Abstract
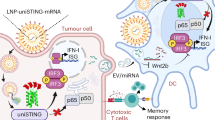
Therapeutic messenger RNA vaccines enable delivery of whole antigens, which can be advantageous over peptide vaccines. However, optimal efficacy requires both intracellular delivery, to allow antigen translation, and appropriate immune activation. Here, we developed a combinatorial library of ionizable lipid-like materials to identify mRNA delivery vehicles that facilitate mRNA delivery in vivo and provide potent and specific immune activation. Using a three-dimensional multi-component reaction system, we synthesized and evaluated the vaccine potential of over 1,000 lipid formulations. The top candidate formulations induced a robust immune response, and were able to inhibit tumor growth and prolong survival in melanoma and human papillomavirus E7 in vivo tumor models. The top-performing lipids share a common structure: an unsaturated lipid tail, a dihydroimidazole linker and cyclic amine head groups. These formulations induce antigen-presenting cell maturation via the intracellular stimulator of interferon genes (STING) pathway, rather than through Toll-like receptors, and result in limited systemic cytokine expression and enhanced anti-tumor efficacy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available form the corresponding author upon reasonable request.
References
Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines—a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018).
Pollard, C. et al. Type I IFN counteracts the induction of antigen-specific immune responses by lipid-based delivery of mRNA vaccines. Mol. Ther. 21, 251–259 (2013).
Jayaraman, M. et al. Maximizing the potency of siRNA lipid nanoparticles for hepatic gene silencing in vivo. Angew. Chem. Int. Ed. Engl. 51, 8529–8533 (2012).
Kauffman, K. J., Webber, M. J. & Anderson, D. G. Materials for non-viral intracellular delivery of messenger RNA therapeutics. J. Control. Release 240, 227–234 (2016).
Richner, J. M. et al. Modified mRNA vaccines protect against Zika virus infection. Cell 168, 1114–1125.e10 (2017).
Kariko, K. et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 16, 1833–1840 (2008).
Li, K., Qu, S., Chen, X., Wu, Q. & Shi, M. Promising targets for cancer immunotherapy: TLRs, RLRs, and STING-mediated innate immune pathways. Int. J. Mol. Sci. 18, pii: E404 (2017).
Zevini, A., Olagnier, D. & Hiscottt, J. Crosstalk between cytoplasmic RIG-I and STING sensing pathways. Trends Immunol. 38, 194–205 (2017).
Barber, G. N. STING: infection, inflammation and cancer. Nat. Rev. Immunol. 15, 760–770 (2015).
Corrales, L. et al. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 11, 1018–1030 (2015).
Kranz, L. M. et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature 534, 396–401 (2016).
Fotin-Mleczek, M. et al. Highly potent mRNA based cancer vaccines represent an attractive platform for combination therapies supporting an improved therapeutic effect. J. Gene Med. 14, 428–439 (2012).
Schlee, M. et al. Recognition of 5’ triphosphate by RIG-I helicase requires short blunt double-stranded RNA as contained in panhandle of negative-strand virus. Immunity 31, 25–34 (2009).
Barbalat, R., Ewald, S. E., Mouchess, M. L. & Barton, G. M. Nucleic acid recognition by the innate immune system. Annu. Rev. Immunol. 29, 185–214 (2011).
Uchida, S. et al. Designing immunostimulatory double stranded messenger RNA with maintained translational activity through hybridization with poly a sequences for effective vaccination. Biomaterials 150, 162–170 (2018).
Van Lint, S. et al. Preclinical evaluation of TriMix and antigen mRNA-based antitumor therapy. Cancer Res. 72, 1661–1671 (2012).
Broos, K. et al. Particle-mediated intravenous delivery of antigen mRNA results in strong antigen-specific T-cell responses despite the induction of type I interferon. Mol. Ther. Nucleic Acids 5, e326 (2016).
Iurescia, S., Fioretti, D. & Rinaldi, M. Nucleic acid sensing machinery: targeting innate immune system for cancer therapy. Recent Pat. Anticancer Drug Discov. 13, 2–17 (2018).
