Abstract
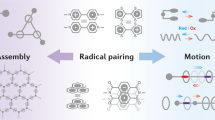
Molecular recognition1,2,3,4 and supramolecular assembly5,6,7,8 cover a broad spectrum9,10,11 of non-covalently orchestrated phenomena between molecules. Catalysis12 of such processes, however, unlike that for the formation of covalent bonds, is limited to approaches13,14,15,16 that rely on sophisticated catalyst design. Here we establish a simple and versatile strategy to facilitate molecular recognition by extending electron catalysis17, which is widely applied18,19,20,21 in synthetic covalent chemistry, into the realm of supramolecular non-covalent chemistry. As a proof of principle, we show that the formation of a trisradical complex22 between a macrocyclic host and a dumbbell-shaped guest—a molecular recognition process that is kinetically forbidden under ambient conditions—can be accelerated substantially on the addition of catalytic amounts of a chemical electron source. It is, therefore, electrochemically possible to control23 the molecular recognition temporally and produce a nearly arbitrary molar ratio between the substrates and complexes ranging between zero and the equilibrium value. Such kinetically stable supramolecular systems24 are difficult to obtain precisely by other means. The use of the electron as a catalyst in molecular recognition will inspire chemists and biologists to explore strategies that can be used to fine-tune non-covalent events, control assembly at different length scales25,26,27 and ultimately create new forms of complex matter28,29,30.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information files.
References
Lehn, J.-M. Supramolecular chemistry—Scope and perspectives. Molecules, supermolecules, and molecular devices (Nobel lecture). Angew. Chem. Int. Ed. Engl. 27, 89–112 (1988).
Cram, D. J. The design of molecular hosts, guests, and their complexes (Nobel lecture). Angew. Chem. Int. Ed. Engl. 27, 1009–1020 (1988).
Philp, D. & Stoddart, J. F. Self-assembly in natural and unnatural systems. Angew. Chem. Int. Ed. Engl. 35, 1154–1196 (1996).
Persch, E., Dumele, O. & Diederich, F. Molecular recognition in chemical and biological systems. Angew. Chem. Int. Ed. 54, 3290–3327 (2015).
Aida, T., Meijer, E. W. & Stupp, S. I. Functional supramolecular polymers. Science 335, 813–817 (2012).
Das, K., Gabrielli, L. & Prins, L. J. Chemically fueled self‐assembly in biology and chemistry. Angew. Chem. Int. Ed. 60, 20120–20143 (2021).
Weißenfels, M., Gemen, J. & Klajn, R. Dissipative self-assembly: fueling with chemicals versus light. Chem 7, 23–37 (2021).
Yin, Z. et al. Dissipative supramolecular polymerization powered by light. CCS Chem. 1, 335–342 (2019).
Wiester, M. J., Ulmann, P. A. & Mirkin, C. A. Enzyme mimics based upon supramolecular coordination chemistry. Angew. Chem. Int. Ed. 50, 114–137 (2010).
Webber, M. J., Appel, E. A., Meijer, E. W. & Langer, R. Supramolecular biomaterials. Nat. Mater. 15, 13–26 (2015).
Amabilino, D. B., Smith, D. K. & Steed, J. W. Supramolecular materials. Chem. Soc. Rev. 46, 2404–2420 (2017).
Wang, Y. et al. What molecular assembly can learn from catalytic chemistry. Chem. Soc. Rev. 43, 399–411 (2014).
Turberfield, A. J. et al. DNA fuel for free-running nanomachines. Phys. Rev. Lett. 90, 118102 (2003).
Zhang, D. Y., Turberfield, A. J., Yurke, B. & Winfree, E. Engineering entropy-driven reactions and networks catalyzed by DNA. Science 318, 1121–1125 (2007).
Song, T. & Liang, H. Synchronized assembly of gold nanoparticles driven by a dynamic DNA-fueled molecular machine. J. Am. Chem. Soc. 134, 10803–10806 (2012).
Li, H. et al. Proton-assisted self-assemblies of linear di-pyridyl polyaromatic molecules at solid/liquid interface. J. Phys. Chem. C 116, 21753–21761 (2012).
Studer, A. & Curran, D. P. The electron is a catalyst. Nat. Chem. 6, 765–773 (2014).
Francke, R. & Little, R. D. Electrons and holes as catalysts in organic electrosynthesis. ChemElectroChem 6, 4373–4382 (2019).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemical methods since 2000: on the verge of a renaissance. Chem. Rev. 117, 13230–13319 (2017).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Romero, N. A. & Nicewicz, D. A. Organic photoredox catalysis. Chem. Rev. 116, 10075–10166 (2016).
Trabolsi, A. et al. Radically enhanced molecular recognition. Nat. Chem. 2, 42–49 (2010).
Aubert, S., Bezagu, M., Spivey, A. C. & Arseniyadis, S. Spatial and temporal control of chemical processes. Nat. Rev. Chem. 3, 706–722 (2019).
Mattia, E. & Otto, S. Supramolecular systems chemistry. Nat. Nanotechnol. 10, 111–119 (2015).
Whitesides, G. M. Self-assembly at all scales. Science 295, 2418–2421 (2002).
Harada, A., Kobayashi, R., Takashima, Y., Hashidzume, A. & Yamaguchi, H. Macroscopic self-assembly through molecular recognition. Nat. Chem. 3, 34–37 (2010).
