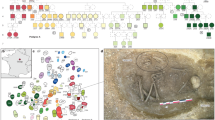
Abstract
To explore kinship practices at chambered tombs in Early Neolithic Britain, here we combined archaeological and genetic analyses of 35 individuals who lived about 5,700 years ago and were entombed at Hazleton North long cairn1. Twenty-seven individuals are part of the first extended pedigree reconstructed from ancient DNA, a five-generation family whose many interrelationships provide statistical power to document kinship practices that were invisible without direct genetic data. Patrilineal descent was key in determining who was buried in the tomb, as all 15 intergenerational transmissions were through men. The presence of women who had reproduced with lineage men and the absence of adult lineage daughters suggest virilocal burial and female exogamy. We demonstrate that one male progenitor reproduced with four women: the descendants of two of those women were buried in the same half of the tomb over all generations. This suggests that maternal sub-lineages were grouped into branches whose distinctiveness was recognized during the construction of the tomb. Four men descended from non-lineage fathers and mothers who also reproduced with lineage male individuals, suggesting that some men adopted the children of their reproductive partners by other men into their patriline. Eight individuals were not close biological relatives of the main lineage, raising the possibility that kinship also encompassed social bonds independent of biological relatedness.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The aligned sequences are available through the European Nucleotide Archive, accession PRJEB46958; the genotype dataset is available as a Supplementary Data file.
References
Saville, A. Hazleton North, Gloucestershire, 1979–82: the Excavation of a Neolithic Long Cairn of the Cotswold-Severn Group (Historic Buildings & Monuments Commission for England, 1990).
Brace, S. et al. Ancient genomes indicate population replacement in Early Neolithic Britain. Nat. Ecol. Evol. 3, 765–771 (2019).
Olalde, I. et al. The Beaker phenomenon and the genomic transformation of northwest Europe. Nature 555, 190–196 (2018).
Cassidy, L. M. et al. A dynastic elite in monumental Neolithic society. Nature 582, 384-388 (2020).
Mittnik, A. et al. Kinship-based social inequality in Bronze Age Europe. Science 366, 731–734 (2019).
Yaka, R. et al. Variable kinship patterns in Neolithic Anatolia revealed by ancient genomes. Curr. Biol. 31, 2455-2468.e18 (2021).
Amorim, C. E. G. et al. Understanding 6th-century barbarian social organization and migration through paleogenomics. Nat. Commun. 9, 3547 (2018).
Sánchez-Quinto, F. et al. Megalithic tombs in western and northern Neolithic Europe were linked to a kindred society. Proc. Natl Acad. Sci. USA 116, 9469–9474 (2019).
Scheib, C. L. et al. East Anglian early Neolithic monument burial linked to contemporary Megaliths. Ann. Hum. Biol. 46, 145–149 (2019).
Schneider, D. M. A Critique of the Study of Kinship (Univ. Michigan Press, 1984).
Carsten, J. After Kinship (Cambridge Univ. Press, 2003).
Brück, J. Ancient DNA, kinship and relational identities in Bronze Age Britain. Antiquity 95, 228–237 (2021).
Meadows, J., Barclay, A. & Bayliss, A. A short passage of time: the dating of the hazleton long cairn revisited. Cambridge Archaeol. J. 17, 45–64 (2007).
Rowley-Conwy, P. & Legge, T. in The Oxford Handbook of Neolithic Europe (eds. Fowler, C., Hofmann, D. & Harding, J.) 429–446 (Oxford Univ. Press, 2015).
Cuthbert, G. S. Enriching the Neolithic: the Forgotten People of the Barrows. PhD thesis, Univ. Exeter (2019).
Rogers, J. et al. in Hazleton North, Gloucestershire, 1979-82: The Excavation of a Neolithic Long Cairn of the Cotswold-Severn Group (ed. A. Saville) 182–198 (Historic Buildings & Monuments Commission for England, 1990).
Hedges, R., Saville, A. & O’Connell, T. Characterizing the diet of individuals at the Neolithic chambered tomb of Hazleton North, Gloucestershire, England, using stable isotopic analysis. Archaeometry 50, 114–128 (2008).
Charlton, S. et al. New insights into Neolithic milk consumption through proteomic analysis of dental calculus. Archaeol. Anthropol. Sci. 11, 6183–6196 (2019).
