Abstract
NPR1 is a master regulator of the defence transcriptome induced by the plant immune signal salicylic acid1,2,3,4. Despite the important role of NPR1 in plant immunity5,6,7, understanding of its regulatory mechanisms has been hindered by a lack of structural information. Here we report cryo-electron microscopy and crystal structures of Arabidopsis NPR1 and its complex with the transcription factor TGA3. Cryo-electron microscopy analysis reveals that NPR1 is a bird-shaped homodimer comprising a central Broad-complex, Tramtrack and Bric-à-brac (BTB) domain, a BTB and carboxyterminal Kelch helix bundle, four ankyrin repeats and a disordered salicylic-acid-binding domain. Crystal structure analysis reveals a unique zinc-finger motif in BTB for interacting with ankyrin repeats and mediating NPR1 oligomerization. We found that, after stimulation, salicylic-acid-induced folding and docking of the salicylic-acid-binding domain onto ankyrin repeats is required for the transcriptional cofactor activity of NPR1, providing a structural explanation for a direct role of salicylic acid in regulating NPR1-dependent gene expression. Moreover, our structure of the TGA32–NPR12–TGA32 complex, DNA-binding assay and genetic data show that dimeric NPR1 activates transcription by bridging two fatty-acid-bound TGA3 dimers to form an enhanceosome. The stepwise assembly of the NPR1–TGA complex suggests possible hetero-oligomeric complex formation with other transcription factors, revealing how NPR1 reprograms the defence transcriptome.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information. The cryo-EM structures of the apo NPR1, NPR12–TGA32 and TGA32–NPR12–TGA32, and X-ray crystal structures of the NPR1(ΔSBD) and TGA3 NID have been deposited at the PDB (www.pdb.org) under accession codes 7MK2, 7TAD, 7TAC, 7MK3 and 7TAE, respectively. The cryo-EM density maps of apo NPR1, NPR1–SA, NPR12–TGA32 and TGA32–NPR12–TGA32 have been deposited to the Electron Microscopy Data Bank under accession codes EMD-23884, EMD-23885, EMD-25771 and EMD-25769, respectively. There are no restrictions on data availability. Source data are provided with this paper.
References
Delaney, T. P., Friedrich, L. & Ryals, J. A. Arabidopsis signal transduction mutant defective in chemically and biologically induced disease resistance. Proc. Natl Acad. Sci. USA 92, 6602–6606 (1995).
Shah, J., Tsui, F. & Klessig, D. F. Characterization of a salicylic acid-insensitive mutant (sai1) of Arabidopsis thaliana, identified in a selective screen utilizing the SA-inducible expression of the tms2 gene. Mol. Plant Microbe Interact. 10, 69–78 (1997).
Cao, H., Bowling, S. A., Gordon, A. S. & Dong, X. Characterization of an Arabidopsis mutant that is nonresponsive to inducers of systemic acquired resistance. Plant Cell 6, 1583–1592 (1994).
Glazebrook, J., Rogers, E. E. & Ausubel, F. M. Isolation of Arabidopsis mutants with enhanced disease susceptibility by direct screening. Genetics 143, 973–982 (1996).
Ryals, J. et al. The Arabidopsis NIM1 protein shows homology to the mammalian transcription factor inhibitor I kappa B. Plant Cell 9, 425–439 (1997).
Cao, H., Glazebrook, J., Clarke, J. D., Volko, S. & Dong, X. The Arabidopsis NPR1 gene that controls systemic acquired resistance encodes a novel protein containing ankyrin repeats. Cell 88, 57–63 (1997).
Backer, R., Naidoo, S. & van den Berg, N. The NONEXPRESSOR OF PATHOGENESIS-RELATED GENES 1 (NPR1) and related family: mechanistic insights in plant disease resistance. Front. Plant Sci. 10, 102 (2019).
