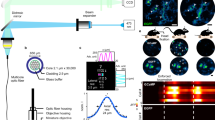
Abstract
Astrocytic calcium dynamics has been implicated in the encoding of sensory information1,2,3,4,5, and modulation of calcium in astrocytes has been shown to affect behaviour6,7,8,9,10. However, longitudinal investigation of the real-time calcium activity of astrocytes in the hippocampus of awake mice is lacking. Here we used two-photon microscopy to chronically image CA1 astrocytes as mice ran in familiar or new virtual environments to obtain water rewards. We found that astrocytes exhibit persistent ramping activity towards the reward location in a familiar environment, but not in a new one. Shifting the reward location within a familiar environment also resulted in diminished ramping. After additional training, as the mice became familiar with the new context or new reward location, the ramping was re-established. Using linear decoders, we could predict the location of the mouse in a familiar environment from astrocyte activity alone. We could not do the same in a new environment, suggesting that the spatial modulation of astrocytic activity is experience dependent. Our results indicate that astrocytes can encode the expected reward location in spatial contexts, thereby extending their known computational abilities and their role in cognitive functions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All source data are provided with the paper. All datasets are provided at GitHub (https://github.com/GoshenLab/Astro_imaging/). Source data are provided with this paper.
Code availability
The custom code used in this paper is provided at GitHub (https://github.com/GoshenLab/Astro_imaging/).
References
Bazargani, N. & Attwell, D. Astrocyte calcium signaling: the third wave. Nat. Neurosci. 19, 182–189 (2016).
Schummers, J., Yu, H. & Sur, M. Tuned responses of astrocytes and their influence on hemodynamic signals in the visual cortex. Science 320, 1638–1643 (2008).
Slezak, M. et al. Distinct mechanisms for visual and motor-related astrocyte responses in mouse visual cortex. Curr. Biol. 29, 3120–3127 (2019).
Wang, X. et al. Astrocytic Ca2+ signaling evoked by sensory stimulation in vivo. Nat. Neurosci. 9, 816–823 (2006).
Stobart, J. L. et al. Cortical circuit activity evokes rapid astrocyte calcium signals on a similar timescale to neurons. Neuron 98, 726–735 (2018).
Nagai, J. et al. Behaviorally consequential astrocytic regulation of neural circuits. Neuron 109, 576–596 (2021).
Adamsky, A. & Goshen, I. Astrocytes in memory function: pioneering findings and future directions. Neuroscience 370, 14–26 (2018).
Yu, X. et al. Reducing astrocyte calcium signaling in vivo alters striatal microcircuits and causes repetitive behavior. Neuron 99, 1170–1187 (2018).
Kol, A. et al. Astrocytes contribute to remote memory formation by modulating hippocampal-cortical communication during learning. Nat. Neurosci. 23, 1229–1239 (2020).
Wahis, J. et al. Astrocytes mediate the effect of oxytocin in the central amygdala on neuronal activity and affective states in rodents. Nat. Neurosci. 24, 529–541 (2021).
Volterra, A., Liaudet, N. & Savtchouk, I. Astrocyte Ca2+ signalling: an unexpected complexity. Nat. Rev. Neurosci. 15, 327–335 (2014).
Thrane, A. S. et al. General anesthesia selectively disrupts astrocyte calcium signaling in the awake mouse cortex. Proc. Natl Acad. Sci. USA 109, 18974–18979 (2012).
Curreli, S., Bonato, J., Romanzi, S., Panzeri, S. & Fellin, T. Complementary encoding of spatial information in hippocampal astrocytes. PLoS Biol. 20, e3001530 (2022).
Adamsky, A. et al. Astrocytic activation generates de novo neuronal potentiation and memory enhancement. Cell 174, 59–71 (2018).
Srinivasan, R. et al. Ca2+ signaling in astrocytes from Ip3r2−/− mice in brain slices and during startle responses in vivo. Nat. Neurosci. 18, 708–717 (2015).
Paukert, M. et al. Norepinephrine controls astroglial responsiveness to local circuit activity. Neuron 82, 1263–1270 (2014).
Vaidyanathan, T. V., Collard, M., Yokoyama, S., Reitman, M. E. & Poskanzer, K. E. Cortical astrocytes independently regulate sleep depth and duration via separate GPCR pathways. eLife 10, e63329 (2021).
Corkrum, M. et al. Dopamine-evoked synaptic regulation in the nucleus accumbens requires astrocyte activity. Neuron 105, 1036–1047 (2020).
