Abstract
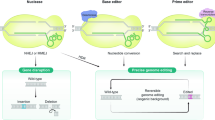
High-throughput functional characterization of genetic variants in their endogenous locus has so far been possible only with methods that rely on homology-directed repair, which are limited by low editing efficiencies. Here, we adapted CRISPR prime editing for high-throughput variant classification and combined it with a strategy that allows for haploidization of any locus, which simplifies variant interpretation. We demonstrate the utility of saturation prime editing (SPE) by applying it to the NPC intracellular cholesterol transporter 1 gene (NPC1), mutations in which cause the lysosomal storage disorder Niemann–Pick disease type C. Our data suggest that NPC1 is very sensitive to genetic perturbation, with 410 of 706 assayed missense mutations being classified as deleterious, and that the derived function score of variants is reflective of diverse molecular defects. We further applied our approach to the BRCA2 gene, demonstrating that SPE is translatable to other genes with an appropriate cellular assay. In sum, we show that SPE allows for efficient, accurate functional characterization of genetic variants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Next-generation sequencing data have been deposited to the NCBI Sequence Read Archive database under accession PRJNA728726. All function scores and corresponding classifications can be found in Supplementary Tables 1–3. The protein subgrouping underlying Fig. 4d is found in Supplementary Table 7. CADD scores underlying the plots in Figs. 4b,c and 5g are found in Supplementary Tables 1 and 3. Source data are provided with this paper.
Code availability
The custom Python scripts used in determining the function score and classification of each variant are available upon request.
References
Cooper, G. M. Parlez-vous VUS? Genome Res. 25, 1423–1426 (2015).
Starita, L. M. et al. Variant interpretation: functional assays to the rescue. Am. J. Hum. Genet. 101, 315–325 (2017).
Fowler, D. M. & Fields, S. Deep mutational scanning: a new style of protein science. Nat. Methods 11, 801–807 (2014).
Majithia, A. R. et al. Prospective functional classification of all possible missense variants in PPARG. Nat. Genet. 48, 1570–1575 (2016).
Sun, S. et al. A proactive genotype-to-patient-phenotype map for cystathionine beta-synthase. Genome Med. 12, 13 (2020).
Findlay, G. M., Boyle, E. A., Hause, R. J., Klein, J. C. & Shendure, J. Saturation editing of genomic regions by multiplex homology-directed repair. Nature 513, 120–123 (2014).
Findlay, G. M. et al. Accurate classification of BRCA1 variants with saturation genome editing. Nature 562, 217–222 (2018).
Meitlis, I. et al. Multiplexed functional assessment of genetic variants in CARD11. Am. J. Hum. Genet. 107, 1029–1043 (2020).
Kweon, J. et al. A CRISPR-based base-editing screen for the functional assessment of BRCA1 variants. Oncogene 39, 30–35 (2020).
Hanna, R. E. et al. Massively parallel assessment of human variants with base editor screens. Cell 184, 1064–1080 e1020 (2021).
Huang, C., Li, G., Wu, J., Liang, J. & Wang, X. Identification of pathogenic variants in cancer genes using base editing screens with editing efficiency correction. Genome Biol. 22, 80 (2021).
Anzalone, A. V. et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576, 149–157 (2019).
Carette, J. E. et al. Haploid genetic screens in human cells identify host factors used by pathogens. Science 326, 1231–1235 (2009).
Vanier, M. T. Niemann–Pick disease type C. Orphanet J. Rare Dis. 5, 16 (2010).
Wassif, C. A. et al. High incidence of unrecognized visceral/neurological late-onset Niemann–Pick disease, type C1, predicted by analysis of massively parallel sequencing data sets. Genet. Med. 18, 41–48 (2016).
Lin, Y. C. et al. Genome dynamics of the human embryonic kidney 293 lineage in response to cell biology manipulations. Nat. Commun. 5, 4767 (2014).
