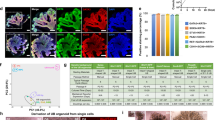
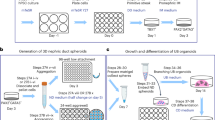
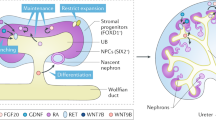
Abstract
Directed differentiation of human pluripotent stem cells (hPSCs) into functional ureteric and collecting duct (CD) epithelia is essential to kidney regenerative medicine. Here we describe highly efficient, serum-free differentiation of hPSCs into ureteric bud (UB) organoids and functional CD cells. The hPSCs are first induced into pronephric progenitor cells at 90% efficiency and then aggregated into spheres with a molecular signature similar to the nephric duct. In a three-dimensional matrix, the spheres form UB organoids that exhibit branching morphogenesis similar to the fetal UB and correct distal tip localization of RET expression. Organoid-derived cells incorporate into the UB tips of the progenitor niche in chimeric fetal kidney explant culture. At later stages, the UB organoids differentiate into CD organoids, which contain >95% CD cell types as estimated by single-cell RNA sequencing. The CD epithelia demonstrate renal electrophysiologic functions, with ENaC-mediated vectorial sodium transport by principal cells and V-type ATPase proton pump activity by FOXI1-induced intercalated cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The scRNA-sequencing data from UB organoid differentiation are available at the Gene Expression Omnibus under accession number GSE206153. The RNA-seq data from microdissected mouse collecting ducts were download from the RNA-seq data from the Mouse Renal Epithelial Cell Atlas (https://esbl.nhlbi.nih.gov/MRECA/Nephron/).
Code availability
Relevant code will be made available upon reasonable request.
References
Kim, J., Koo, B. K. & Knoblich, J. A. Human organoids: model systems for human biology and medicine. Nat. Rev. Mol. Cell Biol. 21, 571–584 (2020).
Lehmann, R. et al. Human organoids: a new dimension in cell biology. Mol. Biol. Cell 30, 1129–1137 (2019).
McMahon, A. P. Development of the mammalian kidney. Curr. Top. Dev. Biol. 117, 31–64 (2016).
Takasato, M. et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature 526, 564–568 (2015).
Morizane, R. et al. Nephron organoids derived from human pluripotent stem cells model kidney development and injury. Nat. Biotechnol. 33, 1193–1200 (2015).
Taguchi, A. & Nishinakamura, R. Higher-order kidney organogenesis from pluripotent stem cells. Cell Stem Cell 21, 730–746 (2017).
Kuraoka, S. et al. PKD1-dependent renal cystogenesis in human induced pluripotent stem cell-derived ureteric bud/collecting duct organoids. J. Am. Soc. Nephrol. 31, 2355–2371 (2020).
Zeng, Z. et al. Generation of patterned kidney organoids that recapitulate the adult kidney collecting duct system from expandable ureteric bud progenitors. Nat. Commun. 12, 3641 (2021).
Mae, S. I. et al. Expansion of human ipsc-derived ureteric bud organoids with repeated branching potential. Cell Rep. 32, 107963 (2020).
Howden, S. E. et al. Plasticity of distal nephron epithelia from human kidney organoids enables the induction of ureteric tip and stalk. Cell Stem Cell 28, 671–684 (2021).
Bens, M. et al. Corticosteroid-dependent sodium transport in a novel immortalized mouse collecting duct principal cell line. J. Am. Soc. Nephrol. 10, 923–934 (1999).
Prie, D. et al. Role of adenosine on glucagon-induced cAMP in a human cortical collecting duct cell line. Kidney Int. 47, 1310–1318 (1995).
Fejes-Toth, G. & Naray-Fejes-Toth, A. Differentiation of renal beta-intercalated cells to alpha-intercalated and principal cells in culture. Proc. Natl Acad. Sci. USA 89, 5487–5491 (1992).
Qiao, J., Sakurai, H. & Nigam, S. K. Branching morphogenesis independent of mesenchymal–epithelial contact in the developing kidney. Proc. Natl Acad. Sci. USA 96, 7330–7335 (1999).