Wang, J., Li, P. & Wu, M. X. Natural STING agonist as an “ideal” adjuvant for cutaneous vaccination. J. Invest. Dermatol. 136, 2183–2191 (2016).
Luo, M. et al. A STING-activating nanovaccine for cancer immunotherapy. Nat. Nanotechnol. 12, 648–654 (2017).
Caucheteux, S. M. & Piguet, V. New cutaneous vaccine adjuvant that STINGs a little less. J. Invest. Dermatol. 136, 2127–2128 (2016).
Iribarren, K. et al. Trial watch: immunostimulation with Toll-like receptor agonists in cancer therapy. Oncoimmunology 5, e1088631 (2016).
Holm, C. K. et al. Virus-cell fusion as a trigger of innate immunity dependent on the adaptor STING. Nat. Immunol. 13, 737–743 (2012).
Fu, J. et al. STING agonist formulated cancer vaccines can cure established tumors resistant to PD-1 blockade. Sci. Transl. Med. 7, 283ra252 (2015).
Wilson, D. R. et al. Biodegradable STING agonist nanoparticles for enhanced cancer immunotherapy. Nanomedicine 14, 237–246 (2018).
Demaria, O. et al. STING activation of tumor endothelial cells initiates spontaneous and therapeutic antitumor immunity. Proc. Natl Acad. Sci. USA 112, 15408–15413 (2015).
Hulme, C. & Gore, V. “Multi-component reactions: emerging chemistry in drug discovery” ‘from xylocain to crixivan’. Curr. Med. Chem. 10, 51–80 (2003).
Sahay, G. et al. Efficiency of siRNA delivery by lipid nanoparticles is limited by endocytic recycling. Nat. Biotechnol. 31, 653–658 (2013).
Gilleron, J. et al. Image-based analysis of lipid nanoparticle-mediated siRNA delivery, intracellular trafficking and endosomal escape. Nat. Biotechnol. 31, 638–646 (2013).
Cheng, N. et al. A nanoparticle-incorporated STING activator enhances antitumor immunity in PD-L1-insensitive models of triple-negative breast cancer. JCI Insight 3, pii: 120638 (2018).
Tanaka, Y., Hasui, T. & Suginome, M. Acid-free, aminoborane-mediated Ugi-type reaction leading to general utilization of secondary amines. Org. Lett. 9, 4407–4410 (2007).
Kazmaier, U. & Ackermann, S. A straightforward approach towards thiazoles and endothiopeptides via Ugi reaction. Org. Biomol. Chem. 3, 3184–3187 (2005).
Pan, S. C. & List, B. Catalytic three-component Ugi reaction. Angew. Chem. Int. Ed. Engl. 47, 3622–3625 (2008).
Fenton, O. S. et al. Bioinspired alkenyl amino alcohol ionizable lipid materials for highly potent in vivo mRNA delivery. Adv. Mater. 28, 2939–2943 (2016).
Akinc, A. et al. A combinatorial library of lipid-like materials for delivery of RNAi therapeutics. Nat. Biotechnol. 26, 561–569 (2008).
Koopmanschap, G., Ruijter, E. & Orru, R. V. Isocyanide-based multicomponent reactions towards cyclic constrained peptidomimetics. Beilstein J. Org. Chem. 10, 544–598 (2014).
Whitehead, K. A. et al. Degradable lipid nanoparticles with predictable in vivo siRNA delivery activity. Nat. Commun. 5, 4277 (2014).
Semple, S. C. et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 28, 172–176 (2010).
Kauffman, K. J. et al. Optimization of lipid nanoparticle formulations for mRNA delivery in vivo with fractional factorial and definitive screening designs. Nano Lett. 15, 7300–7306 (2015).
Whitehead, K. A. et al. In vitro-in vivo translation of lipid nanoparticles for hepatocellular siRNA delivery. ACS Nano 6, 6922–6929 (2012).
Chahal, J. S. et al. Dendrimer-RNA nanoparticles generate protective immunity against lethal Ebola, H1N1 influenza, and Toxoplasma gondii challenges with a single dose. Proc. Natl Acad. Sci. USA 113, E4133–E4142 (2016).