Hsu, P. D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 31, 827–832 (2013).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Wu, X. et al. Genome-wide binding of the CRISPR endonuclease Cas9 in mammalian cells. Nat. Biotechnol. 32, 670–676 (2014).
Duan, J. et al. Genome-wide identification of CRISPR/Cas9 off-targets in human genome. Cell Res. 24, 1009–1012 (2014).
Rose, J. C. et al. Suppression of unwanted CRISPR–Cas9 editing by co-administration of catalytically inactivating truncated guide RNAs. Nat. Commun. 11, 2697 (2020).
Coelho, M. A. et al. CRISPR GUARD protects off-target sites from Cas9 nuclease activity using short guide RNAs. Nat. Commun. 11, 4132 (2020).
Kopitz, J., Gerhard, C., Hofler, P. & Cantz, M. [14C]Methylamine accumulation in cultured human skin fibroblasts—a biochemical test for lysosomal storage and lysosomal diseases. Clin. Chim. Acta 227, 121–133 (1994).
Lachmann, R. H. et al. Treatment with miglustat reverses the lipid-trafficking defect in Niemann–Pick disease type C. Neurobiol. Dis. 16, 654–658 (2004).
te Vruchte, D. et al. Relative acidic compartment volume as a lysosomal storage disorder-associated biomarker. J. Clin. Invest. 124, 1320–1328 (2014).
Xu, M. et al. A phenotypic compound screening assay for lysosomal storage diseases. J. Biomol. Screen. 19, 168–175 (2014).
Millat, G. et al. Niemann–Pick C1 disease: the I1061T substitution is a frequent mutant allele in patients of Western European descent and correlates with a classic juvenile phenotype. Am. J. Hum. Genet. 65, 1321–1329 (1999).
Millat, G. et al. Niemann–Pick C1 disease: correlations between NPC1 mutations, levels of NPC1 protein, and phenotypes emphasize the functional significance of the putative sterol-sensing domain and of the cysteine-rich luminal loop. Am. J. Hum. Genet. 68, 1373–1385 (2001).
Dardis, A. et al. Molecular genetics of Niemann–Pick type C disease in Italy: an update on 105 patients and description of 18 NPC1 novel variants. J. Clin. Med. 9, 3 (2020).
Anzalone, A. V., Koblan, L. W. & Liu, D. R. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat. Biotechnol. 38, 824–844 (2020).
Kim, H. K. et al. Predicting the efficiency of prime editing guide RNAs in human cells. Nat. Biotechnol. 39, 198–206 (2021).
Dougherty, M. et al. Genome sequencing in a case of Niemann–Pick type C. Cold Spring Harb. Mol. Case Stud. 2, a001222 (2016).
Sevin, M. et al. The adult form of Niemann–Pick disease type C. Brain 130, 120–133 (2007).
Greenberg, C. R., Barnes, J. G., Kogan, S. & Seargeant, L. E. A rare case of Niemann–Pick disease type C without neurological involvement in a 66-year-old patient. Mol. Genet. Metab. Rep. 3, 18–20 (2015).
Rentzsch, P., Witten, D., Cooper, G. M., Shendure, J. & Kircher, M. CADD: predicting the deleteriousness of variants throughout the human genome. Nucleic Acids Res. 47, D886–D894 (2019).
Gelsthorpe, M. E. et al. Niemann–Pick type C1 I1061T mutant encodes a functional protein that is selected for endoplasmic reticulum-associated degradation due to protein misfolding. J. Biol. Chem. 283, 8229–8236 (2008).
Zampieri, S., Bembi, B., Rosso, N., Filocamo, M. & Dardis, A. Treatment of human fibroblasts carrying NPC1 missense mutations with MG132 leads to an improvement of intracellular cholesterol trafficking. JIMD Rep. 2, 59–69 (2012).
