Abstract
Sex chromosome disorders severely compromise gametogenesis in both males and females. In oogenesis, the presence of an additional Y chromosome or the loss of an X chromosome disturbs the robust production of oocytes1,2,3,4,5. Here we efficiently converted the XY chromosome set to XX without an additional Y chromosome in mouse pluripotent stem (PS) cells. In addition, this chromosomal alteration successfully eradicated trisomy 16, a model of Down’s syndrome, in PS cells. Artificially produced euploid XX PS cells differentiated into mature oocytes in culture with similar efficiency to native XX PS cells. Using this method, we differentiated induced pluripotent stem cells from the tail of a sexually mature male mouse into fully potent oocytes, which gave rise to offspring after fertilization. This study provides insights that could ameliorate infertility caused by sex chromosome or autosomal disorders, and opens the possibility of bipaternal reproduction.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The DNA-seq data have been deposited at the Sequence Read Archive (SRA) database under accession number PRJNA766461 and the RNA-seq data have been deposited at the Gene Expression Omnibus (GEO) database under accession number GSE184771. Source data are provided with this paper.
Code availability
Custom code used in this article can be accessed at https://github.com/kentamurakami1986/oocytes-from-male-mice.
References
Vernet, N. et al. The expression of Y-linked Zfy2 in XY mouse oocytes leads to frequent meiosis 2 defects, a high incidence of subsequent early cleavage stage arrest and infertility. Development 141, 855–866 (2014).
Amleh, A., Ledee, N., Saeed, J. & Taketo, T. Competence of oocytes from the B6.YDOM sex-reversed female mouse for maturation, fertilization, and embryonic development in vitro. Dev. Biol. 178, 263–275 (1996).
Mahadevaiah, S. K., Lovell-Badge, R. & Burgoyne, P. S. Tdy-negative XY, XXY and XYY female mice: breeding data and synaptonemal complex analysis. J. Reprod. Fertil. 97, 151–160 (1993).
Taketo-Hosotani, T., Nishioka, Y., Nagamine, C. M., Villalpando, I. & Merchant-Larios, H. Development and fertility of ovaries in the B6.YDOM sex-reversed female mouse. Development 107, 95–105 (1989).
Lavery, R. et al. XY Sox9 embryonic loss-of-function mouse mutants show complete sex reversal and produce partially fertile XY oocytes. Dev. Biol. 354, 111–122 (2011).
Saitou, M. & Yamaji, M. Primordial germ cells in mice. Cold Spring Harb. Perspect. Biol. 4, a008375 (2012).
Adams, I. R. & McLaren, A. Sexually dimorphic development of mouse primordial germ cells: switching from oogenesis to spermatogenesis. Development 129, 1155–1164 (2002).
Koopman, P., Gubbay, J., Vivian, N., Goodfellow, P. & Lovell-Badge, R. Male development of chromosomally female mice transgenic for Sry. Nature 351, 117–121 (1991).
Taketo, T. The role of sex chromosomes in mammalian germ cell differentiation: can the germ cells carrying X and Y chromosomes differentiate into fertile oocytes? Asian J. Androl. 17, 360–366 (2015).
Taketo, T. & Naumova, A. K. Oocyte heterogeneity with respect to the meiotic silencing of unsynapsed X chromosomes in the XY female mouse. Chromosoma 122, 337–349 (2013).
Alton, M., Lau, M. P., Villemure, M. & Taketo, T. The behavior of the X- and Y-chromosomes in the oocyte during meiotic prophase in the B6.Y(TIR)sex-reversed mouse ovary. Reproduction 135, 241–252 (2008).
Hikabe, O. et al. Reconstitution in vitro of the entire cycle of the mouse female germ line. Nature 539, 299–303 (2016).
Hamada, N. et al. Germ cell-intrinsic effects of sex chromosomes on early oocyte differentiation in mice. PLoS Genet. 16, e1008676 (2020).
Yamamoto, S. et al. Rapid selection of XO embryonic stem cells using Y chromosome-linked GFP transgenic mice. Transgenic Res. 23, 757–765 (2014).
Deng, J. M. et al. Generation of viable male and female mice from two fathers. Biol. Reprod. 84, 613–618 (2011).
Eggan, K. et al. Male and female mice derived from the same embryonic stem cell clone by tetraploid embryo complementation. Nat. Biotechnol. 20, 455–459 (2002).
Rudd, M. K. et al. Monosomy X rescue explains discordant NIPT results and leads to uniparental isodisomy. Prenat. Diagn. 38, 920–923 (2018).