Neil, S., Evans, J., Montgomery, J. & Scarre, C. Isotopic evidence for residential mobility of farming communities during the transition to agriculture in Britain. R. Soc. Open Sci. 3, 150522 (2016).
Smith, M. & Brickley, M. People of the Long Barrows: Life, Death and Burial in Earlier Neolithic Britain (The History Press, 2009).
Surowiec, A., Snyder, K. T. & Creanza, N. A worldwide view of matriliny: using cross-cultural analyses to shed light on human kinship systems. Philos. Trans. R. Soc. B Biol. Sci. 374, 20180077 (2019).
Fowler, C. Social arrangements: kinship, descent and affinity in the mortuary architecture of Early Neolithic Britain and Ireland. Archaeol. Dialogues (in the press).
Robb, J. & Harris, O. J. T. Becoming gendered in European prehistory: was Neolithic gender fundamentally different? Am. Antiq. 83, 128–147 (2018).
Stone, L. & King, D. E. Kinship and Gender: An Introduction (Routledge, 2018).
Robb, J. What can we really say about skeletal part representation, MNI and funerary ritual? A simulation approach. J. Archaeol. Sci. Reports 10, 684–692 (2016).
Rohland, N., Glocke, I., Aximu-Petri, A. & Meyer, M. Extraction of highly degraded DNA from ancient bones, teeth and sediments for high-throughput sequencing. Nat. Protoc. 13, 2447–2461 (2018).
Gansauge, M. T., Aximu-Petri, A., Nagel, S. & Meyer, M. Manual and automated preparation of single-stranded DNA libraries for the sequencing of DNA from ancient biological remains and other sources of highly degraded DNA. Nat. Protoc. 15, 2279–2300 (2020).
Rohland, N., Harney, E., Mallick, S., Nordenfelt, S. & Reich, D. Partial uracil–DNA–glycosylase treatment for screening of ancient DNA. Philos. Trans. R. Soc. Lond. B 370, 20130624 (2015).
Fu, Q. et al. An early modern human from Romania with a recent Neanderthal ancestor. Nature 524, 216–219 (2015).
Fu, Q. et al. DNA analysis of an early modern human from Tianyuan Cave, China. Proc. Natl Acad. Sci. USA 110, 2223–2227 (2013).
Olalde, I. et al. The genomic history of the Iberian Peninsula over the past 8000 years. Science 363, 1230–1234 (2019).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Korneliussen, T. S., Albrechtsen, A. & Nielsen, R. ANGSD: analysis of next generation sequencing data. BMC Bioinformatics 15, 356 (2014).
Fu, Q. et al. A revised timescale for human evolution based on ancient mitochondrial genomes. Curr. Biol. 23, 553–559 (2013).
Weissensteiner, H. et al. HaploGrep 2: mitochondrial haplogroup classification in the era of high-throughput sequencing. Nucleic Acids Res. 44, W58-W63 (2016).
Kennett, D. J. et al. Archaeogenomic evidence reveals prehistoric matrilineal dynasty. Nat. Commun. 8, 14115 (2017).
van de Loosdrecht, M. et al. Pleistocene North African genomes link Near Eastern and sub-Saharan African human populations. Science 360, 548–552 (2018).
Busing, F. M. T. A., Meijer, E. & Van Der Leeden, R. Delete- m Jackknife for Unequal m. Stat. Comput. 9, 3–8 (1999).
Monroy Kuhn, J. M., Jakobsson, M. & Günther, T. Estimating genetic kin relationships in prehistoric populations. PLoS ONE 13, e0195491 (2018).
Williams, C. et al. A rapid, accurate approach to inferring pedigrees in endogamous populations. Preprint at https://doi.org/10.1101/2020.02.25.965376 (2020).
Kong, A. et al. Fine-scale recombination rate differences between sexes, populations and individuals. Nature 467, 1099–1103 (2010).
Hanghøj, K., Moltke, I., Andersen, P. A., Manica, A. & Korneliussen, T. S. Fast and accurate relatedness estimation from high-throughput sequencing data in the presence of inbreeding. Gigascience 8, giz034 (2019).
Patterson, N. et al. Ancient admixture in human history. Genetics 192, 1065–1093 (2012).
