Abstract
The mammalian body plan is established shortly after the embryo implants into the maternal uterus, and our understanding of post-implantation developmental processes remains limited. Although pre- and peri-implantation mouse embryos are routinely cultured in vitro1,2, approaches for the robust culture of post-implantation embryos from egg cylinder stages until advanced organogenesis remain to be established. Here we present highly effective platforms for the ex utero culture of post-implantation mouse embryos, which enable the appropriate development of embryos from before gastrulation (embryonic day (E) 5.5) until the hindlimb formation stage (E11). Late gastrulating embryos (E7.5) are grown in three-dimensional rotating bottles, whereas extended culture from pre-gastrulation stages (E5.5 or E6.5) requires a combination of static and rotating bottle culture platforms. Histological, molecular and single-cell RNA sequencing analyses confirm that the ex utero cultured embryos recapitulate in utero development precisely. This culture system is amenable to the introduction of a variety of embryonic perturbations and micro-manipulations, the results of which can be followed ex utero for up to six days. The establishment of a system for robustly growing normal mouse embryos ex utero from pre-gastrulation to advanced organogenesis represents a valuable tool for investigating embryogenesis, as it eliminates the uterine barrier and allows researchers to mechanistically interrogate post-implantation morphogenesis and artificial embryogenesis in mammals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Bulk and single-cell RNA-seq data have been deposited in the Gene Expression Omnibus (GEO) database under accession number GSE149372. Source data are provided with this paper.
References
Bedzhov, I. & Zernicka-Goetz, M. Self-organizing properties of mouse pluripotent cells initiate morphogenesis upon implantation. Cell 156, 1032–1044 (2014).
White, M. D. et al. Long-lived binding of Sox2 to DNA predicts cell fate in the four-cell mouse embryo. Cell 165, 75–87 (2016).
New, D. A. T. Whole-embryo culture and the study of mammalian embryos during organogenesis. Biol. Rev. Camb. Philos. Soc. 53, 81–122 (1978).
Huang, Q. et al. Intravital imaging of mouse embryos. Science 368, 181–186 (2020).
Nicholas, J. S. & Rudnick, D. The development of rat embryos in tissue culture. Proc. Natl Acad. Sci. USA 20, 656–658 (1934).
New, D. A. T. & Stein, K. F. Cultivation of mouse embryos in vitro. Nature 199, 297–299 (1963).
New, D. A. T., Coppola, P. T. & Terry, S. Culture of explanted rat embryos in rotating tubes. J. Reprod. Fertil. 35, 135–138 (1973).
New, D. A. T. Development of explanted rat embryos in circulating medium. Development 17, 513–525 (1967).
Beddington, R. S. Induction of a second neural axis by the mouse node. Development 120, 613–620 (1994).
Parameswaran, M. & Tam, P. P. L. Regionalisation of cell fate and morphogenetic movement of the mesoderm during mouse gastrulation. Dev. Genet. 17, 16–28 (1995).
Tam, P. P. Postimplantation mouse development: whole embryo culture and micro-manipulation. Int. J. Dev. Biol. 42, 895–902 (1998).
Cantor, J. R. et al. Physiologic medium rewires cellular metabolism and reveals uric acid as an endogenous inhibitor of UMP synthase. Cell 169, 258–272 (2017).
Nagamatsu, G., Shimamoto, S., Hamazaki, N., Nishimura, Y. & Hayashi, K. Mechanical stress accompanied with nuclear rotation is involved in the dormant state of mouse oocytes. Sci. Adv. 5, eaav9960 (2019).
Ueda, Y. et al. Intrauterine pressures adjusted by Reichert’s membrane are crucial for early mouse morphogenesis. Cell Rep. 31, 107637 (2020).
Downs, K. M. & Davies, T. Staging of gastrulating mouse embryos by morphological landmarks in the dissecting microscope. Development 118, 1255–1266 (1993).
van Maele-Fabry, G., Delhaise, F. & Picard, J. J. Evolution of the developmental scores of sixteen morphological features in mouse embryos displaying 0 to 30 somites. Int. J. Dev. Biol. 36, 161–167 (1992).
Stelzer, Y. et al. Parent-of-origin DNA methylation dynamics during mouse development. Cell Rep. 16, 3167–3180 (2016).
McDole, K. et al. In toto imaging and reconstruction of post-implantation mouse development at the single-cell level. Cell 175, 859–876 (2018).
Ibarra-Soria, X. et al. Defining murine organogenesis at single-cell resolution reveals a role for the leukotriene pathway in regulating blood progenitor formation. Nat. Cell Biol. 20, 127–134 (2018).