Luo, Y. et al. Uniparental disomy of the entire X chromosome in Turner syndrome patient-specific induced pluripotent stem cells. Cell Discov. 1, 15022 (2015).
Denes, A. M., Landin-Wilhelmsen, K., Wettergren, Y., Bryman, I. & Hanson, C. The proportion of diploid 46,XX cells increases with time in women with Turner syndrome-a 10-year follow-up study. Genet. Test. Mol. Biomarkers 19, 82–87 (2015).
Bolton, H. et al. Mouse model of chromosome mosaicism reveals lineage-specific depletion of aneuploid cells and normal developmental potential. Nat. Commun. 7, 11165 (2016).
Nakai-Futatsugi, Y. & Niwa, H. Zscan4 is activated after telomere shortening in mouse embryonic stem cells. Stem Cell Rep. 6, 483–495 (2016).
Hayashi, K. et al. Offspring from oocytes derived from in vitro primordial germ cell-like cells in mice. Science 338, 971–975 (2012).
Codner, G. F. et al. Aneuploidy screening of embryonic stem cell clones by metaphase karyotyping and droplet digital polymerase chain reaction. BMC Cell Biol. 17, 30 (2016).
Gaztelumendi, N. & Nogues, C. Chromosome instability in mouse embryonic stem cells. Sci Rep. 4, 5324 (2014).
Roper, R. J., St John, H. K., Philip, J., Lawler, A. & Reeves, R. H. Perinatal loss of Ts65Dn Down syndrome mice. Genetics 172, 437–443 (2006).
Hayashi, K., Ohta, H., Kurimoto, K., Aramaki, S. & Saitou, M. Reconstitution of the mouse germ cell specification pathway in culture by pluripotent stem cells. Cell 146, 519–532 (2011).
Severance, A. L., Midic, U. & Latham, K. E. Genotypic divergence in mouse oocyte transcriptomes: possible pathways to hybrid vigor impacting fertility and embryogenesis. Physiol. Genomics 52, 96–109 (2020).
Xie, B. et al. Poly(ADP-ribose) mediates asymmetric division of mouse oocyte. Cell Res. 28, 462–475 (2018).
Zuo, E. et al. CRISPR/Cas9-mediated targeted chromosome elimination. Genome Biol. 18, 224 (2017).
Choi, J. et al. Prolonged Mek1/2 suppression impairs the developmental potential of embryonic stem cells. Nature 548, 219–223 (2017).
Zhang, M. et al. Aneuploid embryonic stem cells exhibit impaired differentiation and increased neoplastic potential. EMBO J. 35, 2285–2300 (2016).
Hirota, T. et al. Fertile offspring from sterile sex chromosome trisomic mice. Science 357, 932–935 (2017).
Liu, P. et al. Passage number is a major contributor to genomic structural variations in mouse iPSCs. Stem Cells 32, 2657–2667 (2014).
Young, M. A. et al. Background mutations in parental cells account for most of the genetic heterogeneity of induced pluripotent stem cells. Cell Stem Cell 10, 570–582 (2012).
Liang, Q., Conte, N., Skarnes, W. C. & Bradley, A. Extensive genomic copy number variation in embryonic stem cells. Proc. Natl Acad. Sci. USA 105, 17453–17456 (2008).
Li, Z. K. et al. Generation of bimaternal and bipaternal mice from hypomethylated haploid ESCs with imprinting region deletions. Cell Stem Cell 23, 665–676.e664 (2018).
Vaz, B., El Mansouri, F., Liu, X. & Taketo, T. Premature ovarian insufficiency in the XO female mouse on the C57BL/6J genetic background. Mol. Hum. Reprod. 26, 678–688 (2020).
Ishikura, Y. et al. In vitro derivation and propagation of spermatogonial stem cell activity from mouse pluripotent stem cells. Cell Rep. 17, 2789–2804 (2016).
Ohta, H. et al. In vitro expansion of mouse primordial germ cell-like cells recapitulates an epigenetic blank slate. EMBO J. 36, 1888–1907 (2017).
Ying, Q. L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008).
Hayashi, K. & Saitou, M. Generation of eggs from mouse embryonic stem cells and induced pluripotent stem cells. Nat. Protoc. 8, 1513–1524 (2013).
D’Hulst, C., Parvanova, I., Tomoiaga, D., Sapar, M. L. & Feinstein, P. Fast quantitative real-time PCR-based screening for common chromosomal aneuploidies in mouse embryonic stem cells. Stem Cell Rep. 1, 350–359 (2013).