Grote, D., Souabni, A., Busslinger, M. & Bouchard, M. Pax 2/8-regulated Gata3 expression is necessary for morphogenesis and guidance of the nephric duct in the developing kidney. Development 133, 53–61 (2006).
Barak, H., Rosenfelder, L., Schultheiss, T. M. & Reshef, R. Cell fate specification along the anterior-posterior axis of the intermediate mesoderm. Dev. Dyn. 232, 901–914 (2005).
Loh, K. M. et al. Mapping the pairwise choices leading from pluripotency to human bone, heart, and other mesoderm cell types. Cell 166, 451–467 (2016).
Ornitz, D. M. & Itoh, N. The fibroblast growth factor signaling pathway. Wiley Interdiscip. Rev. Dev. Biol. 4, 215–266 (2015).
Perantoni, A. O. et al. Inactivation of FGF8 in early mesoderm reveals an essential role in kidney development. Development 132, 3859–3871 (2005).
Warga, R. M., Mueller, R. L., Ho, R. K. & Kane, D. A. Zebrafish Tbx16 regulates intermediate mesoderm cell fate by attenuating Fgf activity. Dev. Biol. 383, 75–89 (2013).
Mae, S. I. et al. Generation of branching ureteric bud tissues from human pluripotent stem cells. Biochem. Biophys. Res. Commun. 495, 954–961 (2018).
Bohnenpoll, T. et al. Tbx18 expression demarcates multipotent precursor populations in the developing urogenital system but is exclusively required within the ureteric mesenchymal lineage to suppress a renal stromal fate. Dev. Biol. 380, 25–36 (2013).
Attia, L., Schneider, J., Yelin, R. & Schultheiss, T. M. Collective cell migration of the nephric duct requires FGF signaling. Dev. Dyn. 244, 157–167 (2015).
Atsuta, Y. & Takahashi, Y. FGF8 coordinates tissue elongation and cell epithelialization during early kidney tubulogenesis. Development 142, 2329–2337 (2015).
Sanchez-Ferras, O. et al. A coordinated progression of progenitor cell states initiates urinary tract development. Nat. Commun. 12, 2627 (2021).
Pohl, M., Stuart, R. O., Sakurai, H. & Nigam, S. K. Branching morphogenesis during kidney development. Annu. Rev. Physiol. 62, 595–620 (2000).
Yuri, S., Nishikawa, M., Yanagawa, N., Jo, O. D. & Yanagawa, N. In vitro propagation and branching morphogenesis from single ureteric bud cells. Stem Cell Rep. 8, 401–416 (2017).
Vega, Q. C., Worby, C. A., Lechner, M. S., Dixon, J. E. & Dressler, G. R. Glial cell line-derived neurotrophic factor activates the receptor tyrosine kinase RET and promotes kidney morphogenesis. Proc. Natl Acad. Sci. USA 93, 10657–10661 (1996).
Michos, O. et al. Kidney development in the absence of Gdnf and Spry1 requires Fgf10. PLoS Genet. 6, e1000809 (2010).
Lu, B. C. et al. Etv4 and Etv5 are required downstream of GDNF and Ret for kidney branching morphogenesis. Nat. Genet. 41, 1295–1302 (2009).
Bush, K. T. et al. TGF-β superfamily members modulate growth, branching, shaping, and patterning of the ureteric bud. Dev. Biol. 266, 285–298 (2004).
Maeshima, A., Vaughn, D. A., Choi, Y. & Nigam, S. K. Activin A is an endogenous inhibitor of ureteric bud outgrowth from the Wolffian duct. Dev. Biol. 295, 473–485 (2006).
Michos, O. et al. Reduction of BMP4 activity by gremlin 1 enables ureteric bud outgrowth and GDNF/WNT11 feedback signalling during kidney branching morphogenesis. Development 134, 2397–2405 (2007).
Chi, X. et al. Ret-dependent cell rearrangements in the Wolffian duct epithelium initiate ureteric bud morphogenesis. Dev. Cell 17, 199–209 (2009).
