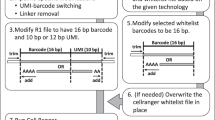
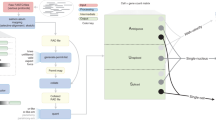
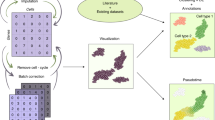
Abstract
We describe a workflow for preprocessing of single-cell RNA-sequencing data that balances efficiency and accuracy. Our workflow is based on the kallisto and bustools programs, and is near optimal in speed with a constant memory requirement providing scalability for arbitrarily large datasets. The workflow is modular, and we demonstrate its flexibility by showing how it can be used for RNA velocity analyses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
A diverse set of 20 datasets was compiled for the purpose of benchmarking preprocessing workflows. Datasets produced and distributed by 10x Genomics were downloaded from the 10x Genomics data downloads page: https://support.10xgenomics.com/single-cell-gene-expression/datasets. Six v3 chemistry datasets and two v2 chemistry datasets were downloaded and processed (Supplementary Table 3). Another 12 datasets were obtained from either the SRA or the European Nucleotide Archive; all were produced with 10x Genomics v2 chemistry. For six of the datasets (SRR6956073, SRR6998058, SRR7299563, SRR8206317, SRR8327928 and SRR8524760), the BAM files were downloaded and the Cell Ranger utility bamtofastq was run to produce FASTQ files for preprocessing from Cell Ranger–structured BAM files. FASTQ files were downloaded directly for the datasets E-MTAB-7320, SRR8257100, SRR8513910, SRR8599150 (available at https://github.com/bustools/getting_started/releases/download/getting_started/SRR8599150_S1_L001_R1_001.fastq.gz and https://github.com/bustools/getting_started/releases/download/getting_started/SRR8599150_S1_L001_R2_001.fastq.gz), SRR8611943 and SRR8639063.
Details of all datasets and their accession numbers can be found in Supplementary Table 3. All genome annotations and reference transcriptomes can be found at https://doi.org/10.22002/D1.1876.
Code availability
The software versions used for the results in the paper were: Alevin v0.13.1, bustools v0.39.1, Cell Ranger v3.0.0, DropletUtils v1.6.1, kallisto v0.46.0, Python 3.7, R v3.5.2, Scanpy v1.4.1, scvelo 0.1.17, Seurat v3.0, snakemake v5.3.0, STARsolo v2.7.0e, velocyto v0.17.17, wc v8.22 (GNU coreutils) and zcat v1.5 (gzip). All programs were run with default options unless otherwise specified. The code to reproduce the findings of this paper is available at https://github.com/pachterlab/MBLGLMBHGP_2021/, kallisto is available at https://github.com/pachterlab/kallisto/ and bustools is available at https://github.com/BUStools/bustools/. Documentation and tutorials for using the kallisto bustools scRNA-seq workflow are available at http://pachterlab.github.io/kallistobustools.
References
Tian, L. et al. scPipe: a flexible R/Bioconductor preprocessing pipeline for single-cell RNA-sequencing data. PLoS Comput. Biol. 14, e1006361 (2018).
Conesa, A. et al. A survey of best practices for RNA-seq data analysis. Genome Biol. 17, 13 (2016).
Kivioja, T. et al. Counting absolute numbers of molecules using unique molecular identifiers. Nat. Methods 9, 72–74 (2011).
Parekh, S., Ziegenhain, C., Vieth, B., Enard, W. & Hellmann, I. zUMIs - a fast and flexible pipeline to process RNA sequencing data with UMIs. Gigascience 7, giy059 (2018).
Srivastava, A., Malik, L., Smith, T., Sudbery, I. & Patro, R. Alevin efficiently estimates accurate gene abundances from dscRNA-seq data. Genome Biol. 20, 65 (2019).
Svensson, V., Vento-Tormo, R. & Teichmann, S. A. Exponential scaling of single-cell RNA-seq in the past decade. Nat. Protoc. 13, 599–604 (2018).
Zheng, G. X. Y. et al. Massively parallel digital transcriptional profiling of single cells. Nat. Commun. 8, 14049 (2017).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Svensson, V. et al. Power analysis of single-cell RNA-sequencing experiments. Nat. Methods 14, 381–387 (2017).