Watari, H. et al. Mutations in the leucine zipper motif and sterol-sensing domain inactivate the Niemann–Pick C1 glycoprotein. J. Biol. Chem. 274, 21861–21866 (1999).
Kuchenbaecker, K. B. et al. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA 317, 2402–2416 (2017).
Olopade, O. I. & Artioli, G. Efficacy of risk-reducing salpingo-oophorectomy in women with BRCA-1 and BRCA-2 mutations. Breast J. 10, S5–S9 (2004).
Rebbeck, T. R. et al. Bilateral prophylactic mastectomy reduces breast cancer risk in BRCA1 and BRCA2 mutation carriers: the PROSE Study Group. J. Clin. Oncol. 22, 1055–1062 (2004).
Labun, K. et al. CHOPCHOP v3: expanding the CRISPR web toolbox beyond genome editing. Nucleic Acids Res. 47, W171–W174 (2019).
Hart, S. N. et al. Comprehensive annotation of BRCA1 and BRCA2 missense variants by functionally validated sequence-based computational prediction models. Genet. Med. 21, 71–80 (2019).
Paquet, D. et al. Efficient introduction of specific homozygous and heterozygous mutations using CRISPR/Cas9. Nature 533, 125–129 (2016).
Liu, Y. et al. Enhancing prime editing by Csy4-mediated processing of pegRNA. Cell Res. 31, 1134–1136 (2021).
Nelson, J. W. et al. Engineered pegRNAs improve prime editing efficiency. Nat. Biotechnol. https://doi.org/10.1038/s41587-021-01039-7 (2021).
Kweon, J. et al. Engineered prime editors with PAM flexibility. Mol. Ther. https://doi.org/10.1016/j.ymthe.2021.02.022 (2021).
Liu, P. et al. Improved prime editors enable pathogenic allele correction and cancer modelling in adult mice. Nat. Commun. 12, 2121 (2021).
Hua, K., Jiang, Y., Tao, X. & Zhu, J. K. Precision genome engineering in rice using prime editing system. Plant Biotechnol. J. 18, 2167–2169 (2020).
Erwood, S. et al. Modeling Niemann–Pick disease type C in a human haploid cell line allows for patient variant characterization and clinical interpretation. Genome Res. 29, 2010–2019 (2019).
Kowarz, E., Loscher, D. & Marschalek, R. Optimized Sleeping Beauty transposons rapidly generate stable transgenic cell lines. Biotechnol. J. 10, 647–653 (2015).
Koblan, L. W. et al. Improving cytidine and adenine base editors by expression optimization and ancestral reconstruction. Nat. Biotechnol. 36, 843–846 (2018).
Walton, R. T., Christie, K. A., Whittaker, M. N. & Kleinstiver, B. P. Unconstrained genome targeting with near-PAMless engineered CRISPR-Cas9 variants. Science 368, 290–296 (2020).
Ran, F. A. et al. Genome engineering using the CRISPR–Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer-Verlag, 2016).
Kluesner, M. G. et al. EditR: a method to quantify base editing from Sanger sequencing. CRISPR J. 1, 239–250 (2018).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Comaniciu, D. & Meer, P. Mean shift: a robust approach toward feature space analysis. IEEE Trans. Pattern Anal. Mach. Intell. 24, 603–619 (2002).
Pedregosa, F. et al. Scikit-learn: machine learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Chernick, M. R. Bootstrap Methods: A Guide for Practitioners and Researchers Vol. 619 (John Wiley & Sons, 2011).
Chung, N. C. Statistical significance of cluster membership for unsupervised evaluation of cell identities. Bioinformatics 36, 3107–3114 (2020).
Henderson, A. R. The bootstrap: a technique for data-driven statistics. Using computer-intensive analyses to explore experimental data. Clin. Chim. Acta 359, 1–26 (2005).
