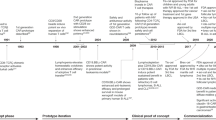
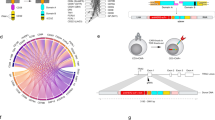
Abstract
Despite the global rapid increase in the number of clinical trials employing chimeric antigen receptors (CARs), no comprehensive survey of their scope, targets and design exists. In this study, we present an interactive CAR clinical trial database, spanning 64 targets deployed in T cells (CAR-T), natural killer cells (CAR-NK) or mixtures (CAR-NK/T) from over 500 clinical trials in 20 countries, encompassing >20,000 patients. By combining these data with transcriptional and proteomic data, we create a ‘targetable landscape’ for CAR cell therapies based on 13,206 proteins and RNAs across 78 tissues, 124 cell types and 20 cancer types. These data suggest a landscape of over 100 single targets and over 100,000 target pairs using logical switches for CAR cell engineering. Our analysis of the CAR cellular therapeutic landscape may aid the design of future therapies, improve target-to-patient matching, and ultimately help inform our understanding of CAR therapy’s safety and efficacy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
We have created an indexed and searchable online site for displaying clinical CAR targets (https://carglobaltrials.com), which also features the normal (tissue group, tissue and cell type) and pathological gene expression data for those targets.
Code availability
Relevant code for identification of new targets and target pairs, and for parsing the current CAR targets is available in the Supplementary Materials.
References
Perna, F. et al. Integrating proteomics and transcriptomics for systematic combinatorial chimeric antigen receptor therapy of AML. Cancer Cell. 32, 506–519.e5 (2017).
Klebanoff, C. A., Rosenberg, S. A. & Restifo, N. P. Prospects for gene-engineered T cell immunotherapy for solid cancers. Nat. Med. 22, 26–36 (2016).
Uhlén, M. et al. A human protein atlas for normal and cancer tissues based on antibody proteomics. Mol. Cell Proteom. 4, 1920–1932 (2005).
Uhlén, M. et al. Proteomics. tissue-based map of the human proteome. Science 347, 1260419 (2015).
Arcangeli, S. et al. Balance of anti-CD123 chimeric antigen receptor binding affinity and density for the targeting of acute myeloid leukemia. Mol. Ther. 25, 1933–1945 (2017).
Guo, D. L., Dong, M., Wang, L., Sun, L. P. & Yuan, Y. Expression of gastric cancer-associated MG7 antigen in gastric cancer, precancerous lesions and H. pylori-associated gastric diseases. World J. Gastroenterol. 8, 1009–1013 (2002).
Gan, H. K., Cvrljevic, A. N. & Johns, T. G. The epidermal growth factor receptor variant III (EGFRvIII): where wild things are altered. FEBS J. 280, 5350–5370 (2013).
Liu, H. P. et al. Epstein–Barr virus-encoded LMP1 interacts with FGD4 to activate Cdc42 and thereby promote migration of nasopharyngeal carcinoma cells. PLoS Pathog. 8, e1002690 (2012).
Morgan, R. A. et al. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing Erbb2. Mol. Ther. 18, 843–851 (2010).
Ahmed, N. et al. HER2-specific chimeric antigen receptor-modified virus-specific t cells for progressive glioblastoma: a phase 1 dose-escalation trial. JAMA Oncol. 3, 1094–1101 (2017).
Feng, K. et al. Phase I study of chimeric antigen receptor modified T cells in treating HER2-positive advanced biliary tract cancers and pancreatic cancers. Protein Cell 9, 838–847 (2018).
Poh, A. JCAR015 in ALL: a root-cause investigation. Cancer Discovery 8, 4.3–5 (2018).
Drent, E. et al. A rational strategy for reducing on-target off-tumor effects of cd38-chimeric antigen receptors by affinity optimization. Mol. Ther. 25, 1946–1958 (2017).
Uhlén, M. et al. A pathology atlas of the human cancer transcriptome. Science 357, eaan2507 (2017).
Yang, Q. et al. dbDEPC 3.0: the database of differentially expressed proteins in human cancer with multi-level annotation and drug indication. Database 2018, bay015 (2018).
Neelapu, S. S. et al. Axicabtagene ciloleucel car t-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Maude, S. L. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. New Engl. J. Med. 378, 439–448 (2018).
Liu, H. et al. Targeting alpha-fetoprotein (AFP)–MHC complex with CAR T-cell therapy for liver cancer. Clin. Cancer Res. 23, 478–488 (2017).
Turazzi, N. et al. Engineered T cells towards TNFRSF13C (BAFFR): a novel strategy to efficiently target B-cell acute lymphoblastic leukaemia. Br. J. Haematol. 182, 939–943 (2018).
Scarfò, I. et al. Anti-CD37 chimeric antigen receptor T cells are active against B- and T-cell lymphomas. Blood 132, 1495–1506 (2018).