Silva, K. J. P., Mahna, N., Mou, Z. & Folta, K. M. NPR1 as a transgenic crop protection strategy in horticultural species. Hortic. Res. 5, 15 (2018).
Stogios, P. J., Downs, G. S., Jauhal, J. J., Nandra, S. K. & Prive, G. G. Sequence and structural analysis of BTB domain proteins. Genome Biol. 6, R82 (2005).
Stogios, P. J. & Prive, G. G. The BACK domain in BTB-kelch proteins. Trends Biochem. Sci. 29, 634–637 (2004).
Canning, P. et al. Structural basis for Cul3 protein assembly with the BTB-Kelch family of E3 ubiquitin ligases. J. Biol. Chem. 288, 7803–7814 (2013).
Zhuang, M. et al. Structures of SPOP-substrate complexes: insights into molecular architectures of BTB-Cul3 ubiquitin ligases. Mol. Cell 36, 39–50 (2009).
Errington, W. J. et al. Adaptor protein self-assembly drives the control of a cullin-RING ubiquitin ligase. Structure 20, 1141–1153 (2012).
Gorina, S. & Pavletich, N. P. Structure of the p53 tumor suppressor bound to the ankyrin and SH3 domains of 53BP2. Science 274, 1001–1005 (1996).
Li, J., Mahajan, A. & Tsai, M. D. Ankyrin repeat: a unique motif mediating protein-protein interactions. Biochemistry 45, 15168–15178 (2006).
Sedgwick, S. G. & Smerdon, S. J. The ankyrin repeat: a diversity of interactions on a common structural framework. Trends Biochem. Sci. 24, 311–316 (1999).
Wang, W. et al. Structural basis of salicylic acid perception by Arabidopsis NPR proteins. Nature 586, 311–316 (2020).
Mou, Z., Fan, W. & Dong, X. Inducers of plant systemic acquired resistance regulate NPR1 function through redox changes. Cell 113, 935–944 (2003).
Canet, J. V., Dobon, A., Roig, A. & Tornero, P. Structure-function analysis of npr1 alleles in Arabidopsis reveals a role for its paralogs in the perception of salicylic acid. Plant Cell Environ. 33, 1911–1922 (2010).
Bombarda, E., Cherradi, H., Morellet, N., Roques, B. P. & Mely, Y. Zn2+ binding properties of single-point mutants of the C-terminal zinc finger of the HIV-1 nucleocapsid protein: evidence of a critical role of cysteine 49 in Zn2+ dissociation. Biochemistry 41, 4312–4320 (2002).
Hol, W. G. Effects of the alpha-helix dipole upon the functioning and structure of proteins and peptides. Adv. Biophys. 19, 133–165 (1985).
Tada, Y. et al. Plant immunity requires conformational changes of NPR1 via S-nitrosylation and thioredoxins. Science 321, 952–956 (2008).
Manohar, M. et al. Identification of multiple salicylic acid-binding proteins using two high throughput screens. Front. Plant Sci. 5, 777 (2014).
Ding, Y. et al. Opposite roles of salicylic acid receptors NPR1 and NPR3/NPR4 in transcriptional regulation of plant immunity. Cell 173, 1454–1467 (2018).
Waterhouse, A. et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Saleh, A. et al. Posttranslational modifications of the master transcriptional regulator NPR1 enable dynamic but tight control of plant immune responses. Cell Host Microbe 18, 169–182 (2015).
Maier, F. et al. NONEXPRESSOR OF PATHOGENESIS-RELATED PROTEINS1 (NPR1) and some NPR1-related proteins are sensitive to salicylic acid. Mol. Plant Pathol. 12, 73–91 (2011).
Hermann, M. et al. The Arabidopsis NIMIN proteins affect NPR1 differentially. Front. Plant Sci. 4, 88 (2013).
Lebel, E. et al. Functional analysis of regulatory sequences controlling PR-1 gene expression in Arabidopsis. Plant J. 16, 223–233 (1998).