Santos, P. J., Gabrys, P. A., Zornberg, L. Z., Lee, M. S. & Macfarlane, R. J. Macroscopic materials assembled from nanoparticle superlattices. Nature 591, 586–591 (2021).
Lehn, J. M. Toward self-organization and complex matter. Science 295, 2400–2403 (2002).
Vantomme, G. & Meijer, E. W. The construction of supramolecular systems. Science 363, 1396–1397 (2019).
Pezzato, C., Cheng, C., Stoddart, J. F. & Astumian, R. D. Mastering the non-equilibrium assembly and operation of molecular machines. Chem. Soc. Rev. 46, 5491–5507 (2017).
Luca, O. R., Gustafson, J. L., Maddox, S. M., Fenwick, A. Q. & Smith, D. C. Catalysis by electrons and holes: formal potential scales and preparative organic electrochemistry. Org. Chem. Front. 2, 823–848 (2015).
Whitesides, G. M. et al. Noncovalent synthesis: using physical-organic chemistry to make aggregates. Acc. Chem. Res. 28, 37–44 (1995).
Reinhoudt, D. N. Synthesis beyond the molecule. Science 295, 2403–2407 (2002).
Frasconi, M. et al. Redox control of the binding modes of an organic receptor. J. Am. Chem. Soc. 137, 11057–11068 (2015).
Wang, Y. et al. Symbiotic control in mechanical bond formation. Angew. Chem. Int. Ed. 55, 12387–12392 (2016).
Cai, K. et al. Molecular Russian dolls. Nat. Commun. 9, 5275 (2018).
Acknowledgements
We thank Northwestern University (NU) for its continued support of this research and acknowledge the Integrated Molecular Structure Education and Research Center (IMSERC) at NU for providing access to equipment for relevant experiments. The computational investigations at California Institute of Technology were supported by National Science Foundation grant no. CBET-2005250 (W.-G.L. and W.A.G.). This work was also supported by the Department of Energy, Office of Science, Office of Basic Energy Sciences under Award DE-FG02-99ER14999 (M.R.W.) and the Natural Science Foundation of Anhui Province grant no. 2108085MB31 (D.S.).
Author information
Authors and Affiliations
Contributions
Y.J., Y.Q. and J.F.S. conceived the idea for this project. L.Z. proposed a key mechanistic conjecture. W.-G.L. and W.A.G. performed quantum mechanical calculations. Y.J. and Y.Q. synthesized and characterized the materials with the help of H.C., Y.F., K.C., D.S., B.S., X.-Y.C., X.L. and X.Z. H.M., R.M.Y. and M.R.W. performed the electron paramagnetic resonance characterizations and detailed analyses. C.L.S. performed the single-crystal X-ray diffraction. R.D.A. contributed to the theoretical analyses on the mechanism of electron catalysis. Y.J., Y.Q. and J.F.S. wrote the first and second drafts of the paper. J.F.S. and W.A.G. directed the project. All the authors participated in evaluating the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Y.J., Y.Q. and J.F.S. have filed a patent application lodged with Northwestern University (INVO reference no. NU 2021-248) based on this work.
Peer review
Peer review information
Nature thanks Robert Francke and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 The electron as an efficient catalyst for both covalent reactions and molecular recognition.
a, Electron-catalysed covalent reactions are well established in synthetic covalent chemistry, particularly for radical-mediated photoredox catalysis and organic electrosynthesis. Consider a reaction A + B → A–B, where a high energy barrier causes the reaction to be very slow. Injection of an electron reduces one of the substrates (A) to a highly reactive radical species (A•−), which can rapidly form a covalent bond with the other substrate (B). The resulting intermediate (A•−–B) releases the electron to afford the final product (A–B). Whereas the overall process is redox neutral, i.e., the redox state remains the same during the transformation from the substrates to product, the catalytic pathway, which involves the temporary addition of an electron, leads to a substantially lower energy barrier, thereby expediting the formation of the covalent bond. In this process, the electron has acted as an effective catalyst. b, The research reported in this article aims to extend the paradigm of electron catalysis to promoting and controlling molecular recognition. The trajectory for this noncovalent process is similar to that for electron-catalysed covalent reactions, except that the product is a supramolecular complex wherein molecular components are assembled courtesy of noncovalent bonding interaction(s), rather than a molecule whose atoms are connected by covalent bond(s).
Supplementary information
Supplementary Information
Supplementary Figs. 1–45, Schemes 1–9, Tables 1–8, supplementary text and notes, extensive experimental data, quantum mechanical calculation results and detailed discussions.
Supplementary Data
The crystallographic data for the [2]catenane in its bisradical dicationic state, which is available free of charge from the Cambridge Crystallographic Data Centre. The CCDC number is 2125118.
Rights and permissions
About this article
Cite this article
Jiao, Y., Qiu, Y., Zhang, L. et al. Electron-catalysed molecular recognition. Nature 603, 265–270 (2022). https://doi.org/10.1038/s41586-021-04377-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-04377-3
This article is cited by
-
Analytical noncovalent electrochemistry for battery engineering
Nature Chemical Engineering (2024)
-
Artificial molecular pumps
Nature Reviews Methods Primers (2024)
-
An electric molecular motor
Nature (2023)
-
Pillararene incorporated metal–organic frameworks for supramolecular recognition and selective separation
Nature Communications (2023)
-
Ultimate structures in catalysis: Single atoms, subnano-clusters, and electrons
Science China Materials (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.