Melsted, P., Ntranos, V. & Pachter, L. The barcode, UMI, set format and BUStools. Bioinformatics 35, 4472–4473 (2019).
La Manno, G. et al. RNA velocity of single cells. Nature 560, 494–498 (2018).
Petukhov, V. et al. dropEst: pipeline for accurate estimation of molecular counts in droplet-based single-cell RNA-seq experiments. Genome Biol. 19, 78 (2018).
Hayer, K. E., Pizarro, A., Lahens, N. F., Hogenesch, J. B. & Grant, G. R. Benchmark analysis of algorithms for determining and quantifying full-length mRNA splice forms from RNA-seq data. Bioinformatics 31, 3938–3945 (2015).
Hwang, B., Lee, J. H. & Bang, D. Single-cell RNA sequencing technologies and bioinformatics pipelines. Exp. Mol. Med. 50, 1–14 (2018).
Ding, J., Adiconis, X., Simmons, S.K. et al. Systematic comparison of single-cell and single-nucleus RNA-sequencing methods. Nat. Biotechnol. 38, 737–746 (2020).
Yi, L., Liu, L., Melsted, P. & Pachter, L. A direct comparison of genome alignment and transcriptome pseudoalignment. Preprint at bioRxiv https://doi.org/10.1101/444620 (2018).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Habib, N. et al. Massively parallel single-nucleus RNA-seq with DroNc-seq. Nat. Methods 14, 955–958 (2017).
Ryu, K. H., Huang, L., Kang, H. M. & Schiefelbein, J. Single-cell RNA sequencing resolves molecular relationships among individual plant cells. Plant Physiol. 179, 1444–1456 (2019).
Packer, J. S. et al. A lineage-resolved molecular atlas of C. elegans embryogenesis at single-cell resolution. Science 365, eaax1971 (2019).
Farrell, J. A. et al. Single-cell reconstruction of developmental trajectories during zebrafish embryogenesis. Science 360, eaar3131 (2018).
Carosso, G. A. et al. Precocious neuronal differentiation and disrupted oxygen responses in Kabuki syndrome. JCI Insight 4, e129375 (2019).
Merino, D. et al. Barcoding reveals complex clonal behavior in patient-derived xenografts of metastatic triple negative breast cancer. Nat. Commun. 10, 766 (2019).
O’Koren, E. G. et al. Microglial function is distinct in different anatomical locations during retinal homeostasis and degeneration. Immunity 50, 723–737 (2019).
Jin, R. M., Warunek, J. & Wohlfert, E. A. Chronic infection stunts macrophage heterogeneity and disrupts immune-mediated myogenesis. JCI Insight 3, e121549 (2018).
Miller, B. C. et al. Subsets of exhausted CD8+ T cells differentially mediate tumor control and respond to checkpoint blockade. Nat. Immunol. 20, 326–336 (2019).
Delile, J. et al. Single cell transcriptomics reveals spatial and temporal dynamics of gene expression in the developing mouse spinal cord. Development 146, dev173807. (2019).
Guo, L. et al. Resolving cell fate decisions during somatic cell reprogramming by single-cell RNA-seq. Mol. Cell 73, 815–829 (2019).
Traag, V. A., Waltman, L. & van Eck, N. J. From Louvain to Leiden: guaranteeing well-connected communities. Sci. Rep. 9, 5233 (2019).
Clark, B. S. et al. Single-cell RNA-seq analysis of retinal development identifies NFI factors as regulating mitotic exit and late-born cell specification. Neuron 102, 1111–1126 (2019).
Ntranos, V., Yi, L., Melsted, P. & Pachter, L. A discriminative learning approach to differential expression analysis for single-cell RNA-seq. Nat. Methods 16, 163–166 (2019).
Soós, S. Age-sensitive bibliographic coupling reflecting the history of science: the case of the Species Problem. Scientometrics 98, 23–51 (2014).
Lun, A. T. L. et al. EmptyDrops: distinguishing cells from empty droplets in droplet-based single-cell RNA sequencing data. Genome Biol. 20, 63 (2019).