Kachroo, A. & Kachroo, P. Fatty acid-derived signals in plant defense. Annu. Rev. Phytopathol. 47, 153–176 (2009).
Fu, Z. Q. & Dong, X. Systemic acquired resistance: turning local infection into global defense. Annu. Rev. Plant Biol. 64, 839–863 (2013).
Wang, D., Amornsiripanitch, N. & Dong, X. A genomic approach to identify regulatory nodes in the transcriptional network of systemic acquired resistance in plants. PLoS Pathog. 2, e123 (2006).
Pape, S., Thurow, C. & Gatz, C. The Arabidopsis PR-1 promoter contains multiple integration sites for the coactivator NPR1 and the repressor SNI1. Plant Physiol. 154, 1805–1818 (2010).
Sievers, F. et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 7, 539 (2011).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Scheres, S. H. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Afonine, P. V. et al. Real-space refinement in PHENIX for cryo-EM and crystallography. Acta Crystallogr. D 74, 531–544 (2018).
Bepler, T. et al. Positive-unlabeled convolutional neural networks for particle picking in cryo-electron micrographs. Nat. Methods 16, 1153–1160 (2019).
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Li, C., Tan, B. K., Zhao, J. & Guan, Z. In vivo and in vitro synthesis of phosphatidylglycerol by an Escherichia coli cardiolipin synthase. J. Biol. Chem. 291, 25144–25153 (2016).
Spoel, S. H. et al. Proteasome-mediated turnover of the transcription coactivator NPR1 plays dual roles in regulating plant immunity. Cell 137, 860–872 (2009).
Zavaliev, R., Mohan, R., Chen, T. & Dong, X. Formation of NPR1 condensates promotes cell survival during the plant immune response. Cell 182, 1093–1108 (2020).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Zavaliev, R. & Epel, B. L. Imaging callose at plasmodesmata using aniline blue: quantitative confocal microscopy. Methods Mol. Biol. 1217, 105–119 (2015).
Acknowledgements
We thank Y. Tada for discussion on the project and Z. Yu, X. Zhao, S. Yang and R. Yan for support in cryo-EM data collection at the Janelia Research Campus. This work was supported by grants from the National Institutes of Health R01 GM115355 to P.Z., R35 GM118036 to X.D., R01 GM141223 to A.B., R01 AI148366 to Z.G., the Duke University School of Medicine Bridge Fund to P.Z., the Howard Hughes Medical Institute to X.D. and the Intramural Research Program of the National Institute of Environmental Health Sciences (NIEHS) grant ZIC ES103326 to M.J.B. X-ray fluorescence and diffraction data were collected at the Northeastern Collaborative Access Team beamline 24-ID-C, which is funded by the National Institute of General Medical Sciences (grant P30 GM124165). The Pilatus 6M detector on the 24-ID-C beamline is funded by the NIH Office of Research Infrastructure Programs (Hight-End Instrumentation grant S10 RR029205). The SAXS measurements were performed at Beamline 12-ID-B. This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by the Argonne National Laboratory under contract DE-AC02-06CH11357. Cryo-EM data were collected using the Thermo Fisher Scientific Titan Krios transmission electron microscopes at the Janelia Research Campus and Duke University Shared Materials Instrumentation Facility (SMIF), and the Thermo Fisher Scientific Talos Arctica transmission electron microscopes at NIEHS and the University of North Carolina at Chapel Hill (UNC).
Author information
Authors and Affiliations
Contributions
S.K., R.Z., Q.W., J.W., X.D. and P.Z. conceived the project. S.K., Q.W., Y.Z., L.D., J.C., M.J.B., A.B. and P.Z. conducted cryo-EM sample preparation, data collection and processing, and model building. S.K. performed the SAXS study. R.Z. performed in vivo bioassays, including the functional analyses. J.C., Q.W. and P.Z. performed crystallographic studies and data analysis. Q.W. and J.C. performed in vitro biochemical assays. J.Z. and Z.G. performed the MS analysis. J.P. performed the promoter analysis for SA-induced genes. S.K., R.Z., X.D. and P.Z. wrote the manuscript with input from all of the authors.