Costantini, F. GDNF/Ret signaling and renal branching morphogenesis: from mesenchymal signals to epithelial cell behaviors. Organogenesis 6, 252–262 (2010).
Tsujimoto, H. et al. A modular differentiation system maps multiple human kidney lineages from pluripotent stem cells. Cell Rep. 31, 107476 (2020).
Uchimura, K., Wu, H., Yoshimura, Y. & Humphreys, B. D. Human pluripotent stem cell-derived kidney organoids with improved collecting duct maturation and injury modeling. Cell Rep. 33, 108514 (2020).
Unbekandt, M. & Davies, J. A. Dissociation of embryonic kidneys followed by reaggregation allows the formation of renal tissues. Kidney Int. 77, 407–416 (2010).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587 (2021).
Lake, B. B. et al. An atlas of healthy and injured cell states and niches in the human kidney. Preprint at https://www.biorxiv.org/content/10.1101/2021.07.28.454201v1 (2021).
Kleyman, T. R. & Cragoe, E. J. Jr. Amiloride and its analogs as tools in the study of ion transport. J. Membr. Biol. 105, 1–21 (1988).
Chen, L., Chou, C. L. & Knepper, M. A. A comprehensive map of mRNAs and their isoforms across All 14 renal tubule segments of mouse. J. Am. Soc. Nephrol. 32, 897–912 (2021).
Werth, M. et al. Transcription factor TFCP2L1 patterns cells in the mouse kidney collecting ducts. eLife 6, e24265 (2017).
Park, J. et al. Single-cell transcriptomics of the mouse kidney reveals potential cellular targets of kidney disease. Science 360, 758–763 (2018).
Blomqvist, S. R. et al. Distal renal tubular acidosis in mice that lack the forkhead transcription factor Foxi1. J. Clin. Invest. 113, 1560–1570 (2004).
Toka, H. R., Toka, O., Hariri, A. & Nguyen, H. T. Congenital anomalies of kidney and urinary tract. Semin. Nephrol. 30, 374–386 (2010).
Kuure, S. & Sariola, H. Mouse models of congenital kidney anomalies. Adv. Exp. Med. Biol. 1236, 109–136 (2020).
Shah, M. M. et al. The instructive role of metanephric mesenchyme in ureteric bud patterning, sculpting, and maturation and its potential ability to buffer ureteric bud branching defects. Am. J. Physiol. Renal Physiol. 297, F1330–F1341 (2009).
Schwartz, G. J. et al. Acid incubation reverses the polarity of intercalated cell transporters, an effect mediated by hensin. J. Clin. Invest. 109, 89–99 (2002).
Schwartz, G. J., Barasch, J. & Al-Awqati, Q. Plasticity of functional epithelial polarity. Nature 318, 368–371 (1985).
Christensen, B. M. et al. Changes in cellular composition of kidney collecting duct cells in rats with lithium-induced NDI. Am. J. Physiol. Cell Physiol. 286, C952–C964 (2004).
Davies, J. A., Unbekandt, M., Ineson, J., Lusis, M. & Little, M. H. Dissociation of embryonic kidney followed by re-aggregation as a method for chimeric analysis. Methods Mol. Biol. 886, 135–146 (2012).
Acknowledgements
We thank members of the Bonventre and Valerius laboratories for reagents and feedback related to this project, especially M. Zarei for assistance in electrophysiology experiments. We thank L. Palmer (Weill Cornell Medicine) for kindly providing purified antibodies to ENaC subunits. We appreciate the outstanding technical expertise from both the Single Cell Core and the BioPolymers Facility at Harvard Medical School for their assistance in completing the scRNA-seq experiments. We also thank J. Thiagarajah and W. Lencer (Boston Children’s Hospital) and the Harvard Digestive Disease Center (P30DK034854) for access and assistance with the Ussing chamber. This work is supported by grants from the National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases and National Institutes of Health/National Center for Advancing Translational Sciences (UH3 TR002155 and R37 DK39773, to J.V.B) and a grant from the University of Virginia Pediatric Center of Excellence Pilot and Feasibility Program (P50 DK096373). K.W.M. was also supported by National Institutes of Health training grant T32 DK00772, and K.W.M. and A.B.P. were supported by grants from the Benjamin J. Lipps Fellowship from the American Society of Nephrology. M.S. was supported by a scholarship from West China School of Medicine/West China Hospital of Sichuan University. W.Z. was supported by a scholarship from Zhongshan Hospital of Fudan University. BioRender software was used in the production of Fig. 3a.