Pijuan-Sala, B. et al. A single-cell molecular map of mouse gastrulation and early organogenesis. Nature 566, 490–495 (2019).
Cao, J. et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature 566, 496–502 (2019).
Saito, T. & Nakatsuji, N. Efficient gene transfer into the embryonic mouse brain using in vivo electroporation. Dev. Biol. 240, 237–246 (2001).
Beronja, S., Livshits, G., Williams, S. & Fuchs, E. Rapid functional dissection of genetic networks via tissue-specific transduction and RNAi in mouse embryos. Nat. Med. 16, 821–827 (2010).
Morgani, S., Nichols, J. & Hadjantonakis, A. K. The many faces of pluripotency: in vitro adaptations of a continuum of in vivo states. BMC Dev. Biol. 17, 7 (2017).
Kojima, Y. et al. The transcriptional and functional properties of mouse epiblast stem cells resemble the anterior primitive streak. Cell Stem Cell 14, 107–120 (2014).
Wu, J. et al. An alternative pluripotent state confers interspecies chimaeric competency. Nature 521, 316–321 (2015).
Huang, Y., Osorno, R., Tsakiridis, A. & Wilson, V. In vivo differentiation potential of epiblast stem cells revealed by chimeric embryo formation. Cell Rep. 2, 1571–1578 (2012).
van Wilgenburg, B., Browne, C., Vowles, J. & Cowley, S. A. Efficient, long term production of monocyte-derived macrophages from human pluripotent stem cells under partly-defined and fully-defined conditions. PLoS One 8, e71098 (2013).
Gafni, O. et al. Derivation of novel human ground state naive pluripotent stem cells. Nature 504, 282–286 (2013).
Harrison, S. E., Sozen, B., Christodoulou, N., Kyprianou, C. & Zernicka-Goetz, M. Assembly of embryonic and extraembryonic stem cells to mimic embryogenesis in vitro. Science 356, eaal1810 (2017).
Aguilera-Castrejon, A. & Hanna, J. H. Highly conducive ex utero mouse embryogenesis from pre-gastrulation to late organogenesis. Protoc. Exch. https://doi.org/10.21203/rs.3.pex-1372/v1 (2021).
Renier, N. et al. iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging. Cell 159, 896–910 (2014).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Aibar, S. et al. SCENIC: single-cell regulatory network inference and clustering. Nat. Methods 14, 1083–1086 (2017).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Acknowledgements
This work was funded by Pascal and Ilana Mantoux; the European Research Council (ERC-CoG-2016 726497-Cellnaivety); the Flight Attendant Medical Research Council (FAMRI); an Israel Cancer Research Fund (ICRF) professorship; BSF; the Helen and Martin Kimmel Institute for Stem Cell Research; the Helen and Martin Kimmel Award for Innovative Investigation; the Israel Science Foundation (ISF); Minerva; the Sherman Institute for Medicinal Chemistry; the Nella and Leon Benoziyo Center for Neurological Diseases; the David and Fela Shapell Family Center for Genetic Disorders Research; the Weizmann–U. Michigan program; the Kekst Family Institute for Medical Genetics; the Dr. Beth Rom-Rymer Stem Cell Research Fund; the Edmond de Rothschild Foundations; the Zantker Charitable Foundation; and the Estate of Zvia Zeroni. We thank O. Reiner and T. Sapir for help with mouse embryo electroporations; the Crown Genomics institute of the Nancy and Stephen Grand Israel National Center for Personalized Medicine at the Weizmann Institute for support with scRNA-seq; and the Weizmann Institute management and board for providing critical financial and infrastructural support. We dedicate this paper to the memories of R. Massarwa and H. Garty.