Acknowledgements
This study was supported by Niemann–Pick Canada and the SickKids Foundation. Additional support was provided by a research grant from the University of Pennsylvania Orphan Disease Center in partnership with The Andrew Coppola Foundation (grant no. MDBR-21-113-NPC to E.A.I.) and by the Canadian Institute for Health Research (project grant no. 462511 to E.A.I.). We also thank the Hospital for Sick Children (Restracomp to S.E). We thank D. Durocher for sharing the TP53-null RPE1 cells. We thank B. Thiruvahindrapuram and T. Nalpathamkalam of the Centre for Applied Genomics, The Hospital for Sick Children, Toronto, Canada for assistance with applying their variant annotation pipeline. We are grateful to T. Durbic and the rest of the Donnelly Sequencing Centre staff for their assistance with sequencing experiments. This study was conducted with the support of the Ontario Institute for Cancer Research’s Genomics and Bioinformatics platform (genomics.oicr.on.ca) through funding provided by the Government of Ontario.
Author information
Authors and Affiliations
Contributions
S.E., T.M.I.B. and J.L. designed experiments. S.E., T.M.I.B., J.L., J.Y., N.G., R.A.B. and L.Z. performed experiments and analyzed data. J.L. designed and implemented the statistical framework for variant classification. S.E. and T.M.I.B. wrote the manuscript. All authors edited and revised the manuscript. E.A.I., L.P. and R.D.C. supervised the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characterization of the NPC1 haploidized HEK293T cell line.
(a) Schematic illustrating the position of digital droplet PCR probes (blue arrowheads) used to confirm haploidization. (b) Plot showing the droplet count values from assays used to measure NPC1 copy number relative to RNase P. The assay using probe 1, located in the unedited region, shows comparable counts between NPC1 and RNase P, while the assay from probe 2, located within the haploidized region, shows approximately one third the NPC1 droplets compared to RNase P. (c) Immunoblot for NPC1 demonstrating the relative NPC1 protein expression in wild type HEK293T cells versus NPC1 haploidized HEK293T cells. Values indicate quantity of protein lysate loaded in each lane. Representative image from one of two biological replicates. (d) Filipin staining was performed to visualize cholesterol distribution throughout wild type HEK293T cells, NPC1 haploidized HEK293T cells, and NPC1 haploidized NPC1 p.C909X cells. Distinct foci of cholesterol accumulation are apparent in NPC1 haploidized NPC1 p.C909X cells, which is absent from both wild type HEK293T cells and NPC1 haploidized HEK293T cells. Representative images from one of two biological replicates. Scale bars = 10 µm.
Extended Data Fig. 2 Functional scores for both NPC1 and BRCA2 were highly reproducible.
(a) Correlation of normalized functional scoring between replicates in NPC1 SPE experiments. (b) Correlation of normalized functional scoring between replicates in BRCA2 SPE experiments.
Extended Data Fig. 3 The three synonymous variants classified as deleterious disrupt proper NPC1 splicing.
(a) Immunoblot for NPC1 protein expression in each of the models isolated. Representative image from one of two biological replicates. (b) PCR amplification of cDNA from each model isolated revealed a distinct lower product that was absent in wild type control cDNA. Representative image from one of two biological replicates. (c) Sequencing of the lower PCR product depicted in (b) revealed errant splice products. These products are illustrated in the exon-intron diagrams. Arrowhead indicates approximate position of the mutation.
Extended Data Fig. 4 SPE is characterized by high editing efficiencies and product purity.
Data plotted as the mean ± standard deviation, (n = 8). PBS, primer binding site; RTT, reverse transcription template.
Extended Data Fig. 5 SPE is consistent along the length of the reverse transcription template.
Editing efficiencies of each individual mutation across the RT template are illustrated from four representative NPC1 SPE experiments (n = 8). The silent PAM-destroying is indicated within the square box. RTT, reverse transcription template.
Extended Data Fig. 6 Saturation prime editing is possible in RPE1 cells.