Navarrete, M. et al. Astrocytes mediate in vivo cholinergic-induced synaptic plasticity. PLoS Biol. 10, e1001259 (2012).
O'Keefe, J. & Dostrovsky, J. The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Res. 34, 171–175 (1971).
Sosa, M. & Giocomo, L. M. Navigating for reward. Nat. Rev. Neurosci. 22, 472–487 (2021).
Xu, H., Baracskay, P., O’Neill, J. & Csicsvari, J. Assembly responses of hippocampal CA1 place cells predict learned behavior in goal-directed spatial tasks on the radial eight-arm maze. Neuron 101, 119–132 (2019).
Hollup, S. A., Molden, S., Donnett, J. G., Moser, M. B. & Moser, E. I. Accumulation of hippocampal place fields at the goal location in an annular watermaze task. J. Neurosci. 21, 1635–1644 (2001).
Dupret, D., O'Neill, J., Pleydell-Bouverie, B. & Csicsvari, J. The reorganization and reactivation of hippocampal maps predict spatial memory performance. Nat. Neurosci. 13, 995–1002 (2010).
Zaremba, J. D. et al. Impaired hippocampal place cell dynamics in a mouse model of the 22q11.2 deletion. Nat. Neurosci. 20, 1612–1623 (2017).
Sato, M. et al. Distinct mechanisms of over-representation of landmarks and rewards in the hippocampus. Cell Rep. 32, 107864 (2020).
Danielson, N. B. et al. Sublayer-specific coding dynamics during spatial navigation and learning in hippocampal area CA1. Neuron 91, 652–665 (2016).
Gauthier, J. L. & Tank, D. W. A dedicated population for reward coding in the hippocampus. Neuron 99, 179–193 (2018).
Rubin, A., Geva, N., Sheintuch, L. & Ziv, Y. Hippocampal ensemble dynamics timestamp events in long-term memory. eLife 4, e12247 (2015).
Dong, C., Madar, A. D. & Sheffield, M. E. J. Distinct place cell dynamics in CA1 and CA3 encode experience in new environments. Nat. Commun. 12, 2977 (2021).
McNamara, C. G., Tejero-Cantero, A., Trouche, S., Campo-Urriza, N. & Dupret, D. Dopaminergic neurons promote hippocampal reactivation and spatial memory persistence. Nat. Neurosci. 17, 1658–1660 (2014).
Hainmueller, T. & Bartos, M. Parallel emergence of stable and dynamic memory engrams in the hippocampus. Nature 558, 292–296 (2018).
Allegra, M., Posani, L., Gomez-Ocadiz, R. & Schmidt-Hieber, C. Differential relation between neuronal and behavioral discrimination during hippocampal memory encoding. Neuron 108, 1103–1112 (2020).
Turi, G. F. et al. Vasoactive intestinal polypeptide-expressing interneurons in the hippocampus support goal-oriented spatial learning. Neuron 101, 1150–1165 (2019).
Mederos, S. et al. Melanopsin for precise optogenetic activation of astrocyte-neuron networks. Glia 67, 915–934 (2019).
Wang, Y. et al. Accurate quantification of astrocyte and neurotransmitter fluorescence dynamics for single-cell and population-level physiology. Nat. Neurosci. 22, 1936–1944 (2019).
Dombeck, D. A., Khabbaz, A. N., Collman, F., Adelman, T. L. & Tank, D. W. Imaging large-scale neural activity with cellular resolution in awake, mobile mice. Neuron 56, 43–57 (2007).
Li, N., Daie, K., Svoboda, K. & Druckmann, S. Robust neuronal dynamics in premotor cortex during motor planning. Nature 532, 459–464 (2016).
Bromberg-Martin, E. S., Matsumoto, M. & Hikosaka, O. Distinct tonic and phasic anticipatory activity in lateral habenula and dopamine neurons. Neuron 67, 144–155 (2010).
Hanks, T. D. et al. Distinct relationships of parietal and prefrontal cortices to evidence accumulation. Nature 520, 220–223 (2015).
Maimon, G. & Assad, J. A. A cognitive signal for the proactive timing of action in macaque LIP. Nat. Neurosci. 9, 948–955 (2006).
Murakami, M., Vicente, M. I., Costa, G. M. & Mainen, Z. F. Neural antecedents of self-initiated actions in secondary motor cortex. Nat. Neurosci. 17, 1574–1582 (2014).
Kim, H. R. et al. A unified framework for dopamine signals across timescales. Cell 183, 1600–1616 (2020).