Author information
Authors and Affiliations
Contributions
A.A.-C. designed and conducted most of the wet lab, embryology, sequencing and imaging experiments, established the ex utero culture protocol and co-wrote the manuscript. B.O. conducted embryo injections, performed human microglia cultures and generated human–mouse chimeras, assisted in culture condition testing and processed cryosections. T.S. conducted bioinformatics analysis with N.N. supervising. R.M. helped to reproduce previously published protocols for ex utero culture and taught immunohistochemical protocols to our team. I.M. submitted Helsinki approval, collected cord blood and calibrated human cord serum production. N.G. recruited donors and performed cord blood extraction during caesarean sections. C.I. and S.S. assisted with human cord serum production. S.T. generated lentiviruses and assisted in embryo immunostaining and lentiviral infection of embryos. J.B., D.S. and S.V. performed tissue culture and bulk RNA sequencing of mouse pluripotent stem cells. V.C., S.A. and L.L. assisted with embryo immunostaining. N.L. performed characterization of cultured cells by qPCR. M.A. and H.K.-S. assisted with library preparation and single-cell RNA sequencing. Y.A. assisted with light sheet microscopy and live imaging. Y.R., S.C. and Y.S. generated tdTomato reporter embryos and assisted with allele imprinting experiments. M.Z. assisted with embryo injections. R.S. assembled and maintained the gas-pressure regulator module. J.H.H. conceived the idea for this project, conceptually designed the gas regulator module, established the ex utero culture protocol, supervised execution of experiments and adequate analysis of data, and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.H.H is an advisor to Biological Industries Ltd, and submitted a patent application that covers the roller and static culture conditions described herein (filed by J.H.H. and the Weizmann Institute of Science). R.S. is CEO of Arad Technologies Ltd. All other authors declare no competing interests.
Additional information
Peer review information Nature thanks Hiromitsu Nakauchi, Magdalena Zernicka-Goetz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Optimized gas-regulating module for roller culture incubators.
a, Diagram depicting the configuration of the gas-mixing box for gas concentration and pressure regulation. N2 and CO2 enter the gas-mixing box and are mixed by a centrifugal blower. Gases are then injected into a water bottle inside the incubator by a pressure pump that allows control of the gas pressure in the gas-mixing box that is transmitted to the sealed embryo bottle apparatus. The voltage on the pressure transmitter controls the pressure generated by the pressure pump in the gas mixing box. Lph, litres per hour. b, c, Top and front views of the gas controller module. d, e, Picture displaying the localization of the main components in the gas regulation module (d; listed in e). f, Interior of the precision incubator system (by B.T.C. Engineering, Cullum Starr Ltd) showing the direction of the gas flow (white arrowheads). g, Image of day 3 (E10.5) embryos cultured in rotating bottles (yellow arrowheads).
Extended Data Fig. 2 Establishment and optimization of a mouse embryo ex utero culture from late gastrulation (E7.5) until advanced organogenesis (E11).
a, E7.5 embryo dissection overview (see Methods). b, Percentage of normally developed embryos under different gas pressures and glucose or oxygen concentrations. Blue numbers indicate the conditions that yielded the highest efficiency of embryo survival. Values in parentheses denote the number of embryos assessed per condition at every sampled time point. Embryos that were dissected, fixed or moved to other conditions are subtracted from the total. Representative bright-field images of embryos cultured under certain conditions are shown to the right. c, Efficiency of normal embryonic development evaluated in different mouse genetic backgrounds. Parental mouse lines are indicated on the left (female: male). Values in parentheses show the numbers of embryos evaluated. PYS, parietal yolk sac; RS, rat serum. Scale bars, 500 μm.
Extended Data Fig. 3 Spatio-temporal expression patterns of ectoderm- and mesoderm-related lineage markers are recapitulated in ex utero cultured embryos.
Maximum intensity projections of embryos developed in utero and ex utero, fixed and immunostained for SOX2, OTX2, TUJ1, PAX6, SOX9, Brachyury, CDX2 and MHC-II (myosin heavy chain-II) at the indicated stages. Blue, DAPI. Images are representative of a minimum of three biological replicates. Scale bars, 100 μm (E7.5), 200 μm (E8.5, E9.5), and 500 μm (E10.5, E11.5).
Extended Data Fig. 4 In vivo spatio-temporal expression patterns of endoderm-related lineage markers are recapitulated in cultured embryos.
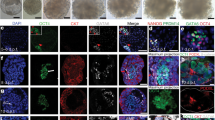
Maximum intensity projections of embryos developed in utero and ex utero, fixed and immunostained for SOX17, FOXA2 and GATA4 at the indicated stages. Blue, DAPI. For SOX17, insets are enlargements of the dashed boxes. Representative immunohistochemistry (mid-section, sagittal plane) images are shown for FOXA2 and GATA4 at the last time point (far-right panels). Images represent a minimum of three biological replicates. Scale bars, 100 μm (E7.5), 200 μm (E8.5, E9.5), and 500 μm (E10.5, E11.5).