Cassidy, L. M. et al. Neolithic and Bronze Age migration to Ireland and establishment of the insular Atlantic genome. Proc. Natl Acad. Sci. USA 113, 368–373 (2016).
Lazaridis, I. et al. Ancient human genomes suggest three ancestral populations for present-day Europeans. Nature 513, 409–413 (2014).
Biagini, S. A. et al. People from Ibiza: an unexpected isolate in the Western Mediterranean. Eur. J. Hum. Genet. 27, 941–951 (2019).
Ringbauer, H., Novembre, J. & Steinrücken, M. Parental relatedness through time revealed by runs of homozygosity in ancient DNA. Nat. Commun. 12, 5425 (2021).
Acknowledgements
This work was supported by US National Institutes of Health grant GM100233, by the Allen Discovery Center programme, a Paul G. Allen Frontiers Group advised programme of the Paul G. Allen Family Foundation, by John Templeton Foundation grant 61220, by a gift from J.-F. Clin, and by the Howard Hughes Medical Institute. I.O. is supported by a Ramón y Cajal grant from Ministerio de Ciencia e Innovación, Spanish Government (RYC2019-027909-I/AEI/10.13039/501100011033). We thank J. Harris and A. Brookes at the Corinium Museum for providing permission to sample skeletal material from Hazleton North; T. Booth, A. Mittnik, H. Ringbauer and A. Whittle for valuable discussions; and N. Adamski, R. Bernardos, G. Bravo, K. Callan, E. Curtis, A. M. Lawson, M. Mah, S. Mallick, A. Micco, L. Qiu, K. Stewardson, A. Wagner, J. N. Workman and F. Zalzala for contributions to laboratory and bioinformatic work.
Author information
Authors and Affiliations
Contributions
N.R., O.C., S.C., R.P. and D.R. performed or supervised laboratory work. C.F., V.C., I.A., L.B. and S.C. assembled or contextualized archaeological material. C.F. and I.O. analysed data. C.F., I.O. and D.R. wrote the manuscript. C.F., I.O. and S.C. wrote the supplement.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Neil Carlin, Mehmet Somel and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 The Hazleton North chambered tomb.
a, Distribution of human remains in both chambers. The schematics in a are adapted from ref. 1, original figures © Historic England. b, Right humerus from Individual C showing helical fracture (red arrow), tooth marks (yellow arrow) and gnawed proximal and distal ends (white arrows).
Extended Data Fig. 2 Degrees of biological relatedness among individuals at Hazleton North.
(Supplementary Information Section 2.2). Pairs with fewer than 15,000 overlapping SNPs are indicated with an asterisk.
Extended Data Fig. 3 Using allelic mismatch rate patterns along the chromosomes to differentiate types of relationships for individuals sharing the same amount of DNA.
a, Differentiating between parent-offspring and sibling relationships. Allelic mismatch rate values across sliding windows of 20 Mb, moving by 1 Mb each step. As an example, we show values at chromosome 17 and include for reference a comparison between two unrelated Neolithic individuals from Britain (in brown), and a comparison between one individual and himself (in purple) to show how mismatch rates behave when two chromosomes are shared. The mismatch rate pattern for SP1m-SC1f is compatible with one chromosome shared along the entire chromosome 14 (in fact, along all autosomal chromosomes (Supplementary Table 6)), indicating a parent-offspring relationship. In contrast, the NC7f-SP3m comparison shows regions on chromosome 17 where no chromosome is shared (~65–70 Mb), other regions where two chromosomes are shared (~0–25 Mb) and other regions where one chromosome is shared (~25–60 Mb), compatible with a sibling relationship. b, Comparing DNA sharing patterns between SC9f and her paternal grandparents. We show mismatch rate values at chromosome 2 and include for reference a parent-offspring comparison (SE1m-SP2m; in blue) to show how mismatch rates behave when one chromosome is shared. Two recombination events (one at ~145 Mb and other at ~220 Mb) in SC9f’s father’s gamete result in SC9f sharing one chromosome with SC3m from the start of the chromosome to ~145 Mb, one chromosome with SC4f from 145 to 220 Mb and one chromosome with SC3m from 220 Mb to the end of the chromosome. This pattern of sharing one chromosome with either SC3m or SC4f (but never both) at every location of the genome is characteristic of comparisons between a grandchild and his/her two grandparents and is also observed in the other autosomal chromosomes.
