de Waard, M. A. & Kema, G. H. J. Isolation and characterization of the mating-type idiomorphs from the wheat septoria leaf blotch fungus Mycosphaerella graminicola. Fungal Genet. Biol. 35, 277–286 (2002).
Rudd, J. J. et al. Transcriptome and metabolite profiling of the infection cycle of Zymoseptoria tritici on wheat reveals a biphasic interaction with plant immunity involving differential pathogen chromosomal contributions and a variation on the hemibiotrophic lifestyle definition. Plant Physiol. 167, 1158–1185 (2015).
Zhong, Z. et al. A small secreted protein in Zymoseptoria tritici is responsible for avirulence on wheat cultivars carrying the Stb6 resistance gene. New Phytol. 214, 619–631 (2017).
Leonard, K. J. Genetic equilibria in host-pathogen systems. Phytopathology 59, 1858–1863 (1969).
Tellier, A. & Brown, J. K. M. Polymorphism in multilocus host parasite coevolutionary interactions. Genetics 177, 1777–1790 (2007).
Kema, G. H. J. & van Silfhout, C. H. Genetic variation for virulence and resistance in the wheat-Mycosphaerella graminicola pathosystem III. Comparative seedling and adult plant experiments. Phytopathology 87, 266–272 (1997).
Brown, J. K. M. Durable resistance of crops to disease: a Darwinian perspective. Annu. Rev. Phytopathol. 53, 513–539 (2015).
Sierotzki, H., Wullschleger, J. & Gisi, U. Point mutation in cytochrome b gene conferring resistance to strobilurin fungicides in Erysiphe graminis f. sp tritici field isolates. Pestic. Biochem. Physiol. 68, 107–112 (2000).
Fraaije, B. A. et al. Role of ascospores in further spread of QoI-resistant cytochrome b alleles (G143A) in field populations of Mycosphaerella graminicola. Phytopathology 95, 933–941 (2005).
Torriani, S. F. F., Brunner, P. C., McDonald, B. A. & Sierotzki, H. QoI resistance emerged independently at least 4 times in European populations of Mycosphaerella graminicola. Pest Manag. Sci. 65, 155–162 (2009).
Amil, A. F., Heaney, S. P., Stanger, C. & Shaw, M. W. Dynamics of QoI sensitivity in Mycosphaerella fijiensis in Costa Rica during 2000 to 2003. Phytopathology 97, 1451–1457 (2007).
Arango Isaza, R. E. et al. Combating a global threat to a clonal crop: banana black sigatoka pathogen Pseudocercospora fijiensis (synonym Mycosphaerella fijiensis) genomes reveal clues for disease control. PLoS Genet. 12, e1005876 (2016).
Price, C. L., Parker, J. E., Warrilow, A. G. S., Kelly, D. E. & Kelly, S. L. Azole fungicides: understanding resistance mechanisms in agricultural fungal pathogens. Pest Manag. Sci. 71, 1054–1058 (2015).
Diaz-Trujillo, C. et al. A new mechanism for reduced sensitivity to demethylation-inhibitor fungicides in the fungal banana black Sigatoka pathogen Pseudocercospora fijiensis. Mol. Plant Pathol. https://doi.org/10.1111/mpp.12637 (2017).
O’Gorman, C. M., Fuller, H. & Dyer, P. S. Discovery of a sexual cycle in the opportunistic fungal pathogen Aspergillus fumigatus. Nature 457, 471–474 (2009).
Verweij, P. E., Chowdhary, A., Melchers, W. J. G. & Meis, J. F. Azole resistance in Aspergillus fumigatus: can we retain the clinical use of mold-active antifungal azoles? Clin. Infect. Dis. 62, 362–368 (2016).
Annone, J. Cultural types of Septoria tritici Rob. ex Desm.: Occurrence in Washington, Stability and Virulence. MSc thesis, Washington State University (1984).
Alexopoulos, C. J. Introductory Mycology. (John Wiley & Sons, New York, 1962).
