Abstract
Endothermy underpins the ecological dominance of mammals and birds in diverse environmental settings1,2. However, it is unclear when this crucial feature emerged during mammalian evolutionary history, as most of the fossil evidence is ambiguous3,4,5,6,7,8,9,10,11,12,13,14,15,16,17. Here we show that this key evolutionary transition can be investigated using the morphology of the endolymph-filled semicircular ducts of the inner ear, which monitor head rotations and are essential for motor coordination, navigation and spatial awareness18,19,20,21,22. Increased body temperatures during the ectotherm–endotherm transition of mammal ancestors would decrease endolymph viscosity, negatively affecting semicircular duct biomechanics23,24, while simultaneously increasing behavioural activity25,26 probably required improved performance27. Morphological changes to the membranous ducts and enclosing bony canals would have been necessary to maintain optimal functionality during this transition. To track these morphofunctional changes in 56 extinct synapsid species, we developed the thermo-motility index, a proxy based on bony canal morphology. The results suggest that endothermy evolved abruptly during the Late Triassic period in Mammaliamorpha, correlated with a sharp increase in body temperature (5–9 °C) and an expansion of aerobic and anaerobic capacities. Contrary to previous suggestions3,4,5,6,7,8,9,10,11,12,13,14, all stem mammaliamorphs were most probably ectotherms. Endothermy, as a crucial physiological characteristic, joins other distinctive mammalian features that arose during this period of climatic instability28.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The raw datasets used in this study are available in the Supplementary Dataset. Links for CT scan datasets and bony labyrinth 3D meshes obtained from https://www.morphosource.org/ can be found in Supplementary Data 3. Some bird skull measurements and fish lengths were obtained from https://skullsite.com/ and fishbase.org, respectively (Supplementary Data 2). Time calibrations between most extant species were obtained from timetree.org. Body mass and Tb of some extant species were obtained from https://eol.org/traitbank (Supplementary Data 2). Source data are provided with this paper.
Code availability
The R scripts used in this study are available in the Supplementary Dataset.
Change history
09 January 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41586-023-05700-w
References
Lovegrove, B. G. Fires of Life: Endothermy in Birds and Mammals (Yale Univ. Press, 2019).
Benton, M. J. The origin of endothermy in synapsids and archosaurs and arms races in the Triassic. Gondwana Res. 100, 261–289 (2021).
Kubo, T. & Benton, M. J. Tetrapod postural shift estimated from Permian and Triassic trackways. Palaeontology 52, 1029–1037 (2009).
Huttenlocker, A. K. & Farmer, C. G. Bone microvasculature tracks red blood cell size diminution in Triassic mammal and dinosaur forerunners. Curr. Biol. 27, 48–54 (2017).
Jones, K. E., Angielczyk, K. D. & Pierce, S. E. Stepwise shifts underlie evolutionary trends in morphological complexity of the mammalian vertebral column. Nature Commun. 10, 5071 (2019).
Faure-Brac, M. G. & Cubo, J. Were the synapsids primitively endotherms? A palaeohistological approach using phylogenetic eigenvector maps. Phil. Trans. R. Soc. B 375, 20190138 (2020).
Crompton, A. W. et al. Structure of the nasal region of non-mammalian cynodonts and mammaliaforms: speculations on the evolution of mammalian endothermy. J. Vertebr. Paleontol. 37, e1269116 (2017).
Bajdek, P. et al. Microbiota and food residues including possible evidence of pre-mammalian hair in Upper Permian coprolites from Russia. Lethaia 49, 455–477 (2016).
Farmer, C. G. Parental care, destabilizing selection, and the evolution of tetrapod endothermy. Physiology 35, 160–176 (2020).
Rey, K. et al. Oxygen isotopes suggest elevated thermometabolism within multiple Permo–Triassic therapsid clades. eLife 6, e28589 (2017).
Newham, E., Gill, P. G. & Corfe, I. J. New tools suggest a middle Jurassic origin for mammalian endothermy: advances in state‐of‐the‐art techniques uncover new insights on the evolutionary patterns of mammalian endothermy through time. BioEssays 44, 2100060 (2022).