Author information
Authors and Affiliations
Contributions
M.S., K.W.M. and J.V.B. conceived the study and experimental design, performed and analyzed experiments and co-wrote the manuscript. A.B.P. designed and performed experiments related to transepithelial physiology studies. W.Z. and L.E. generated the GATA3 reporter cell line and FOXI1 gain-of-function constructs. M.T.V. conceived and performed fetal kidney explant studies. J.V.B. and K.W.M. acquired funding for this work.
Corresponding authors
Ethics declarations
Competing interests
J.V.B. is an inventor on kidney organoid patents assigned to Mass General Brigham and is co-founder and holds equity in Goldfinch Bio. K.W.M. has kidney organoid patents pending. J.V.B.ʼs interests were reviewed and are managed by Brigham and Women’s Hospital and Mass General Brigham in accordance with their conflict of interest policies. The other authors have no competing financial interests to declare.
Peer review
Peer review information
Nature Biotechnology thanks Cristina Cebrian and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Generation of GATA3-mScarlet reporter allele.
Schematic representation depicting targeting scheme for CRISPR/Cas9-mediated knock-in of mScarlet into human GATA3 locus in H9 hESC line. The donor vector containing P2A-mScarlet and Hygro resistance cassette with flanking homology arms was co-transfected into cells with plasmid co-expressing Cas9 and gRNA targeting GATA3 stop codon.
Extended Data Fig. 2 Efficient induction of pronephric IM cells at day 3.
a, Brightfield micrographs demonstrating appearance of undifferentiated hESCs at day 0, as well cultures after 24 and 30 hours exposure to primitive streak-inducing factors. At 24 hours, there was still significant colony-like morphology consistent with incomplete induction of mesendoderm cells, but after six additional hours the colonies were nearly completely dissociated into single, mesenchymal-like cells. b, Quantification of IF staining for TBXT (as shown in Fig. 1b) revealed >95% efficiency after 30 hours exposure. n = 6 quantified fields from three independent replicates. c, Timecourse qPCR analysis corresponding to Fig. 1c. The primitive streak marker MIXL1 was maximally expressed at day 1 and was subsequently quickly down-regulated, while IM markers OSR1, HOXB7, HNF1B, and SOX9 were increased by day 3. n = 3 independent biological replicates per timepoint. d, Efficient specification and expression of pronephric IM genes at day 3 was dependent on the combinatorial effects of FGF2, RA, TGFβ inhibition (A83-01), and BMP inhibition (LDN193189) during days 1-3. n = 3 independent biological replicates per condition. e, In the pronephric IM cultures at days 3 and 7, there was low level of expression of posterior IM markers (WT1, SIX2, EYA1, and HOXA11), which were derived from a differentiation protocol for inducing metanephric progenitor cells. *, p < 0.05; **p < 0.005; two-tailed Student’s t-test individually comparing day 3 and day 7 against the day 9 posterior IM samples; n = 4 biological replicates, data representative of 2 independent experiments. Specifically p-values for comparisons using day 3 were 0.005, 0.006, 0.0005, and 0.017 for WT1, SIX2, EYA1, and HOXA11, respectively; at day 7, they were 0.007, 0.007, 0.0002, and 0.015. f, From days 1-3, the TGFβ inhibitor A8301 was required for suppression of definitive endoderm (SOX17) fate, whereas BMP inhibition with LDN193189 inhibited formation of lateral plate mesoderm (FOXF1). *p < 0.005; two-tailed Student’s t-test; n = 3 biological replicates, data representative of 2 independent experiments. Scale bar 200 μm (a). Column and error bars represent mean and standard deviation, respectively.