Cho, J. H., Collins, J. J. & Wong, W. W. Universal chimeric antigen receptors for multiplexed and logical control of T cell responses. Cell 173, 1426–1438.e11 (2018).
Stoeckius, M. et al. Simultaneous epitope and transcriptome measurement in single cells. Nat. Methods. 14, 865–868 (2017).
Garrett-Bakelman, F. E. et al. The NASA Twins Study: a multi-omic, molecular, physiological, and behavioral analysis of a year-long human spaceflight. Science 364, eaau8650 (2019).
Neelapu, S. S. et al. Chimeric antigen receptor T-cell therapy—assessment and management of toxicities. Nat. Rev. Clin. Oncol. 15, 47–62 (2018).
Lohmueller, J. J. et al. mSA2 affinity-enhanced biotin-binding CAR T cells for universal tumor targeting. OncoImmunol. 7, e1368604 (2017).
Acknowledgements
This work was supported by STARR Cancer Consortium grants (nos. I7-A765, I9-A9-071) and funding from the Irma T. Hirschl and Monique Weill-Caulier Charitable Trusts, Bert L. and N. Kuggie Vallee Foundation, Igor Tulchinsky and the WorldQuant Foundation, the Pershing Square Sohn Cancer Research Alliance, the National Institutes of Health (grant nos. P01CA214274, 1R01MH117406) and the Leukemia and Lymphoma Society (grant nos. LLS 9238-16, Mak, LLS-MCL-982, Chen-Kiang). Funding also came from the National Natural Science Foundation of China (grant nos. 81402529 and 81672994), Zhejiang Provincial Foundation for Natural Sciences (grant no. LZ15H220001), Zhejiang Provincial Medical Scientific Research Foundation of China (grant nos. 2015KYB325 and 2015PYA009), Hangzhou City Medical Scientific Research Foundation of Zhejiang Province (grant no. 2015Z04) and Hangzhou City Scientific Technology Research Foundation of Zhejiang Province (grant no. 20150733Q64) of the Hangzhou Science and Technology Development Program (grant no. 20150733Q63).
Author information
Authors and Affiliations
Contributions
C.E.M., M.M. and S.W. conceived the study. C.E.M. and M.M. performed the clinical site setup and framework. A.M.M., G.R. and M.G. updated the clinical annotation and target maps. C.E.M. and S.W. provided study support and logistics. S.W. led the on-site validation. M.M. led the code and analysis of public and clinical trial data and created the site code base. E.A. and J.R. validated the clinical trial targets. C.H., M.K., N.B., B.O. and L.L. from the Yale CAR Annotation Team cross-validated the parameters.
Corresponding authors
Ethics declarations
Competing interests
There are no specific, relevant competing interests related to this work. However, in the interest of full disclosure, C.M. is a cofounder and board member for Biotia and Onegevity Health, as well as an advisor or compensated speaker for Abbvie, Acuamark Diagnostics, ArcBio, BioRad, DNA Genotek, Genialis, Genpro, Karius, Illumina, New England Biolabs, QIAGEN, Whole Biome and Zymo Research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 CAR stratification by normal expression and recent usage in the clinic.
(a) The number of trials from 2017–2019 that used a given CAR compare to the percent of tissues with medium-high levels of expression of the CAR target. (b) The percentage of tissue groups with not-detected, low, medium, or high levels of target expression based on maximum expression seen within a cell type for CARs used in multiple recent trials. (c) Tissue group, (d) tissue, and (e) cell types with a given CAR target highly expressed. CARs examined are listed in (b).
Supplementary information
Supplementary Materials
Supplementary Fig. 1.
Supplementary Tables
Supplementary Tables 1–3.
Analysis code
CAR_NBT_analysis_expression.js, CAR_NBT_analysis_location.js, CAR_NBT_analysis_logicalAndNotPairs.R, CAR_NBT_analysis_logicalAndPairs.R, CAR_NBT_analysis_meta.js, CAR_NBT_analysis_singleTargets.R
Rights and permissions
About this article
Cite this article
MacKay, M., Afshinnekoo, E., Rub, J. et al. The therapeutic landscape for cells engineered with chimeric antigen receptors. Nat Biotechnol 38, 233–244 (2020). https://doi.org/10.1038/s41587-019-0329-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-019-0329-2
This article is cited by
-
Novel insights into TCR-T cell therapy in solid neoplasms: optimizing adoptive immunotherapy
Experimental Hematology & Oncology (2024)
-
Chimeric antigen receptor-based natural killer cell immunotherapy in cancer: from bench to bedside
Cell Death & Disease (2024)
-
Making drugs from T cells: The quantitative pharmacology of engineered T cell therapeutics
npj Systems Biology and Applications (2024)
-
Targeted immunotherapy: harnessing the immune system to battle multiple myeloma
Cell Death Discovery (2024)
-
Telomeres and aging: on and off the planet!
Biogerontology (2024)