Angielczyk, K. D. & Schmitz, L. Nocturnality in synapsids predates the origin of mammals by over 100 million years. Proc. R. Soc. B 281, 20141642 (2014).
Botha, J. & Huttenlocker, A. in Vertebrate Skeletal Histology and Paleohistology (eds. de Buffrénil, V. et al.) 550–563 (CRC Press, 2021).
Grigg, G. et al. Whole‐body endothermy: ancient, homologous and widespread among the ancestors of mammals, birds and crocodylians. Biol. Rev. 97, 766–801 (2022).
Benoit, J. et al. The sixth sense in mammalian forerunners: variability of the parietal foramen and the evolution of the pineal eye in South African Permo–Triassic eutheriodont therapsids. Acta Palaeontol. Pol. 61, 777–789 (2016).
Benoit, J., Manger, P. R. & Rubidge, B. S. Palaeoneurological clues to the evolution of defining mammalian soft tissue traits. Sci. Rep. 6, 25604 (2016).
Rowe, T. B., Macrini, T. E. & Luo, Z.-X. Fossil evidence on origin of the mammalian brain. Science 332, 955–957 (2011).
Rabbitt, R. D., Damiano, E. R. & Grant, J. W. in The Vestibular System (eds. Highstein, S. M. et al.) 153–201 (Springer, 2004).
David, R. et al. Assessing morphology and function of the semicircular duct system: introducing new in-situ visualization and software toolbox. Sci. Rep. 6, 32772 (2016).
Highstein, S. M., Fay, R. R. & Popper, A. N. The Vestibular System (Springer, 2004).
Angelaki, D. E. & Cullen, K. E. Vestibular system: the many facets of a multimodal sense. Annu. Rev. Neurosci. 31, 125–150 (2008).
Fitzpatrick, R. C., Butler, J. E. & Day, B. L. Resolving head rotation for human bipedalism. Curr. Biol. 16, 1509–1514 (2006).
Ten Kate, J. H. & Kuiper, J. W. The viscosity of the Pike’s endolymph. J. Exp. Biol. 53, 495–500 (1970).
Oman, C. M. in The Vestibular System: Function and Morphology (ed. Gualtierotti, T.) 251–274 (Springer, 1981).
Garland, T. & Albuquerque, R. L. Locomotion, energetics, performance, and behavior: a mammalian perspective on lizards, and vice versa. Integr. Comp. Biol. 57, 252–266 (2017).
Hirt, M. R. et al. A general scaling law reveals why the largest animals are not the fastest. Nat. Ecol. Evol. 1, 1116–1122 (2017).
Jones, G. M. & Spells, K. E. A theoretical and comparative study of the functional dependence of the semicircular canal upon its physical dimensions. Proc. R. Soc. Lond. B 157, 403–419 (1963).
Scotese, C. R., Song, H., Mills, B. J. & van der Meer, D. G. Phanerozoic paleotemperatures: the earth’s changing climate during the last 540 million years. Earth Sci. Rev. 215, 103503 (2021).
Storey, K. B. & Storey, J. M. Metabolic rate depression and biochemical adaptation in anaerobiosis, hibernation and estivation. Quart. Rev. Biol. 65, 145–174 (1990).
Legendre, L. J. & Davesne, D. The evolution of mechanisms involved in vertebrate endothermy. Phil. Trans. R. Soc. B 375, 20190136 (2020).
Woodley, R. & Buffenstein, R. Thermogenic changes with chronic cold exposure in the naked mole-rat (Heterocephalus glaber). Comp. Biochem. Phys. A 133, 827–834 (2002).
Pianka, E. R. in Ecology and Natural History of Desert Lizards: Analyses of the Ecological Niche and Community Structure (ed Pianka, E. R.) 35–47 (Princetown Univ. Press, 1986).
Grigg, G. C., Beard, L. A. & Augee, M. L. The evolution of endothermy and its diversity in mammals and birds. Physiol. Biochem. Zool. 77, 982–997 (2004).