Scheinin, I. et al. DNA copy number analysis of fresh and formalin-fixed specimens by shallow whole-genome sequencing with identification and exclusion of problematic regions in the genome assembly. Genome Res. 24, 2022–2032 (2014).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Danecek, P. et al. Twelve years of SAMtools and BCFtools. Gigascience 10, giab008 (2021).
Hayashi, K., Hikabe, O., Obata, Y. & Hirao, Y. Reconstitution of mouse oogenesis in a dish from pluripotent stem cells. Nat. Protoc. 12, 1733–1744 (2017).
Kim, D., Paggi, J. M., Park, C., Bennett, C. & Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 37, 907–915 (2019).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Sasaki, K. et al. Robust in vitro induction of human germ cell fate from pluripotent stem cells. Cell Stem Cell 17, 178–194 (2015).
Acknowledgements
We thank K. Kitajima and C. Meno for providing microscopes; N. Konishi for technical support; H. Toh for advice about DNA-seq; and Research Support Center, Research Center for Human Disease Modeling, Kyushu University Graduate School of Medical Sciences for technical assistance. This study was supported in part by Grant-in-Aid for Scientific Research on Innovative Areas from JSPS (grants 18H05544 and 18H05545 to K.H. and 18H05549 to T.S.K.); by a Grant-in-Aid for Specially Promoted Research from JSPS (grants 17H06098 and 22H04920 to M.S.); by the Takeda Science Foundation (K.H.); by the Luca Bella Foundation (K.H.); and by a Grant-in-Aid from The Open Philanthropy Project (K.H.).
Author information
Authors and Affiliations
Contributions
K.H. conceived the project. K.M., N. Hamazaki and K.H. performed the investigation. N. Hamazaki, T.S.K. and I.O. contributed the methodology. K.M. performed the DNA-seq and RNA-seq analyses. N. Hamada, G.N., H.O., Y.N., Y.I., Y.S., Y.K., F.A., K.A. and M.S. provided the resources. K.K. and K.H. supervised the project. K.M. and K.H. wrote the manuscript, incorporating feedback from all the authors.
Corresponding author
Ethics declarations
Competing interests
K.H. is an inventor on patent applications relating to in vitro oocyte production in mice filed by Kyushu University.
Peer review
Peer review information
Nature thanks Diana Laird and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer review reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Generation of XX ES cells from XO ES cells with reversine.
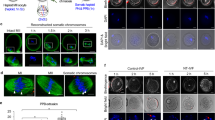
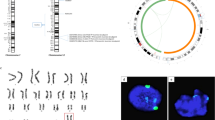
(a) Knock-in of DsRed into the X chromosome. The schematic diagram shows the gRNA position, primers for genotyping (arrows), and expected sizes of the amplicons. The right image shows the result of PCR using the primers. The black dots indicate the ES cell lines used in this study. M, size marker. W, wild type. (b) Removal of the Y chromosome. Shown are the results of PCR analysis of sex chromosomes. Each sex chromosome is indicated at the left side of the image. Black dots indicate XO ES cell clones. The lane with an asterisk was excluded from the count because the band was faint. (c) Effect of reversine on the proliferation of ES cells at each passage. The results of three independent experiments are shown as mean±s.e.m. (d) The effect of reversine treatment on the proliferation of ES cells at day 2. The results of three independent experiments are shown as mean±s.e.m. P value was determined by One-way ANOVA with Bonferroni adjustment. (e) Effect of reversine on DsRed intensity of XRO ES cells in the total cell population and G1 population. The top row shows the DsRed intensity of the total cell population cultured without (Control) or with reversine at each concentration. The numbers show the percentage of the cell population at each reversine concentration. The second row shows the overlay of the DsRed intensity of the total cell population cultured without (grey) and with reversine (red) at each concentration. The third row shows the overlay of the cell size measured by forward scatter (FSC) of the total cell population cultured without (grey) and with reversine (red) at each concentration. The fourth row shows the DNA content in the total cell population cultured without (Control) or with reversine at each concentration. The G1 population in each plot was analyzed in the fifth and bottom rows. The fifth row shows the overlay of the DsRed intensity of the G1 population cultured without (grey) and with reversine (red) at each concentration. The bottom row shows the overlay of the cell size measured by forward scatter (FSC) of the G1 population cultured without (grey) and with reversine (red) at each concentration. Note that DsRed-highly positive cells increased in proportion to the concentration of reversine in both the total cell and G1 populations, whereas the cell size became diverse in both the small and large fractions. (f) DNA-seq analysis of sex-converted ES cell clones. Shown are the results of sex-converted ES cell clones apart from the ES cell clone shown in Fig. 1e. Two copies are plotted at 0 in log2 ratio. The numbers below the plot are the chromosome numbers.