(a) Fluorescent distributions derived from fluorescence-activated cell sorting of LysoTracker-stained RPE1 wild type versus NPC1-deficient cell lines. (b) Plot of prime editing efficiency and outcomes in NPC1 haploidized RPE1 cells. PBS, primer binding site; RTT, reverse transcription template. Data plotted as the mean ± standard deviation (c) A plot illustrating the high correlation observed between function scores derived from saturation prime editing experiments in NPC1 haploidized HEK293T versus RPE cells (Pearson’s r = 0.92, n = 88). The functional consequence of each variant is indicated by colour.
Extended Data Fig. 7 NPC1 saturation prime editing function score reflects protein expression.
Immunoblot from subset of clones isolated from saturation prime editing experiments. The function scores followed by protein subgroups are listed in brackets. Colour of function score and protein category indicates functional classification with purple being deleterious and light turquoise being functional. Representative images from one of two biological replicates.
Extended Data Fig. 8 Copy number determination in the BRCA2 haploidized HEK293T cell line.
(a) Schematic illustrating the position of digital droplet PCR probes (blue arrowheads) used to confirm haploidization. (b) Plot showing the droplet count values from assays used to measure BRCA2 copy number relative to RNase P. The assay using probe 1, located in the haploidized region, shows approximately one third the droplets for BRCA2 compared to RNase P, while the assay from probe 2, located within the unedited region, shows comparable droplet counts between BRCA2 and RNase P.
Extended Data Fig. 9 Deleterious mutations result in impaired fitness in BRCA2 haploidized HEK293T cells.
(a,b) Plots showing the frequency of varied indels over time in BRCA2 haploidized HEK293T cells across two different loci. Frameshifting indels were markedly depleted between the two time points. Data plotted as the mean of two experimental replicates. (c-e) Editing frequency of each unique allele was determined by high-throughput sequencing over two timepoints. (c) BRCA2 p.Y2624C was introduced using base editing then depleted between the assayed time points. (d) BRCA2 p.S2691F was introduced using base editing then depleted between the assayed time points. (e) BRCA2 p.Y2726C was introduced using base editing then depleted between the assayed time points. Coloured letters indicate the amino acid change resulting from base editing.
Extended Data Fig. 10 Representative gating strategy used in the assay of NPC1 variants.
(a) Representative image depicting the initial gating of the population. (b) Representative image depicting the gating to exclude cell doublets. (c-d) Representative images depicting the linear gating strategy defining the ‘low’ and ‘high’ fluorescent populations. (c) Representative image depicting the distribution of LysoTrackerTM signal derived from unedited control NPC1 haploidized HEK293T cells. (d) Representative image depicting the distribution of LysoTrackerTM signal derived from NPC1 haploidized HEK293T cells that had been subject to saturation prime editing.
Supplementary information
Supplementary Information
Supplementary Notes 1 and 2.
Supplementary Tables
Supplementary Tables 1–7.
Source data
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 3
Unprocessed western blot and agarose gel.
Source Data Extended Data Fig. 7
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Erwood, S., Bily, T.M.I., Lequyer, J. et al. Saturation variant interpretation using CRISPR prime editing. Nat Biotechnol 40, 885–895 (2022). https://doi.org/10.1038/s41587-021-01201-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-021-01201-1
This article is cited by
-
scSNV-seq: high-throughput phenotyping of single nucleotide variants by coupled single-cell genotyping and transcriptomics
Genome Biology (2024)
-
High-throughput evaluation of genetic variants with prime editing sensor libraries
Nature Biotechnology (2024)
-
Precise genome-editing in human diseases: mechanisms, strategies and applications
Signal Transduction and Targeted Therapy (2024)
-
A prime editor mouse to model a broad spectrum of somatic mutations in vivo
Nature Biotechnology (2024)
-
Benchmarking splice variant prediction algorithms using massively parallel splicing assays
Genome Biology (2023)