Bennett, A. F. & Ruben, J. A. Endothermy and activity in vertebrates. Science 206, 649–654 (1979).
Spoor, F. The semicircular canal system and locomotor behaviour, with special reference to hominin evolution. Cour. Forschungsinst. Senckenberg 243, 93–104 (2003).
Kemp, A. D. & Christopher Kirk, E. Eye size and visual acuity influence vestibular anatomy in mammals. Anat. Rec. 297, 781–790 (2014).
Money, K. E. et al. Physical properties of fluids and structures of vestibular apparatus of the pigeon. Am. J. Physiol. 220, 140–147 (1971).
Rowe, T. Definition, diagnosis, and origin of Mammalia. J. Vertebr. Palaeontol. 8, 241–264 (1988).
Nicol, S. C. Energy homeostasis in monotremes. Front. Neurosci. 11, 195 (2017).
Spoor, F. & Thewissen, J. G. M. in Senses on the Threshold: Adaptations in Secondarily Aquatic Vertebrates (eds Thewissen, J. G. M. & Nummela, S.). 257–286 (Univ. California Press, 2008).
Opiang, M. D. Home ranges, movement, and den use in long-beaked echidnas, Zaglossus bartoni, from Papua New Guinea. J. Mammal. 90, 340–346 (2009).
Viglietti, P. A. et al. Evidence from South Africa for a protracted end-Permian extinction on land. Proc. Nat. Acad. Sci. USA 118, e2017045118 (2021).
Jones, K. E. et al. Regionalization of the axial skeleton predates functional adaptation in the forerunners of mammals. Nature Ecol. Evol. 4, 470–478 (2020).
Lautenschlager, S. et al. The role of miniaturization in the evolution of the mammalian jaw and middle ear. Nature 561, 533–537 (2018).
Donnelly, H. T. Oxygen consumption, activity and body fat in normal and hairless mice. Lab. Animals 16, 167–171 (1982).
Bal, N. C. & Periasamy, M. Uncoupling of sarcoendoplasmic reticulum calcium ATPase pump activity by sarcolipin as the basis for muscle non-shivering thermogenesis. Phil. Trans. R. Soc. B 375, 20190135 (2020).
Rowland, L. A., Bal, N. C. & Periasamy, M. The role of skeletal‐muscle‐based thermogenic mechanisms in vertebrate endothermy. Biol. Rev. 90, 1279–1297 (2015).
Benson, R. B., Butler, R., Close, R. A., Saupe, E. & Rabosky, D. L. Biodiversity across space and time in the fossil record. Curr. Biol. 31, R1225–R1236 (2021).
Close, R. A. et al. Diversity dynamics of Phanerozoic terrestrial tetrapods at the local-community scale. Nat. Ecol. & Evol. 3, 590–597 (2019).
Fernandez, V. et al. Synchrotron reveals Early Triassic odd couple: injured amphibian and aestivating therapsid share burrow. PLoS ONE 8, e64978 (2013).
Evers, S. W. et al. Neurovascular anatomy of the protostegid turtle Rhinochelys pulchriceps and comparisons of membranous and endosseous labyrinth shape in an extant turtle. Zool. J. Linn. Soc. 187, 800–828 (2019).
Mason, M. J. et al. The frog inner ear: picture perfect? J. Assoc. Res. Otolaryngol. 16, 171–188 (2015).
Schulz-Mirbach, T. et al. A unique swim bladder-inner ear connection in a teleost fish revealed by a combined high-resolution microtomographic and three-dimensional histological study. BMC Biol. 11, 75 (2013).
Schulz-Mirbach, T., Heß, M. & Metscher, B. D. Sensory epithelia of the fish inner ear in 3D: studied with high-resolution contrast enhanced microCT. Front. Zool. 10, 63 (2013).
Schulz-Mirbach, T., Heß, M. & Plath, M. Inner ear morphology in the Atlantic molly Poecilia mexicana—first detailed microanatomical study of the inner ear of a cyprinodontiform species. PLoS ONE 6, e27734 (2011).