Extended Data Fig. 4 Alternative family tree fitting all the genetic evidence except the IBD breakpoints co-localization analysis.
(Supplementary Section 2.4, Extended Data Figure 5). Individuals are coloured according to the female sub-lineage they belong to (NC1m and NC5m do not belong to any of the four major sub-lineages and are thus given a different color).
Extended Data Fig. 5 Using co-localization of IBD breakpoints to disambiguate between family tree in Fig. 1c and family tree in Extended Data Fig. 4.
a, We show mismatch rate values across sliding windows of 20 Mb on chromosome 3, moving by 1 Mb each step, for comparisons between SC3m and his four second-degree relatives. b, c, Recombination events on chromosome 3 needed to explain the observed mismatch rate patterns under b, the scenario of tree in Fig. 1c where 4 recombination events are required, or c, the scenario of the tree in Extended Data Fig. 4 where 10 recombination events are required including the extremely implausible occurrence of two recombination events at the same genomic locations in four different gametes.
Extended Data Fig. 6 Testing the validity of the family pedigree in Fig. 1c using X-chromosome relatedness and number of shared IBD segments.
a, Relatedness coefficients in the X-chromosome for first- and second-degree relationships with more than 300 overlapping SNPs. For each comparison, expected values according to the type of relationship in the family tree in Fig. 1c are shown in grey boxes. Bars represent 95% confidence intervals. b, Number of shared IBD segments on chromosomes 1-22 for first- and second-degree relationships. Pairs are grouped according to their type of relationship in the family tree in Fig. 1c.
Extended Data Fig. 7 Testing the consistency of the kinship results using NgsRelate42.
a, Correlation between the relatedness coefficient r and the Theta coefficient computed with NgsRelate, restricting to comparisons with more than 15,000 overlapping SNPs. b, Cotterman coefficients k0 and k2 for first- and second-degree relationships, as computed with NgsRelate.
Extended Data Fig. 8 Comparing autosomal relatedness between reproductive partners, different male reproductive partners of a female and different female reproductive partners of a male.
To estimate relatedness coefficients between unsampled and sampled male reproductive partners of a female, we doubled the relatedness coefficient obtained between the son of the unsampled male and the sampled male, to account for the fact that a son is one degree of relationship further away from their father’s relatives as compared to his father. Bars represent 95% confidence intervals.
Extended Data Fig. 9 Principal Component Analysis and inbreeding analysis.
a, Principal component analysis of Hazleton North individuals and other ancient individuals from Britain and Ireland. Ancient individuals were projected onto the principal components computed on a set of present-day West Eurasians genotyped on the Human Origins Array (not shown in the figure). Individuals with fewer than 15,000 SNPs on the Human Origins dataset were excluded for this analysis. b, Runs of homozygosity (ROH) in different length categories for the Hazleton North individuals with more than 400,000 SNPs covered. ROH were computed using hapROH47. Below, we plot the expected ROH length distribution for the offspring of closely related parents in outbred populations and for individuals from populations with small effective population size47.
Supplementary information
Supplementary Information
This Supplementary Information file contains four sections and additional references. Section 1: Osteological summary of human remains from Hazleton North. Section 2: Genetic analysis of biological relatedness and family tree reconstruction. Section 3: Statistical testing of kinship patterns. Section 4: Comparison of generational reconstruction with Bayesian model of radiocarbon dates from Hazleton North.
Supplementary Tables
This file contains Supplementary Tables 1–7.
Supplementary Data
Genotype dataset.
Rights and permissions
About this article
Cite this article
Fowler, C., Olalde, I., Cummings, V. et al. A high-resolution picture of kinship practices in an Early Neolithic tomb. Nature 601, 584–587 (2022). https://doi.org/10.1038/s41586-021-04241-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-04241-4
This article is cited by
-
Kinship practices at the early bronze age site of Leubingen in Central Germany
Scientific Reports (2024)
-
Accurate detection of identity-by-descent segments in human ancient DNA
Nature Genetics (2024)
-
Windows into the past: recent scientific techniques in dental analysis
British Dental Journal (2024)
-
The Allen Ancient DNA Resource (AADR) a curated compendium of ancient human genomes
Scientific Data (2024)
-
Shared chromosomal segments connect ancient human societies
Nature Genetics (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.