Sahin, U. et al. Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature 547, 222–226 (2017).
Gu, B., Linehan, B. & Tseng, Y. C. Optimization of the buchi B-90 spray drying process using central composite design for preparation of solid dispersions. Int. J. Pharm. 491, 208–217 (2015).
Heyes, J., Palmer, L., Bremner, K. & MacLachlan, I. Cationic lipid saturation influences intracellular delivery of encapsulated nucleic acids. J. Control. Release 107, 276–287 (2005).
Oberli, M. A. et al. Lipid nanoparticle assisted mRNA delivery for potent cancer immunotherapy. Nano Lett. 17, 1326–1335 (2017).
Kuai, R., Ochyl, L. J., Bahjat, K. S., Schwendeman, A. & Moon, J. J. Designer vaccine nanodiscs for personalized cancer immunotherapy. Nat. Mater. 16, 489–496 (2017).
Ma, D. Y. & Clark, E. A. The role of CD40 and CD154/CD40L in dendritic cells. Semin. Immunol. 21, 265–272 (2009).
Walseng, E. et al. Dendritic cell activation prevents MHC class II ubiquitination and promotes MHC class II survival regardless of the activation stimulus. J. Biol. Chem. 285, 41749–41754 (2010).
Deering, R. P., Kommareddy, S., Ulmer, J. B., Brito, L. A. & Geall, A. J. Nucleic acid vaccines: prospects for non-viral delivery of mRNA vaccines. Expert Opin. Drug Deliv. 11, 885–899 (2014).
Bloom, M. B. et al. Identification of tyrosinase-related protein 2 as a tumor rejection antigen for the B16 melanoma. J. Exp. Med. 185, 453–459 (1997).
Aviles, E. et al. Synthesis and preliminary biological evaluation of a small library of hybrid compounds based on Ugi isocyanide multicomponent reactions with a marine natural product scaffold. Bioorg. Med. Chem. Lett. 25, 5339–5343 (2015).
Ugi, I., Werner, B. & Dömling, A. The chemistry of isocyanides, their multicomponent reactions and their libraries. Molecules 8, 4 (2003).
Wada, J. et al. A new nonsteroidal antiinflammatory agent. 2-Substituted 5- or 6-benzothiazoleacetic acids and their derivatives. J. Med. Chem. 16, 930–934 (1973).
Ohkuri, T. et al. Intratumoral administration of cGAMP transiently accumulates potent macrophages for anti-tumor immunity at a mouse tumor site. Cancer Immunol. Immunother. 66, 705–716 (2017).
Rao, R. S., Kumar, C. G., Prakasham, R. S. & Hobbs, P. J. The Taguchi methodology as a statistical tool for biotechnological applications: a critical appraisal. Biotechnol. J. 3, 510–523 (2008).
Belliveau, N. M. et al. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of siRNA. Mol. Ther. Nucleic Acids 1, e37 (2012).
Feig, C. et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl Acad. Sci. USA 110, 20212–20217 (2013).
Maruggi, G. et al. Immunogenicity and protective efficacy induced by self-amplifying mRNA vaccines encoding bacterial antigens. Vaccine 35, 361–368 (2017).
Acknowledgements
This work was supported by Translate Bio (Lexington, MA) and the Juvenile Diabetes Research Foundation (grant nos. 17-2007-1063 and 3-PDF-2015-91-A-N). This work is supported in part by the Cancer Center Support (core) (grant no. P30-CA14051) from the National Institutes of Health. D.G.A. is a consultant for Translate Bio. L.M. was supported by a Misrock postdoctoral fellowship. D.D. was supported by an EPSRC E-TERM Fellowship (EP/I017801/1) and a Marie Sklodowska Curie Fellowship (IF/798348). K.S. was supported by the Ruth L. Kirschstein NRSA Postdoctoral Fellowship (no. 1F32EB025688-01A1) from the National Institute of Biomedical Imaging and Bioengineering of the National Institutes of Health. L.L. and J.H. were supported by a Ming Wai Lau grant from the Ming Wai Lau Centre for Reparative Medicine, Karolinska Institutet. The authors would like to acknowledge the use of resources at the W. M. Keck Biological Imaging Facility (Whitehead Institute) and at the Microscopy, Histology, Animal Imaging & Preclinical Testing and Flow Cytometry Core Facilities (Swanson Biotechnology Center, David H. Koch Institute for Integrative Cancer Research at MIT), and acknowledge Wuxi Further Pharmaceutical Co., Ltd for synthesizing and providing lipid materials.