Ghanem, T. A., Rabbitt, R. D. & Tresco, P. A. Three-dimensional reconstruction of the membranous vestibular labyrinth in the toadfish Opsanus tau. Hearing Res. 124, 27–43 (1998).
Buran, B. N., Deng, X. & Popper, A. N. Structural variation in the inner ears of four deep‐sea elopomorph fishes. J. Morph. 265, 215–225 (2005).
Deng, X., Wagner, H. J. & Popper, A. N. The inner ear and its coupling to the swim bladder in the deep-sea fish Antimora rostrata (Teleostei: Moridae). Deep Sea Res. 58, 27–37 (2011).
Deng, X., Wagner, H. J. & Popper, A. N. Interspecific variations of inner ear structure in the deep‐sea fish family Melamphaidae. Anat. Rec. 296, 1064–1082 (2013).
Evangelista, C. et al. A comparison of the external morphology of the membranous inner ear in elasmobranchs. J. Morphol. 271, 483–495 (2010).
Gauldie, R. W., Mulligan, K. & Thompson, R. K. The otoliths of a chimaera, the New Zealand elephant fish Callorhynchus milii. N. Zeal. J. Mar. Freshw. Res. 21, 275–280 (1987).
Gray, A. A. The Labyrinth of Animals: Including Mammals, Birds, Reptiles and Amphibians, Vol. I (Churchill, 1907).
Gray, A. A. The Labyrinth of Animals: Including Mammals, Birds, Reptiles and Amphibians, Vol. II (Churchill, 1908).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
XLSTAT statistical and data analysis solution (Addinsoft, 2021).
R Core Team. R: a language and environment for statistical computing (R Foundation for Statistical Computing, 2017).
Bjørn-Helge, M., Wehrens, R. & Liland, K. H. pls: partial least squares and principal component regression. R package version 2.7-3 (2020).
Maddison, W. P. & Maddison, D. R. Mesquite: a modular system for evolutionary analysis, Version 3.61 (2018).
Kumar, S. et al. TimeTree: a resource for timelines, timetrees, and divergence times. Mol. Biol. Evol. 34, 1812–1819 (2017).
Prum, R. O. et al. A comprehensive phylogeny of birds (Aves) using targeted next-generation DNA sequencing. Nature 526, 569–573 (2015).
Suh, A. et al. Ancient horizontal transfers of retrotransposons between birds and ancestors of human pathogenic nematodes. Nat. Commun. 7, 11396 (2016).
Naylor, G. J. P. et al. in Biology of Sharks and Their Relatives 2 (eds. Carrier, J. C., Musick, J. A. & Heithaus, M. R.) 31–56 (2012).
Bakke, I. & Steinar, J. Characterization of mitochondrial ribosomal RNA genes in gadiformes: sequence variations, secondary structural features, and phylogenetic implications. Mol. Phylogenetics Evol. 25, 87–100 (2002).
Bakke, I. & Steinar, D. J. Molecular phylogenetics of Gadidae and related Gadiformes based on mitochondrial DNA sequences. Mar. Biotech. 7, 61–69 (2005).
Parham, J. F. et al. Best practices for justifying fossil calibrations. Syst. Biol. 61, 346–359 (2012).
Rüsch, A. & Thurm, U. Cupula displacement, hair bundle deflection, and physiological responses in the transparent semicircular canal of young eel. Pflügers Archiv. 413, 533–545 (1989).
Steer, R. W. The Influence of Angular and Linear Acceleration and Thermal Stimulation on the Human Semicircular Canal. Sc.D. thesis, Massachusetts Institute of Technology (1967).
Rauch, S. Biochemie das Hörorgans (Thieme Verlag, 1964).
Ten Doesschate, G. De eigenschappen van de endolymph van Beenvissche. Sc. D. thesis, Utrecht University (1914).
Money, K. E., Sokoloff, M. & Weaver, R. S. Specific gravity and viscosity of endolymph and perilymph. In NASA, Second Symposium on the Role of the Vestibular Organs 91–98 (NASA, 1966).
Schnieder, E. A. & Schindler, K. in Biochemie des Hörorgans (ed Rauch, S.) 164–168 (Georg Thieme Verlag, 1964).