Extended Data Fig. 3 Characterization of nephric duct spheroids.
a, Pronephric IM cells aggregated at day 3 efficiently formed numerous compact spheroids that maintained high levels of GATA3 expression over the course of their development. b, When either FGF2 or FGF8 was used in place of FGF9 from days 3-5, the spheres were more loosely organized and exhibited lower expression of GATA3. c, Micrograph of a small region of nephric duct progenitors plated on a patterned microwell, demonstrating high degree of uniformity of size, shape, and structure of the spheroids. d, From days 4-7, the spheres maintained expression of early pronephric transcription factors LHX1 and HOXB7, and they also gradually acquired expression of nephric duct markers RET and EMX2. e, Over these several days of culture, qPCR analysis showed developmental increase in nephric duct genes RET, WNT11, WNT9B, and EMX2. n = 3 independent biological replicates per timepoint. f-g, Heatmap and expression plots representing scRNA-seq data from day 7 revealed high expression of nephric duct markers in clusters 2 and 3, with complementary expression of stromal lineage genes in clusters 0 and 1 (in reference to UMAP in Fig. 1f). h, IF staining confirmed a high level of WT1-positivity in nephric duct lineage-negative cells at day 7. i, The differentiation of off-target lineages was not observed in the scRNA-seq dataset. Scale bars, 500 μm (a), 100 μm (b), 400 μm (c), 50 μm (d and h). Column and error bars represent mean and standard deviation, respectively.
Extended Data Fig. 4 UB organoid differentiation protocol is efficient when using hiPSCs.
a, Using the hiPSC line BJFF.6, a 30 hour exposure during the first stage of differentiation was also required for the primitive streak phenotype. b, AIM transcription factors PAX2, GATA3, LHX1, and HOXB7 were induced with very high efficiency by day 3. c, The hiPSC-derived AIM exhibited efficient formation of nephric duct spheroids in 96-well low attachment plates. The spheroids then underwent similar molecular and morphological development compared to those derived from hESCs as shown in Fig. 1. d, Nephric duct spheroids efficiently grew into branched UB organoids. e-f, hiPSC-derived UB organoids exhibited tip-stalk patterning and spontaneously formed differentiated AQP2-positive principal cells. Scale bars, 50 μm (a-d) and 100 μm (e-f).
Extended Data Fig. 5 Branching morphogenesis in UB organoids.
a, After embedding in 3-D matrix at day 7, the UB organoids exhibited several rounds of iterative branching during the first week of culture. The stereomicrograph of day 11 UB organoid demonstrates multiple terminal branching events, with each generation uniquely colored: 1=green, 2=red, 3=blue, 4=orange. b, A characteristic terminal bifurcation was demonstrated in the time course of micrographs. c, At later stages, such as day 18, branching was slowed and instead the distal UB tips formed enlarged knobs that more rarely underwent further cleavage events. d-e, Wholemount IF staining at days 9, 11, and 14 as shown in Fig. 2c with separation of channels. f, Expression of the transcription factor HNF1B increased between days 9 and 14. g, From day 7, the UB organoids showed a gradual decline in the tip marker WNT11 and a corresponding increase in the medullary stalk marker WNT7B. WNT9B, which is more broadly expressed in the stalk components, was maintained at relatively stable levels. n = 3 independent biological replicates per timepoint. Scale bars, 50 μm (a-c) and 200 μm (d-f). Column and error bars represent mean and standard deviation, respectively.