Author information
Authors and Affiliations
Contributions
L.M. and D.G.A. designed experiments and analyzed data. L.L. designed the chemical structures. L.M., Y.H., D.D., Y.S., J.C., K.S., W.G. and J.H. performed experiments. J.L. and L.M. drew the schematic figures. L.M., D.D., J.C.D., Y.H. and D.G.A. wrote the manuscript. R.L. and D.G.A. supervised the study. All authors discussed the results and assisted in the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
L.M., Y.H., L.L. and D.G.A. have filed a patent for the development of the described lipids synthesized using the three-component reaction. R.L. receives licensing fees (to patents in which he was an inventor on) from, invested in, consults (or was on scientific advisory boards or boards of directors) for, lectured (and received a fee), or conducts sponsored research at MIT for which he was not paid for the following entities: 7th Sense, Abpro, Aleph Farms, Alkermes, Allevi, Alnylam, Artificial Cells, Arsenal Medical, BASF, Celero, Cellomics, Cellular Biomedical, Clarus, Clontech, Combined Therapeutics, Conference Forum, Curis, Domain, Eagle, Echo, Edge, Evox, Fate Therapeutics, Frequency Therapeutics, Genscript, Glycobia, Glympse, Grandhope, Greenlight, HKF Technologies, Horizon Discovery, Humacyte, Indivor, Inovio, Institute of Immunology, In Vivo Therapeutics, Ironwood Pharmaceuticals, Kallyope, Kensa, Keratinx, KSQ Therapeutics, Laderatech, Inc., Landsdowne Labs, Like Minds, Luminopia, Luye, Lyndra, Lyra, Medical Kinetics, Merck, Micelle, Moderna, Momenta, Monsanto, Mylan, Nanobiosym, Nanobiotix, Noveome, Particles for Humanity, Perosphere, Pfizer, Polaris, Portal, Pulmatrix, Puretech, Roche, Rubius, Secant, Selecta Biosciences, Setsuro, Shiseido, Sigilon, Sio2, SQZ, Stembiosys, Suono Bio, T2 Biosystems, Tara, Taris Biomedical, Tarveda, Third Rock, Tiba, Tissium, Titan Pharma, Unilever, VasoRX, Verseau Therapeutics, Vivtex, Wiki Foods and Zenomic.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–24, Tables 1, 2 and 4, and Notes 1–4.
Supplementary Table 3
Characterization of the top-performing lipids.
Rights and permissions
About this article
Cite this article
Miao, L., Li, L., Huang, Y. et al. Delivery of mRNA vaccines with heterocyclic lipids increases anti-tumor efficacy by STING-mediated immune cell activation. Nat Biotechnol 37, 1174–1185 (2019). https://doi.org/10.1038/s41587-019-0247-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-019-0247-3
This article is cited by
-
In situ combinatorial synthesis of degradable branched lipidoids for systemic delivery of mRNA therapeutics and gene editors
Nature Communications (2024)
-
Liquid crystalline inverted lipid phases encapsulating siRNA enhance lipid nanoparticle mediated transfection
Nature Communications (2024)
-
Combinatorial development of nebulized mRNA delivery formulations for the lungs
Nature Nanotechnology (2024)
-
Research Progress of Nanomaterials for Prevention, Diagnosis, and Treatment of SARS-CoV-2
BioNanoScience (2024)
-
Natural long-chain saturated fatty acids doped LNPs enabling spleen selective mRNA translation and potent cancer immunotherapy
Nano Research (2024)