Macdonald, J. A. & Wells, R. M. G. in Biology of Antarctic fish (eds di Prisco, G., Maresca, B. & Tota B.) 163–178. (Springer, 1991).
Revell, L. J. phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Orme, D. et al. caper: comparative analyses of phylogenetics and evolution in R. R package version 1.0.1 (2018).
Pennell, M. W. et al. geiger v2.0: an expanded suite of methods for fitting macroevolutionary models to phylogenetic trees. Bioinformatics 30, 2216–2218 (2014).
Schliep, K. P. phangorn: phylogenetic analysis in R. Bioinformatics 27, 592–593 (2011).
Papadakis, M. et al. Rfast: a collection of efficient and extremely fast R functions. R package version 2.0.1 (2020).
Wilkinson, S. P. & Davy, S. K. phylogram: an R package for phylogenetic analysis with nested lists. J. Open Source Softw. 3, 790 (2018).
Paradis, E. & Schliep, K. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35, 526–528 (2019).
Ben-Shachar, M., Lüdecke, D. & Makowski, D. effectsize: estimation of effect size indices and standardized parameters. J. Open Source Softw. 5, 2815 (2020).
Komsta, L. outliers: tests for outliers. R package version 0.14 (2011).
Puttick M. et al. motmot: models of trait macroevolution on trees. R package version 2.1.3 (2019).
Wickham H., François R., Henry L. & Müller K. dplyr: a grammar of data manipulation. R package version 0.7.6 (2018).
Xiang, Y., Yakushin, S. B., Kunin, M., Raphan, T. & Cohen, B. Head stabilization by vestibulocollic reflexes during quadrupedal locomotion in monkey. J. Neurophysiol. 100, 763–780 (2008).
Boucot A. J., Chen, X., Scotese, C. R. & Morley, R. J. in SEPM Concepts in Sedimentology and Paleontology, Vol. 11 (eds Nichols, G. J. & Ricketts, B.) 1–478 (SEPM, 2013).
Rees, P. M. et al. Permian phytogeographic patterns and climate data/model comparisons. J. Geol. https://doi.org/10.1086/324203 (2002).
Rey, K. et al. Global climate perturbations during the Permo–Triassic mass extinctions recorded by continental tetrapods from South Africa. Gondwana Res. 37, 384–396 (2016).
Roscher, M., Stordal, F. & Svensen, H. The effect of global warming and global cooling on the distribution of the latest Permian climate zones. Palaeogeogr. Palaeoclimatol. Palaeoecol. 309, 186–200 (2011).
Gibbs, M. T. et al. Simulations of Permian climate and comparisons with climate-sensitive sediments. J. Geol. 110, 33–55 (2002).
Harris, R. et al. Climate change during the Triassic and Jurassic. Geol. Today 33, 210–215 (2017).
Sellwood, B. W. & Valdes, P. J. Mesozoic climates: general circulation models and the rock record. Sediment. Geol. 190, 269–287 (2006).
Acknowledgements
Funding for this project was provided by the Fundação para a Ciência e a Tecnologia postdoctoral fellowship SFRH/BPD/96205/2013, FCT–AGA KHAN Development Network grant number 333206718, National Geographic Society grant number CP-109R-17, the Field Museum, NSF EAR-1337291, the Max Planck Society and the Calleva Foundation. We acknowledge intellectual contributions from discussions with R. Rabbitt. We thank J. White, S. Walsh, P. Campbell, S. Pierce, C. Capobianco, S. Chapman, J. D. Cundiff, A. Wynn, P. Gill, E. Rayfield, J. Hopson, R. Asher, A. Neander, W. Simpson, A. Stroup, A. Resetar, J. Mata, J.-J. Hublin, D. Plotzki, H. Temming, W. van Gestel, J. Jansen, R. Allain, D. Silvestro, F. Condamine, C. Scotese, R. Mundry, S. W. Evers, M. J. Mason, P.-O. Antoine, S. Hellert, C. Schultz, M. B. Soares and A. Schmitt. We also thank the Institute of Veterinary Pathology and the Veterinary Clinic for Birds and Reptiles at Leipzig University, the Leibniz Institute for Zoo and Wildlife Research and the German Primate Center at Göttingen. We acknowledge the MRI platform member of the national infrastructure France-BioImaging supported by the French National Research Agency (ANR-10-INBS-04, «Investments for the future»), the labex CEMEB (ANR-10-LABX-0004) and NUMEV (ANR-10-LABX-0020). We acknowledge the European Synchrotron Radiation Facility for provision of synchrotron radiation facilities and we would like to thank V. Fernandez and P. Tafforeau for assistance in using beamline BM05 and ID17. IPFN activities received financial support from through projects UIDB/50010/2020 and UIDP/50010/2020. Some silhouettes were obtained from Phylopic.org.