Howe, M. W., Tierney, P. L., Sandberg, S. G., Phillips, P. E. M. & Graybiel, A. M. Prolonged dopamine signalling in striatum signals proximity and value of distant rewards. Nature 500, 575–579 (2013).
Mu, Y. et al. Glia accumulate evidence that actions are futile and suppress unsuccessful behavior. Cell 178, 27–43 (2019).
Aronov, D., Nevers, R. & Tank, D. W. Mapping of a non-spatial dimension by the hippocampal-entorhinal circuit. Nature 543, 719–722 (2017).
Terada, S., Sakurai, Y., Nakahara, H. & Fujisawa, S. Temporal and rate coding for discrete event sequences in the hippocampus. Neuron 94, 1248–1262 (2017).
Radvansky, B. A. & Dombeck, D. A. An olfactory virtual reality system for mice. Nat. Commun. 9, 839 (2018).
Bittner, K. C., Milstein, A. D., Grienberger, C., Romani, S. & Magee, J. C. Behavioral time scale synaptic plasticity underlies CA1 place fields. Science 357, 1033–1036 (2017).
Deemyad, T., Luthi, J. & Spruston, N. Astrocytes integrate and drive action potential firing in inhibitory subnetworks. Nat. Commun. 9, 4336 (2018).
Hippenmeyer, S. et al. A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol. 3, e159 (2005).
Taniguchi, H. et al. A resource of Cre driver lines for genetic targeting of GABAergic neurons in cerebral cortex. Neuron 71, 995–1013 (2011).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Pnevmatikakis, E. A. & Giovannucci, A. NoRMCorre: an online algorithm for piecewise rigid motion correction of calcium imaging data. J. Neurosci. Methods 291, 83–94 (2017).
Dombeck, D. A., Harvey, C. D., Tian, L., Looger, L. L. & Tank, D. W. Functional imaging of hippocampal place cells at cellular resolution during virtual navigation. Nat. Neurosci. 13, 1433–1440 (2010).
Markus, E. J., Barnes, C. A., McNaughton, B. L., Gladden, V. L. & Skaggs, W. E. Spatial information content and reliability of hippocampal CA1 neurons: effects of visual input. Hippocampus 4, 410–421 (1994).
Radvansky, B. A., Oh, J. Y., Climer, J. R. & Dombeck, D. A. Behavior determines the hippocampal spatial mapping of a multisensory environment. Cell Rep. 36, 109444 (2021).
Acknowledgements
We thank all of the members of the Goshen laboratory for their support; and A. Citri, Y. Burak, Y. Loewenstein, E. Malach, A. Kaduri Amichai, A. Adamsky and A. Kol for reading the manuscript. A.D. is supported by the Azrieli fellowship and the ELSC graduate students' scholarship. This project has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant agreement no. 803589), the Israel Science Foundation (ISF grant no. 1815/18) and the Canada-Israel grants (CIHR-ISF, grant no. 2591/18).
Author information
Authors and Affiliations
Contributions
A.D. performed all of the experiments with help from A.B.-C., N.B., T.C., R.R., N.N. and T.K., and performed the analysis with help from A.R. and Y.Z.; A.D. and I.G. wrote the manuscript with assistance from A.R. and Y.Z.; I.G. supervised all aspects of the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Caswell Barry and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Apparatus for Imaging Astrocytes in Behaving Mice.
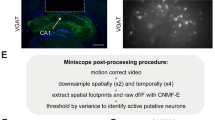
A. Two environments consisting of different belts with tactile cues and virtual reality displays. B. Selective expression of GCaMP6f in CA1 astrocytes following injection of AAV-GFAP::cyto-GCaMP6f to an Ai14XSST-Cre mouse. GCaMP6f was expressed in > 92% of CA1 astrocytes (212/230 cells from 3 mice; 92.73% ± 1.65 of GFAP positive cells were also GCaMP6f positive), with > 94% specificity (212/224 cells from 3 mice; 94.58% ± 0.29 of GCaMP6f positive cells were also GFAP positive). Minimal co-localization with SST or PV positive cells or the neuronal marker NeuN was detected (1.65% expression in neurons, 11/668 cells; 1.55% ± 0.54 of GCaMP6f positive cells were also NeuN positive; scale bars: 50 µm). C. ROI centroid distance was negatively correlated with the mean pairwise event correlation (pooled data from n = 8 mice). D. ROI size was positively correlated with mean event probability (pooled data from n = 8 mice). Data presented as mean±SEM.
Extended Data Fig. 2 Astrocytic Activity Explains Location More Accurately Than Velocity.