Extended Data Fig. 2 PGCL cell induction and transcriptome analysis of oocytes from the sex-converted XX ES cells.
(a) PGCL cell induction from XRY, XRO and XRXR ES cells. The numbers in the FACS analysis images are the percentages of PGCL cells. Scale bars, 200 µm. (b) H3K27me3 states indicative of X chromosome states during PGCL cell induction. Images show immunofluorescence analysis of ES cells and PGCL cells at day 3 of induction (d3 PGCL cells). Note that single H3K27me3 bright spots corresponding to Xi, were observed in cells at day 3 of PGCL cell induction. Scale bars, 10 μm. The right graph shows the percentage of cells with the indicated H3K27me3 state in BV positive and negative cells at day 3 of PGCL cell induction. These results are based on biologically duplicated samples. (c) Gene expression analysis of GV (left) and MII (right) oocytes differentiated from native XX ES cells (BVSCH18) and XRXR ES cells (BVSCN2-4-63R2). Genes expressed more than twofold higher (red) or twofold lower (blue) in XRXR oocytes are shown. The FDR cutoff is 0.01. Biological duplicate samples.
Extended Data Fig. 3 Reversine effect on trisomy 16 ES cells and oocyte production from trisomy 16 XY and XO ES cells.
(a) DNA-seq analysis of a mosaic trisomy 16 clone. (b) DNA-seq analysis of trisomy 16 XRO ES cells. Another trisomy 16 XRO cell line is shown in Fig. 2b. (c) Q-PCR analysis to determine the X chromosome number. Blue and orange dots show the X chromosome content determined using chromosome 7 and chromosome 11, respectively, as references. Details are described in Fig. 1c. The genotypes of sex chromosomes and individual clone IDs are shown below the graph. (d) DNA-seq analysis of FACS sorted trisomy 16 XRO ES cells without (control) or with reversine treatment. The left column shows the whole population, and the right column shows the top 5% fraction of the DsRed-high population. (e) Log2 ratio of autosomes and the X chromosome under each condition in the DNA-seq analysis. Note that the ratio of XRXR ES cells was significantly increased in the top 5% fraction compared to the total cell fraction with 0.02 µM reversine, while autosomes were relatively unaffected. There was no significant difference (P > 0.11) in the ratio of XRXR ES cells in the top 5% fraction without and with 0.02 µM reversine, consistent with the results shown in Fig. 1b. P value was determined by One-way ANOVA with Bonferroni adjustment. The values were compiled from biologically triplicated experiments. (f) PGCL cell induction from trisomy 16 XRY ES cells and trisomy 16 XRO ES cells. Shown are representative images and FACS analyses of the induction of PGCL cells from the ES cells indicated. The numbers in the FACS analysis are the percentages of PGCL cells. Scale bars, 200 µm. (g) IVDi culture using PGCL cells from trisomy 16 XRY ES cells and trisomy 16 XRO ES cells. Representative images of the rOvaries at the days indicated are shown. Scale bars, 200 µm. Similar results were obtained in biologically duplicated experiments.
Extended Data Fig. 4 Generation of XX iPS cells from XY iPS cells followed by oocyte production.
(a) Genotype of XY iPS cells used in this study. Shown are the results of PCR analysis of sex chromosomes (top) and knock-in of DsRed into the X chromosome (bottom). Details for the knock-in are shown in Extended Data Fig. 1a. All six iPS cell lines tested had X and Y chromosomes (top). The black dot in each image indicates the iPS cell clone used in this study. The knock-in alleles were detected in four of the six iPS cell lines. The same results were obtained in PCR experiment repeated twice. (b) Removal of the Y chromosome. Shown are the results of PCR analysis of sex chromosomes after 9 passages. The black dot indicates XRO iPS cells used in this study. The same results were obtained in PCR experiment repeated twice. (c) Q-PCR analysis to determine the X chromosome number. Blue and orange dots show the X chromosome content determined using chromosome 7 and chromosome 11, respectively, as references. Details are described in Fig. 1c. The genotypes of sex chromosomes and individual clone IDs are shown below the graph. (d) DNA-seq analysis of sex-converted iPS cell clones. Two copies are plotted at 0 in log2 ratio. The numbers below the plot are the chromosome numbers. The genotypes and the IDs of the individual clones are displayed on the left side of the plot. (e) PGCL cell induction from XRY, XRO and XRXR iPS cells. Shown are representative images and FACS analyses of the induction of PGCL cells from the iPS cells indicated. The numbers in the FACS analysis are the percentages of PGCL cells. Scale bars, 200 µm. Similar results were obtained in biologically duplicated experiments. (f) IVDi culture using PGCL cells from XRY, XRO and XRXR iPS cells. Shown are representative images of the rOvaries at the days indicated. As a negative control, a representative rOvary containing only embryonic gonadal somatic cells is shown. The images at day 21 of culture are the same as in Fig. 3b. Scale bars, 200 µm. Similar results were obtained in biologically duplicated experiments.