Author information
Authors and Affiliations
Contributions
R.A., R.D. and K.D.A. designed the study and interpreted the data. R.A. and R.D. collected and analysed the data and wrote the manuscript, with specific input by F.S., K.D.A. and P.M.B. R.D. developed the thermo-motility index and wrote the code. K.D.A. and R.A. reviewed time divergence dates and last occurrence data. R.D., F.S. and A.S. collected specimens for membranous labyrinth analysis. All authors contributed raw and/or processed data, discussed the results, and commented on the draft manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Michael Benton and the other, anonymous, reviewers for their contribution to the peer review of this work. Peer review reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Selected computed-tomography sections of inner ears of non-mammalian synapsids.
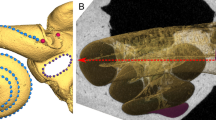
a-e, Anterior (ASC), posterior (PSC), and lateral (LSC) semicircular canals, vestibule (Ve), and crus communis (CC). Scale bar is 5 mm. a, horizontal section of the dicynodont Kawingasaurus (GPIT/RE/9272). b, coronal section of the dicynodont Aulacephalodon (NHCC LB335). c, horizontal section of the probainognathian Chiniquodon (MCZ 3778). d, coronal section of the mammaliamorph Pseudotherium (PVSJ 882). e, horizontal section of the therocephalian Mupashi (NHCC LB44).
Extended Data Fig. 2 Examples of measurements used in this study.
a-i, Views of the bony labyrinth (grey) and the membranous semicircular duct system (red) of an alpaca (a-c, CEB 130038), a domestic turkey (d-f, CEB 130069), and a false gharial (g-i, CEB140070), in the plane of the anterior (a, d, g), posterior (b, e, h), and lateral (c, f, i) semicircular canals. DM a, DM p, and DM l, major axes of the anterior, posterior, and lateral canal tori; Dm a, Dm p, and Dm l, minor axes of the anterior, posterior, and lateral canal tori; dS a, dS p, and dS l, cross-sectional thicknesses of the slender portion of the anterior, posterior, and lateral canals; LS a, LS p, and LS l, lengths of the slender portion of the anterior, posterior and lateral canals; Sa, Sp, and Sl, slender portions of the anterior, posterior, and lateral semicircular ducts; Aa, Ap, and Al, anterior, posterior, and lateral ampullae; Ua, anterior utriculus; CC, common crus; Ve, vestibule.
Extended Data Fig. 3 Distribution of morphological parameters measured on the anterior semicircular canals of tetrapods.
a-d, Boxplots of the radius of curvature relative to body mass (a), average cross-sectional thickness (b), and length (c) of the slender portion relative to the radius of curvature, and eccentricity of the semicircular canal tori (d). Arrows represent the direction of adaptations theoretically expected for endotherms. Boxplot centre, median; box boundaries, first and third quartiles; whiskers, 1.5 × IQR from boundaries. b, The outlier Caecilia has been omitted to standardize the plot area.
Extended Data Fig. 4 Phylogenetic distribution of uncontrolled parameters of the TMI.
a, Optimization of the residual variation of bony/membranous correlations on a time calibrated tree of extant tetrapods. Branch colours reflect the value of the residual variation according to the colour scale. b, Scatterplot of endolymph viscosity against Tb showing all data available (see Methods). Mammals are in blue, euteleosteans in green and birds in red. Dashed curves represent endolymph physicochemical properties that are water-like (large dashes), near water-like (medium dashes) or relatively viscous (small dashes). Animal silhouettes were either created by Ricardo Araújo (guinea pig, whiting, haddock, bald notothen) or are available at phylopic (http://phylopic.org/) under Public Domain license.