Kema, G. H. J., Yu, D. Z., Rijkenberg, F. H. J., Shaw, M. W. & Baayen, R. P. Histology of the pathogenesis of Mycosphaerella graminicola in wheat. Phytopathology 86, 777–786 (1996).
Kema, G. H. J., Sayoud, R., Annone, J. G. & Van Silfhout, C. H. Genetic variation for virulence and resistance in the wheat-Mycosphaerella graminicola pathosystem. II. Analysis of interactions between pathogen isolates and host cultivars. Phytopathology 86, 213–220 (1996).
Cha, R. S., Zarbl, H., Keohavong, P. & Thilly, W. G. Mismatch amplification mutation assay (MAMA): application to the c-H-ras gene. PCR Methods Appl. 2, 14–20 (1992).
Kema, G. H. J. et al. Genetic variation for virulence and resistance in the wheat-Mycosphaerella graminicola pathosystem. I. Interactions between pathogen isolates and host cultivars. Phytopathology 86, 200–212 (1996).
Zadoks, J. C., Chang, T. T. & Konzak, C. F. A decimal code for the growth stages of cereals. Weed Res. 14, 415–421 (1974).
Courtois, B. et al. Genome-wide association mapping of root traits in a japonica rice panel. PLoS One 8, e78037 (2013).
Morgulis, A. et al. Database indexing for production MegaBLAST searches. Bioinformatics 24, 1757–1764 (2008).
Hahne, F. et al. Gviz: Plotting Data and Annotation Information Along Genomic Coordinates. R package v.1 (2013).
Van Ooijen, J.W. JoinMap4, Software for the Calculation of Genetic Linkage Maps in Experimental Populations (Kyazma, 2006).
Holt, C. & Yandell, M. MAKER2: an annotation pipeline and genome-database management tool for second-generation genome projects. BMC Bioinformatics 12, 491 (2011).
Johnson, A. D. et al. SNAP: a web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics 24, 2938–2939 (2008).
Stanke, M., Steinkamp, R., Waack, S. & Morgenstern, B. AUGUSTUS: a web server for gene finding in eukaryotes. Nucleic Acids Res 32, W309–W312 (2004).
Lukashin, A. V. & Borodovsky, M. GeneMark.hmm: new solutions for gene finding. Nucleic Acids Res. 26, 1107–1115 (1998).
Kellner, R. et al. Expression profiling of the wheat pathogen Zymoseptoria tritici reveals genomic patterns of transcription and host-specific regulatory programs. Genome Biol. Evol. 6, 1353–1365 (2014).
Trapnell, C., Pachter, L. & Salzberg, S. L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009).
Trapnell, C. et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 28, 511–515 (2010).
Lee, E. et al. Web Apollo: a web-based genomic annotation editing platform. Genome Biol. 14, R93 (2013).
Palma-Guerrero, J. et al. Comparative transcriptomic analyses of Zymoseptoria tritici strains show complex lifestyle transitions and intraspecific variability in transcription profiles. Mol. Plant Pathol. 17, 845–859 (2016).
Emanuelsson, O., Brunak, S., von Heijne, G. & Nielsen, H. Locating proteins in the cell using TargetP, SignalP and related tools. Nat. Protoc. 2, 953–971 (2007).
Zdobnov, E. M. & Apweiler, R. InterProScan: an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17, 847–848 (2001).
Barratt, R. W., Johnson, G. B. & Ogata, W. N. Wild-type and mutant stocks of Aspergillus nidulans. Genetics 52, 233–246 (1965).
Zwiers, L. H. & De Waard, M. A. Efficient Agrobacterium tumefaciens-mediated gene disruption in the phytopathogen Mycosphaerella graminicola. Curr. Genet. 39, 388–393 (2001).
Mehrabi, R., Zwiers, L. H., de Waard, M. A. & Kema, G. H. J. MgHog1 regulates dimorphism and pathogenicity in the fungal wheat pathogen Mycosphaerella graminicola. Mol. Plant Microbe Interact. 19, 1262–1269 (2006).
Sambrook, J. & Russell, D. W. Purification of nucleic acids by extraction with phenol:chloroform. CSH Protoc. 2006, pdb.prot4455 (2006).