Extended Data Fig. 5 Derivation of offspring from a single iPS cell line.
(a) Chimeric mice harbouring XRY or sex-converted XRXR iPS cells. The graph below the images shows the ratio of iPS cell chimeric mice. The number in the top graph is the percentage of male chimeras harbouring XRY iPS cells and the numbers in the middle and bottom graphs are the percentages of female chimeras harbouring XRXR iPS cells. (b) Offspring from chimeric mice harbouring XRY or sex-converted XRXR iPS cells. The top images show offspring from male chimeric mice harbouring XRY iPS cells (left) and offspring female chimeric mice harbouring XRXR iPS cells (right). The middle graph shows the coat colour of offspring from a mating between chimeric mice and wild-type albino mice. The number in the graph is the percentage of non-albino mice from a mating between chimeric mice and wild type albino mice. The bottom gel image show genotyping of the offspring from iPS cell chimeric mice harbouring XRY iPS cells (left) or sex-converted XRXR iPS cells (right). PCR products from BV and DsRed transgenes, exogenous retroviral Sox2 and Pou5f1, and the Prdm 14 locus as a positive control are shown. The coat colour of number 10 was albino. P, positive control. N, negative control. Details of the positive control and negative control are described in Fig. 3d. (c) Pups from a mating between an XRY iPS cell-chimeric male and XRXR iPS cell-chimeric female mouse. (d) FACS analysis of H-2K haplotypes of offspring obtained from crosses between the chimeric mice. Shown are the FACS plot of H-2K haplotypes in lymphocytes of C57BL/6J (H-2Kb), 129X1/SvJ (H-2Kb), Balb/c (H-2Kd), and F1 mice from a mating between chimeric mice. Note that one of the F1 mice had only H-2Kb, whereas the other had a hybrid haplotype. (e) SNP analysis of offspring. Shown are SNPs at rs3022980 and rs3022981 in C57BL/6J, 129X1/SvJ, BALB/c and F1 mice from a mating between chimeric mice. (f) Ratio of homozygous F1 from a mating between an XRY iPS cell-chimeric mouse and XRXR iPS cell-chimeric mouse. (g) An adult mouse from (c) at 10 months after birth.
Extended Data Fig. 6 Generation of XX ES cells from XO ES cells using anti-CD38 antibodies.
(a) FACS analysis of CD38 expression in C57BL/6J (originally XX) ES cells. Note that there are two types of CD38 expression: low and high. (b) FACS analysis of DsRed and CD38 in XRO and XRXR ES cells. Note that the CD38 intensity was increased in XRXR ES cells in proportion to the intensity of DsRed. (c) The gates used to select XX ES cells from XO ES cells using anti-CD38 antibodies. The gate at left (solid line) was used for the CD38-high cell population and the gate at right (dashed line) was used for the CD38-extremely high cell population. (d) Q-PCR analysis to determine the X chromosome number. The left graph shows the results using BVSC XO ES cells with high CD38 (the solid gate in (c)) and the right graph shows the results using BVSC XO ES cells with extremely high CD38 (the dashed gate in (c)). (e) The graph shows the results using non-reporter XO ES cells with high CD38. The genotypes of sex chromosomes and individual clone IDs are shown below the graph. Blue and orange dots show the X chromosome content determined using chromosome 7 and chromosome 11, respectively, as references. Details are described in Fig. 1c. (f) FACS analysis of PGCL cells derived from non-reporter and X-duplicated ESCs. PGCL cells were identified by antibodies against SSEA1 and CD6126. Similar results were obtained in biologically duplicated experiments.
Supplementary information
Supplementary Information
This file contains Supplementary Figs. 1–7 and Supplementary Tables 1 and 2.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Murakami, K., Hamazaki, N., Hamada, N. et al. Generation of functional oocytes from male mice in vitro. Nature 615, 900–906 (2023). https://doi.org/10.1038/s41586-023-05834-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-05834-x
This article is cited by
-
Will these reprogrammed elephant cells ever make a mammoth?
Nature (2024)
-
Ethical and legal challenges in assisted same-sex conception through in vitro gametogenesis
Nature Medicine (2024)
-
Eggs made from male mouse stem cells using error-prone culture
Nature (2023)
-
Human embryo science: can the world’s regulators keep pace?
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.