Extended Data Fig. 5 Probability distributions of body temperatures of fossil synapsids predicted from the TMI.
a-c, Note the elevated Tb of non-mammalian mammaliamorphs (NMM) and relatively low Tb of non-mammaliamorph probainognathians (NMP, b-c). Overlaps between distributions should be interpreted carefully because predicted temperatures are phylogenetically-dependent (see Table 1). Vertical dashed line represents the lowest Tb observed in extant endotherms (31 °C). Predicted Tb for endotherm and ectotherm (a), phylogenetic clusters best summarizing the data (b), and major groups (c) of fossil synapsids. c, Note that predicted Tb of non-mammaliamorph synapsids conform to a stochastic process.
Extended Data Fig. 6 Phylogenetic distribution of the TMI in fossil tetrapods.
Optimization of the TMI on a time-calibrated tree of major tetrapod clades and all of the extinct tetrapods analysed in this study. Branch colours reflect the likelihood of being endothermic according to the probability colour scale.
Extended Data Fig. 7 Relationship between thermo-motility indices of the lateral and anterior canals.
The solid line represents the PGLS regression for synapsids. The lateral canal best reflects behavioural agility and its TMI, calculated from the saturating velocity, is the least related to Tb (Supplementary Note 2). Note that non-mammalian synapsids mostly plot above extant ectotherms, suggesting increased locomotor activity for similar Tb. Additionally, while non-mammalian mammaliamorphs plot with endotherms for the anterior canal TMI (the main TMI used in this study), they are intermediate between endotherms and ectotherms for the lateral canal TMI, suggesting their locomotor activity was intermediate between basal synapsids and mammals. Animal silhouettes were either created by Ricardo Araújo (Oligokyphus, Cynognathia) or are available at phylopic (http://phylopic.org/) under Public Domain license.
Supplementary information
Supplementary Information
Supplementary Methods, including: sampling rationale; biomechanics of the thermo-motility index; discussion on the endolymph viscosity in vertebrates and a step-by-step summary of the analyses. Supplementary Notes 1–3, including: definitions; description of the statistical analyses; Dimetrodon photogrammetry and divergence times and last occurrence datum.
Supplementary Data 1
Data on stride frequency, maximum aerobic and anaerobic speeds with body temperature and body mass for a vertebrate sample.
Supplementary Data 2
Measurements; body temperature; body size variables; thermo-motility indices; details on PLS regressions.
Supplementary Data 3
Scan data and specimen provenience.
Supplementary Data 4
Data on the age, palaeolatitude and palaeoclimate for sites of non-mammaliamorph synapsids; with predicted body temperature and interpretations of thermoregulatory strategies; data on climate of today’s locations.
Supplementary Data 5
Reproducibility and repeatability tests.
Supplementary Data 6
Phylogenetic tree of all specimens used in the study.
Supplementary Dataset
R custom code and associated datasets.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Araújo, R., David, R., Benoit, J. et al. Inner ear biomechanics reveals a Late Triassic origin for mammalian endothermy. Nature 607, 726–731 (2022). https://doi.org/10.1038/s41586-022-04963-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-04963-z
This article is cited by
-
First evidence of the link between internal and external structure of the human inner ear otolith system using 3D morphometric modeling
Scientific Reports (2023)
-
Mammalian maxilloturbinal evolution does not reflect thermal biology
Nature Communications (2023)
-
Derived faunivores are the forerunners of major synapsid radiations
Nature Ecology & Evolution (2023)
-
A complete skull of a stem mammal from the Late Triassic of Brazil illuminates the early evolution of prozostrodontian cynodonts
Journal of Mammalian Evolution (2023)
-
Revisiting life history and morphological proxies for early mammaliaform metabolic rates
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.