Corresponding authors
Ethics declarations
Competing interests
X.D. is a cofounder of Upstream Biotechnology and a scientific advisory board member of Inari Agriculture. The other authors declare no competing interests.
Peer review
Peer review information
Nature thanks Aardra Kachroo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
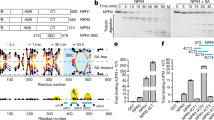
Extended Data Fig. 1 Biochemical characterization and cryo-EM reconstruction of apo NPR1.
a, SDS–PAGE gel of purified NPR1. b, Size-exclusion chromatography shows that NPR1 elutes between molecular weight markers of 440 kDa and 158 kDa on a Superose 6 Increase 10/300 GL column. c, Crosslinking of NPR1 by BS3 at varying concentrations reveals a dominant dimer band. d, Flowchart of the reconstruction. Details are described in Methods. Green circles highlight representative NPR1 particles on the micrograph with the enlarged view in the inset. e, The global Fourier Shell Correlation (FSC) curve. f, Local resolution map. g, Euler angle distribution of the particles. h, Representative regions of the EM density map. Images in a and c are representative of 3 and 2 biological replicates, respectively.
Extended Data Fig. 2 Biochemical characterization of the NPR1 BHB and SBD domains.
a, Interaction of Myc-tagged CUL3A (Myc-CUL3) with free GST, or GST-fused WT NPR1 (GST–NPR1), or NPR1ΔBHB deletion mutant (deletion of E200-L258; GST–NPR1ΔBHB) in E. coli. The proteins were co-expressed in E. coli and total lysate was used for pull-down with glutathione affinity resin. Images are representative of 2 biological replicates. b, Solution small angle X-ray scattering (SAXS) data for the Arabidopsis apo NPR1 SBD purified from insect cells at a concentration of 1 mg ml−1. The SAXS scattering curve, the Kratky plot, and the Guinier analysis are shown in the top, middle, and bottom panels, respectively. The non-bell shape of the Kratky plot indicates the protein is unfolded and exhibits a random-coil behaviour. The Guinier analysis yields a large radius of gyration (Rg) value of 47 Å for the NPR1 SBD (~20 kDa) in comparison with folded lysozyme (14.3 kDa; Rg = ~16 Å) and Bovine serum albumin (66 kDa; Rg = ~30 Å), indicating a disordered conformation.
Extended Data Fig. 3 NPR1 harbours a unique zinc finger.
a, X-ray fluorescence scanning data revealed the presence of Zn2+ in NPR1(ΔSBD) crystals. Scanning results for the NPR1 protein crystal and buffers are shown in the left and right panels, respectively. b, Sequence alignment of BTB domains. Conserved cysteine and histidine residues in a unique cysteine cluster preserved in NPR proteins are highlighted in pink. Dots indicate residues participating in zinc coordination, and triangles denote residues mutated in npr1(dim). Listed plant species include: Arabidopsis thaliana (At), Brassica rapa (Br), Brassica juncea (Bj), Brassica napus (Bn),Raphanus sativus (Rs), Oryza sativa (Os), Nicotiana tabacum (Nt), Populus trichocarpa (Pt), Zea mays (Zm), Solanum lycopersicum (Sl), Vitis vinifera (Vv), Hordeum vulgare (Hv), Medicago truncatula (Mt), and Glycine max (Gm).
Extended Data Fig. 4 Cryo-EM reconstruction of the NPR1-SA complex.
Details of the flowchart are described in Methods. a, Flowchart of the reconstruction. b, Global Fourier Shell Correlation (FSC) curve. c, Euler angle distribution of the particles.