Extended Data Fig. 6 UB progenitor cells participate in niche interactions in chimeric explants but not with hPSC-derived metanephric cells.
a, Cells from induced UB organoids and metanephric kidney organoids were dissociated at day 7, mixed and reaggregated, and cultured as spheres in suspension. Within two days (at day 9) the GATA3/KRT8-expressing UB cells had formed an epithelial network in the inner portion of the organoid. Metanephric cells (PAX2 only) were differentiating around the periphery of the structure in close association with the UB cells. However, we did not observe formation of capping mesenchyme structures or branching within the UB epithelium. b, Brightfield image with visualization of the UB epithelium using GATA3-mScarlet reporter confirmed the absence of significant branching morphogenesis. c, In chimeric explants with mouse fetal kidneys, the human induced UB cells (indicated by human nuclear antigen detection) incorporated into the UB tip (Cdh1-positive) at a high frequency, but never into the surrounding metanephric progenitors (Wt1 and Six2). Shown are six representative examples of progenitor niches that contained human cells. Scale bars, 200 μm (a and b) and 20 μm (c).
Extended Data Fig. 7 Characterization of CD organoids using scRNA-seq.
a, UB and CD transcription factors were abundant in clusters 0-4 identified in Fig. 3e, which contained >97% of the sequenced cells (b). c, Expression of markers associated with other differentiated nephron segments, including podocytes, proximal tubule, thick ascending limb of Henle’s loop (TAL), and distal convoluted tubule (DCT), was largely absent in the dataset. d, Mapping prediction scores from Azimuth analysis were notably higher in clusters 0-2, which contained the more differentiated principal cell populations, while the scores were fairly low in clusters 3 and 4. e, Similarly, the prediction scores for cells that mapped to alternative tubular fates (found almost exclusively in clusters 3 and 4) were generally much worse than those that mapped to collecting duct. f, UMAP of clusters 0-4 following random downsampling to 500 total cells that were used in Monocle trajectory analysis. g, The predicted lineage trajectory plotted by pseudotime, which corresponds to Fig. 3i.
Extended Data Fig. 8 Amiloride-sensitive sodium conductance in two-dimensional culture.
a, Tables showing raw data from voltmeter experiments using hESC-derived collecting duct cells. n.d., not done. b-c, Transepithelial voltage and resistance measurements in response to varying concentrations, including both pre- and post-amiloride treatment. Data shown (n = 4 per dose) are representative of at least three independent experiments. d, Quantification of RNA-seq expression levels of potassium channels from microdissected mouse CD segments42, including connecting tubule (CNT), cortical CD (CCD), outer medullary CD (OMCD) and inner medullary CD (IMCD). Kcnj1 and Kcnj10 were absent in the IMCD, while Kcnj13 and Kcnj16 were expressed in all CD regions. n = 7 independent biological replicates per segment. e, In day 18 CD organoids, only KCNJ16 was expressed at high levels in the scRNA-seq dataset. Column and error bars represent mean and standard deviation, respectively.
Extended Data Fig. 9 CD organoids resemble the IMCD with respect to IC differentiation.
a, Region-specific expression from the mouse CD42 revealed absence of IC-related genes (Atp6v1g3, Foxi1, and Tfcp2l1) in the IMCD, and markers of transitional PC/ICs were either reduced (Sec23b, Parm1) or absent (Syt7) in the IMCD. n = 3 independent biological replicates per timepoint. b, Expression in day 18 CD organoids was comparable to the mouse IMCD, with absence of ATP6V1G3 and FOXI1 and low level of TFCP2L1. The transitional cell gene SEC23B was expressed throughout the principal clusters, but neither PARM1 nor SYT7 were abundant. Column and error bars represent mean and standard deviation, respectively.
Supplementary information
Supplementary Information
Supplementary Tables 1–3
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, M., McCracken, K.W., Patel, A.B. et al. Human ureteric bud organoids recapitulate branching morphogenesis and differentiate into functional collecting duct cell types. Nat Biotechnol 41, 252–261 (2023). https://doi.org/10.1038/s41587-022-01429-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-022-01429-5
This article is cited by
-
Calcium signalling and transport in the kidney
Nature Reviews Nephrology (2024)
-
Replacing renal function using bioengineered tissues
Nature Reviews Bioengineering (2023)
-
How organoids are advancing the understanding of chronic kidney disease
Nature (2023)
-
Directed differentiation of ureteric bud and collecting duct organoids from human pluripotent stem cells
Nature Protocols (2023)
-
Derivation of collecting duct organoids
Nature Reviews Nephrology (2022)