Keon, J. et al. Transcriptional adaptation of Mycosphaerella graminicola to programmed cell death (PCD) of its susceptible wheat host. Mol. Plant Microbe Interact. 20, 178–193 (2007).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 3, 1101–1108 (2008).
Ware, S.B. Aspects of Sexual Reproduction in Mycosphaerella Species on Wheat and Barley: Genetic Studies on Specificity, Mapping, and Fungicide Resistance. PhD thesis, Wageningen University (2006).
Acknowledgements
We acknowledge financial support from the Sixth EU Framework Programme (BioExploit-EU FP6) Food Quality and Safety priority (contract no. 513959 to G.H.J.K. and E.C.P.V.); the Dutch Ministry of Agriculture, Nature and Food Quality; Bayer CropScience; Monsanto’s Beachell-Borlaug International Scholars Program (3340030501 to L.A.); the L’Oréal-UNESCO For Women in Science Fellowship (ERI/RPO/PPF/CDC/10.299 to S.B.M.); the Consejo Nacional de Ciencia y Tecnología, Mexico (CONACyT, no. 87781 to C.D.-T.); the Netherlands Organization for Scientific Research (NWO-VENI 863.15.005 to M.F.S.; NWO-VICI 865.11.003 to B.P.H.J.T.); and the Dioraphte Foundation (14.03.01.00 to H.J.G.M. and G.H.J.K.). We thank O. Mendes, I. de Vries, and C. van Schaik for general support, and B. A. McDonald (ETH, Plant Pathology, Zürich, Switzerland) for sharing unpublished results.
Author information
Authors and Affiliations
Contributions
G.H.J.K., P.J.G.M.d.W., B.P.H.J.T., M.F.S., M.A.d.W., and C.W. designed the study. S.B.W., S.B.M., C.D.-T., E.Z., L.A., and E.C.P.V. performed the crossing assays and collected and analyzed the genotypic and phenotypic data. T.A.J.v.d.L., H.J.S., and L.A. generated the genetic linkage maps, and H.J.S. and L.A. fine mapped AvrStb6. L.A., A.M.G., H.A.Y.G., R.M., and H.J.G.M. performed AvrStb6 candidate analyses and additional genome analyses, and A.M.G. and L.A. performed the functional analysis of AvrStb6. H.A.Y.G. and M.F.S. analyzed the genomic sequences and reannotated the IPO323 and IPO94269 genome sequence. G.H.J.K. and F.v.d.B. conceptualized the EPP model, and F.v.d.B., R.M.-S., V.A.-C., and J.H. mathematically developed the EPP model. G.H.J.K. wrote the paper with substantial input from S.B.W., B.P.H.J.T., H.J.G.M., M.F.S., F.v.d.B., and J.H. G.H.J.K. coordinated the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–18, Supplementary Note and Supplementary Tables 1–3 and 5–8
Supplementary Table
Supplementary Table 4
Rights and permissions
About this article
Cite this article
Kema, G.H.J., Mirzadi Gohari, A., Aouini, L. et al. Stress and sexual reproduction affect the dynamics of the wheat pathogen effector AvrStb6 and strobilurin resistance. Nat Genet 50, 375–380 (2018). https://doi.org/10.1038/s41588-018-0052-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-018-0052-9
This article is cited by
-
The Egyptian wheat cultivar Gemmeiza-12 is a source of resistance against the fungus Zymoseptoria tritici
BMC Plant Biology (2024)
-
Quantitative pathogenicity and host adaptation in a fungal plant pathogen revealed by whole-genome sequencing
Nature Communications (2024)
-
Unlocking the story of resistance to Zymoseptoria tritici in Tunisian old durum wheat germplasm based on population structure analysis
BMC Genomics (2023)
-
Investigation on TOX2 cluster genes of the fungal wheat pathogen Zymoseptoria tritici
European Journal of Plant Pathology (2023)
-
Deciphering resistance to Zymoseptoria tritici in the Tunisian durum wheat landrace accession ‘Agili39’
BMC Genomics (2022)