Extended Data Fig. 5 Cryo-EM reconstruction of the NPR1–TGA3 complex.
a, Flowchart of the reconstruction. b, Local resolution, global Fourier Shell Correlation (FSC) curve, Euler angle distribution of the particles, and representative regions of the EM density map of the TGA32–NPR12–TGA32 complex. c, Local resolution, global FSC curve, Euler angle distribution of the particles, and representative regions of the EM density map of the NPR12–TGA32 complex.
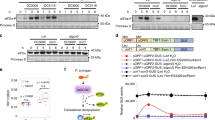
Extended Data Fig. 6 Crystallographic and mass spectrometry characterization of the TGA3 NID–palmitate complex.
a, Crystal structure of the TGA3 NID dimer. The two TGA3-NID molecules are shown in the rainbow colour, with N-terminus coloured in blue and C-terminus coloured in red. b, A zoomed-in view of the location of the palmitate. Polar interactions with the carboxylate group of the palmitates are indicated with dashed lines. For clarity, only one TGA3 molecule is coloured in rainbow, and the other molecule is coloured in grey. Purple meshes in panels a, b represent 2mFo-DFc omit map of the palmitate plotted at the 1.0 σ level. c, Mass spectrometry analysis of the fatty acid extracted from the protein sample, verifying the fatty acid as the palmitic acid (C16:0).
Extended Data Fig. 7 The as-1 elements in SA-induced gene promoters.
a, Distribution of as-1 element in the promoters (3 kb) of SA-uninduced genes compared to the promoters (3 kb) of top 100 SA-induced genes32. A statistically significant difference of as-1 element distribution was seen in the promoters of the top 100 SA-induced genes compared to the promoters of SA-uninduced genes (p-value < 0.001). b, Promoter analysis for as-1 elements of the top 100 SA-induced genes after 8 h treatment. c, Frequency plot of distances between as-1 elements from the promoters of the top 100 SA-induced genes. d, Electrophoresis mobility shift assay of NPR1 and TGA3 using 6-fluorescein-labelled DNA spanning the LS5-to-LS7 region of the PR1 promoter containing two as-1 elements (LS5/LS7) or a single as-1 element in the LS5 region (LS5/LS7-) or in the LS7 region (LS5-/LS7). d is a representative image of 3 biological replicates.
Extended Data Fig. 8 Characterization of the npr1(dim) mutant.
a, Elution profiles of the WT NPR1, npr1(C82A), and npr1(dim) samples on a Superose 6 Increase 10/300 GL column. b, A low resolution cryo-EM map of npr1(dim) reveals the expected shape of a monomeric NPR1. The monomeric model of NPR1 has been fitted into the EM density and displayed in the cartoon representation.
Supplementary information
Supplementary Information
Supplementary Figs 1 and 2 (the uncropped gels) and Supplementary Table 1, which contains a list of the DNA primers that were used in this study.
Rights and permissions
About this article
Cite this article
Kumar, S., Zavaliev, R., Wu, Q. et al. Structural basis of NPR1 in activating plant immunity. Nature 605, 561–566 (2022). https://doi.org/10.1038/s41586-022-04699-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-04699-w
This article is cited by
-
Feedback regulation of salicylic acid signaling by isochorismate synthase1 modulates camptothecin biosynthesis in Camptotheca acuminata
Plant Cell, Tissue and Organ Culture (PCTOC) (2024)
-
Disruption of the Novel Small Protein RBR7 Leads to Enhanced Plant Resistance to Blast Disease
Rice (2023)
-
Different viral effectors suppress hormone-mediated antiviral immunity of rice coordinated by OsNPR1
Nature Communications (2023)
-
A plant RNA virus inhibits NPR1 sumoylation and subverts NPR1-mediated plant immunity
Nature Communications (2023)
-
Plant Immunity: A Plastic System Operated Through Cell-Fate Transition
Journal of Plant Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.