Griffiths, J. A., Richard, A. C., Bach, K., Lun, A. T. L. & Marioni, J. C. Detection and removal of barcode swapping in single-cell RNA-seq data. Nat. Commun. 9, 2667 (2018).
Alexa, A., Rahnenführer, J. & Lengauer, T. Improved scoring of functional groups from gene expression data by decorrelating GO graph structure. Bioinformatics 22, 1600–1607 (2006).
Ashburner, M. et al. Gene Ontology: tool for the unification of biology. Nat. Genet. 25, 25–29 (2000).
The Gene Ontology Consortium. The Gene Ontology Resource: 20 years and still GOing strong. Nucleic Acids Res. 47, D330–D338 (2019).
Aran, D. et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat. Immunol. 20, 163–172 (2019).
Benayoun, B. A. et al. Remodeling of epigenome and transcriptome landscapes with aging in mice reveals widespread induction of inflammatory responses. Genome Res. 29, 697–709 (2019).
Street, K. et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics 19, 477 (2018).
Saelens, W., Cannoodt, R., Todorov, H. & Saeys, Y. A comparison of single-cell trajectory inference methods. Nat. Biotechnol. 37, 547–554 (2019).
Macosko, E. Z. et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell 161, 1202–1214 (2015).
Acknowledgements
We thank V. Ntranos and V. Svensson for helpful suggestions and comments. We thank J. Farrell for the D. rerio gene annotation used to process SRR6956073, J. Schiefelbein for the A. thaliana gene annotation used to process SRR8257100, J. Fear for the D. melanogaster gene annotation used to process SRR8513910, and J. Kim and Q. Zhu for the C. elegans gene annotation used to process SRR8611943. The benchmarking work was made possible, in part, thanks to support from the Beckman Institute Caltech Bioinformatics Resource Center. A.S.B. and L.P. were funded in part by NIH U19MH114830.
Author information
Authors and Affiliations
Contributions
P.M., A.S.B., L. Liu and L.P. developed the algorithms for bustools and P.M., A.S.B. and L. Liu wrote the software. A.S.B. conceived of and performed the UMI and barcode calculations motivating the algorithms. F.G. implemented and performed the benchmarking procedure, and curated indices for the datasets. A.S.B. and E.d.V.B. designed and produced the comparisons between Cell Ranger and kallisto bustools. L. Lu investigated in detail the performance of different workflows on the “10k mouse neuron” data and produced the analysis of that dataset. A.S.B. designed the RNA velocity workflow and performed the RNA velocity analyses. K.M.H contributed to the development of the reproducible workflow. K.E.H. developed and investigated the effect of reference transcriptome sequences for pseudoalignment. J.G. interpreted results and helped to supervise the research. A.S.B. planned, organized and prepared figures. A.S.B., E.d.V.B., P.M. and L.P. planned the manuscript. A.S.B. and L.P. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature Biotechnology thanks the anonymous reviewers for their contribution to the peer review of this work.
Supplementary information
Supplementary Information
Supplementary Figs. 1–15, Note and Table 2.
Supplementary Table 1
Runtime, memory and cost.
Supplementary Table 3
Benchmark panel summary.
Rights and permissions
About this article
Cite this article
Melsted, P., Booeshaghi, A.S., Liu, L. et al. Modular, efficient and constant-memory single-cell RNA-seq preprocessing. Nat Biotechnol 39, 813–818 (2021). https://doi.org/10.1038/s41587-021-00870-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-021-00870-2
This article is cited by
-
Single-nucleus RNA velocity reveals critical synaptic and cell-cycle dysregulations in neuropathologically confirmed Alzheimer’s disease
Scientific Reports (2024)
-
Epithelial zonation along the mouse and human small intestine defines five discrete metabolic domains
Nature Cell Biology (2024)
-
Deep generative modeling of transcriptional dynamics for RNA velocity analysis in single cells
Nature Methods (2024)
-
Early-life prefrontal cortex inhibition and early-life stress lead to long-lasting behavioral, transcriptional, and physiological impairments
Molecular Psychiatry (2024)
-
Distinct pathways drive anterior hypoblast specification in the implanting human embryo
Nature Cell Biology (2024)