A. The mean number of concurrent astrocytic events following reward delivery in all laps of the same mouse shown in Fig. 2a–c. B. Mean normalized number of concurrent events as a function of time following reward delivery in all mice presented in Fig. 2d (blue is the one from A), showing significant reduction over time (Pearson’s r: −0.4 ± 0.04, 1-sided permutation tests, p ≤ 0.006 in all 9 mice). C. The mean normalized ΔF/F as a function of binned location, showing significant ramping in all mice shown in Fig. 2d (Pearson’s r: 0.25 ± 0.03, 1-sided permutation tests, p ≤ 0.018 in all 9 mice). D-E. Both the astrocytic somata and processes show significant ramping. D. The mean normalized number of concurrent events as a function of binned location calculated separately for the somata and processes of the mouse shown in Fig. 2a–c (n = 59 somata and n = 67 processes). E. The correlation between location and concurrent events in the somata is not significantly different from the processes (Pearson’s r: 0.36 ± 0.03 and 0.38 ± 0.03, 1-sided permutation tests, p ≤ 0.014 and p ≤ 0.025 in the somata and processes respectively, n = 424 somata and n = 455 processes from n = 8 mice; 2-sided paired t-test, t(7) = 0.92, p = 0.39). F. The mean normalized number of concurrent events as a function of binned location when the reward was given in random locations along the track, showing no apparent ramping (Pearson’s r: 0.02 ± 0.02, 1-sided permutation tests, p ≥ 0.084 for all 3 mice; The dashed square denotes the previously learnt constant reward location). G. The mean normalized number of concurrent events as a function of binned location when the VR display was turned off, showing significant ramping in most mice (Pearson’s r: 0.2 ± 0.04, 1-sided permutation test, p ≤ 0.009, n = 3 mice; Pearson’s r: 0.11, 1-sided permutation test, p = 0.075, n = 1 mouse). H. Same as Fig. 2f, calculated using the ΔF/F traces, showing ramping towards the reward location in many ROIs with significant spatial information. I. The mean number of concurrent events of the mouse shown in Fig. 2a–c as a function of binned normalized velocities in all laps. Grey bins denote no samples. J. Mean number of concurrent events as a function of binned normalized velocities, normalized by shuffled data in all mice presented in Fig. 2d (blue is the one from A), (Pearson’s r: 0.17 ± 0.04, 1-sided permutation test, p ≥ 0.084 in n = 4 mice, p ≤ 0.025 in n = 5 mice). K. The mean number of concurrent events as a function of location and normalized velocity in the mouse shown in Fig. 2a–c. Ramping is more prominent across locations than velocities. L. The mean STD of the astrocytic population activity across locations for a given velocity (STDlocations|velocity) weighted by the time spent in the location x velocity is significantly larger than vice versa (STDvelocities|location)(STDlocations|velocity: 3.62 ± 0.44, STDvelocities|location: 7.37 ± 0.74, 2-sided paired t-test, n = 9 mice, t(8) = 6.56, p = = 0.0002). M. The mean distribution of the difference between the mean weighted STDlocations|velocity and STDvelocities|location for single ROIs from the 9 mice shown in Fig. 2d. Most ROIs vary more across locations than across velocities. N-O. General linear models were used to fit the number of concurrent astrocytic events as a linear function of different behavioural variables, showing that location had a unique contribution to the variance of astrocytic activity. N. The model performance was significantly better when it was fitted on the actual location data compared to shuffled location (coefficient of determination (r2) between the model prediction and the actual number of concurrent events: 0.25 ± 0.04 and 0.18 ± 0.03 for real and shuffled location inputs respectively, 1-sided permutation test, p ≤ 0.027 in n = 8 mice, p = 0.35 in n = 1 mouse). O. Cross-validated models that included location as their input performed significantly better than reduced models without it (r2: 0.21 ± 0.04 and 0.15 ± 0.03 in full and reduced models respectively, 1-sided permutation test, p < 0.004 in n = 8 mice, p = 0.065 in n = 1 mouse). Data presented as mean (bold line) ±SEM (shaded area). Different mice are colour-coded as in Fig. 2d.
Extended Data Fig. 3 Astrocytic Activity Does Not Ramp Towards Rewarding Location in a New Environment.
A. The correlation between location and astrocytic activity in repeated active ROIs was significantly higher in the familiar environment compared to the new one (Pearson’s r: 0.41 ± 0.03 and 0.2 ± 0.05 in the familiar or new environment, respectively, n = 7 mice, 2-sided paired t-test, t(6) = 8.04, p = 0.0002). B. The repeated active ROI pairs that had significant mutual information (MI) in each environment of the mouse shown in Fig. 3a, b. C. The mean proportion of significant MI repeated active ROI pairs in the familiar environment is significantly higher than in the new environment (0.39 ± 0.08 and 0.16 ± 0.04 in the familiar and new environment, respectively, 2-sided paired t-test, n = 7 mice, t(6) = 2.45, p = 0.0495). D. The mean normalized number of concurrent events as a function of location in the familiar environment and (E) in the new environment for all active ROIs, not just the repeated ones, in the 7 mice shown in Fig. 3c, d. F. The ramping of astrocytic activity is significantly larger in the familiar environment than in the new environment (Pearson’s r: 0.44 ± 0.03 and 0.16 ± 0.03 in the familiar or novel environment, respectively, 2-sided independent samples t-test, n = 7 mice, t(12) = 6.11, p = 0.00005). G. Two mice were imaged for the third time in the familiar environment after the exposure to the new environment. The mean normalized number of concurrent events as a function of location shows that ramping is maintained (Pearson’s r: 0.31 ± 0.11, 1-sided permutation test, p < 0.01). Data presented as mean (bold line) ±SEM (shaded area). Different mice are colour-coded as in Fig. 2d.
Extended Data Fig. 4 Performance of Mice Location Decoders in Familiar Environment.
A-F. Pooled error cumulative probability plots of the mice that appear in the averaged data in Fig. 4. A. Mean error size: 43.7 ± 0.1 and 54.1 ± 0.1, for the decoder trained on the real data and the shuffled data respectively, 1-sided permutation test, p = 0.009). B. Mean error size: 41.4 ± 0.2 and 61.1 ± 0.3, for the decoder trained on the real data and the shuffled data respectively, 1-sided permutation test, p = 0.027). C. Mean error size: 37.8 ± 0.2 and 56.6 ± 0.2, for the decoder trained on the real data and the shuffled data respectively, 1-sided permutation test, p = 0.009). D. Mean error size: 49.6 ± 0.2 and 60.2 ± 0.2, for the decoder trained on the real data and the shuffled data respectively, 1-sided permutation test, p = 0.151). E. Mean error size: 43.9 ± 0.3 and 62.6 ± 0.2, for the decoder trained on the real data and the shuffled data respectively, 1-sided permutation test, p = 0.037). F. Mean error size: 49.9 ± 0.1 and 62.2 ± 0.2, for the decoder trained on the real data and the shuffled data respectively, 1-sided permutation test, p = 0.061). Data presented as mean (bold line) ±SEM (shaded area). Different mice are colour-coded as in Fig. 2d.
Supplementary information
Supplementary Information
Supplementary Table 1: supplementary statics describing the statistics calculated for each mouse presented throughout the paper.
Supplementary Video 1
Two-photon Ca2+ imaging of CA1 astrocytes in two different FOVs. A video showing astrocytic Ca2+ activity in two FOVs, acquired using a fast-tuneable lens focusing on different depths. Imaging in each FOV was acquired at 7.745 frames per second and is shown after motion correction. The video playback is sped up fourfold compared with the original acquisition rate.
Supplementary Video 2
Ca2+ imaging of numerous CA1 astrocytes during virtual navigation. A video showing the Ca2+ dynamics in CA1 astrocytes (left) and the simultaneously recorded mouse location in the virtual environment (right). As the mouse approaches the known reward location, the astrocytic population gradually increases its activity. A total of 126 ROIs were segmented in the complete dataset, which includes an additional FOV (not shown), and their extracted signal is shown in Fig. 2a. Imaging in each FOV was acquired at 7.745 frames per second and is shown after motion correction. The video playback is sped up fourfold compared with the original acquisition rate.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doron, A., Rubin, A., Benmelech-Chovav, A. et al. Hippocampal astrocytes encode reward location. Nature 609, 772–778 (2022). https://doi.org/10.1038/s41586-022-05146-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-05146-6
This article is cited by
-
Multicore fiber optic imaging reveals that astrocyte calcium activity in the mouse cerebral cortex is modulated by internal motivational state
Nature Communications (2024)
-
Astrocytes in the adult dentate gyrus—balance between adult and developmental tasks
Molecular Psychiatry (2024)
-
Network-level encoding of local neurotransmitters in cortical astrocytes
Nature (2024)
-
Centripetal integration of past events in hippocampal astrocytes regulated by locus coeruleus
Nature Neuroscience (2024)
-
Spatial transcriptomics reveal neuron–astrocyte synergy in long-term memory
Nature (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.