Extended Data Fig. 5 Ex utero culture of GFP-reporter transgenic embryos.
a, Bright-field and GFP fluorescence images of ex utero embryos in culture at the specified times expressing the GFP reporter following activation by Wnt1-Cre and Isl1-Cre lineage-specific reporter alleles. n = 7 and 10 embryos for Wnt1-Cre and Isl1-Cre, respectively. Embryos dissected out of the yolk sac at +Day 4 are shown in the far-right panel. Scale bars, 500 μm. b, Representative confocal images of in utero E11.5 and ex utero +Day 4 transgenic mouse embryos expressing GFP following activation by Wnt1-Cre and Isl1-Cre reporter alleles. Scale bars, 1 mm. c, GFP fluorescence and bright-field images of in utero E10.5 and ex utero +Day 3 IG-DMR–GFP reporter embryos. n = 7 in utero; n = 7 ex utero. Scale bars, 500 μm.
Extended Data Fig. 6 Devising a platform for culturing mouse embryos from the onset of gastrulation until advanced organogenesis.
a–o, Schematic protocols indicating the percentages of E6.5 embryos that had developed properly per day in each condition. The medium composition, static or roller culture, and oxygen concentration are specified for each protocol. Values in parentheses denote the number of embryos evaluated per condition. Embryos that were dissected, fixed or moved to other conditions are subtracted from the total. Representative bright-field images of embryos cultured under certain conditions are shown to the right. Numbers in blue indicate the protocol that yielded the highest efficiency of embryo survival and was subsequently used throughout the study. Scale bars, 500 μm.
Extended Data Fig. 7 Embryos grown ex utero since early gastrulation recapitulate the spatio-temporal expression profiles of lineage markers seen in utero.
a–c, Maximum intensity projections of embryos developed ex utero, fixed, and immunostained for eleven specific markers at the indicated time points. Blue, DAPI. Images are representative of a minimum of three biological replicates. Scale bars, 50 μm (E6.5), 100 μm (+Day 1), 200 μm (+Day 2/3), 500 μm (+Day 4/5).
Extended Data Fig. 8 Single-cell transcriptomic analysis of ex utero +Day 2 and +Day 4 cultured embryos compared to in utero E8.5 and E10.5 embryos.
a, Schematic illustration of the embryo culture protocol and sequenced time points. Early-gastrulating (E6.5) embryos grown ex utero were processed for 10x Genomics scRNA-seq after 2 or 4 days of culture. b, Violin plot indicating the number of unique molecular identifiers (UMIs) and genes obtained per condition at each time point. E8.5, median of 9,787 UMIs and 2,989 genes detected per cell; E10.5, median of 4,795 UMIs and 1,789 genes detected per cell. c, d, Lineage annotation at culture days +2 (c) and +4 (d). Dot plots illustrating the area under the curve (AUC) enrichment value of overlapping cells across clusters and tissue lineages. Circle size denotes the magnitude of enrichment. Colours indicate P value (calculated from AUC). e, f, UMAP-based plots illustrating the normalized AUC assigned value of all individual cells for each lineage at culture days +2 (e) and +4 (f). g, Correlation of gene expression of the top 2,000 most variable genes per cluster between in utero E10.5 and ex utero +Day 4 embryos. Differentially expressed genes are named and shown as red dots. Clusters with the highest number of variable genes (range of 2–8 genes only per cluster) are encased in a red box. h, Pie charts depicting the proportional abundance of each cell cluster in both in utero and ex utero developed embryos at +Day 4/E10.5. Asterisks denote clusters with statistically significant differences between the two groups. Cluster 7, P = 0.004; cluster 8, P = 0.009; cluster 15, P = 0.001.
Extended Data Fig. 9 Changes in morphology and size in embryos developing ex utero from pre-gastrulation to the hindlimb formation stage.
a, Proportional increase in size of ex utero embryos grown from the onset of gastrulation (E6.5) to the 44-somite stage. Representative bright-field images of embryos cultured for 5 days are shown at each specific stage. Embryos without yolk sac are shown from day 3 to day 5. n ≥ 119. b, Percentages of normal embryos in cultures started at E6.5. c, Diagram depicting the embryonic axis measured at each stage (length of the antero-posterior axis (A-P) for E6.5 to E8.5 and crown–rump length for later stages). d, Measurements of embryonic length at the indicated time points. Dots represent individual embryos; in utero, n = 72, 25, 13, 19, 15, 38 (left to right); ex utero, n = 68, 29, 8, 19, 24; **Mann–Whitney test; ns, not significant. e, Bright-field images of E5.5 embryos grown ex utero for 6 days until the 42-somite stage. Embryos cultured since E5.5 exhibit a mild developmental delay of about 2–4 pairs of somites when compared to those developed in utero; however, overall morphological development seemed to occur correctly. f, Percentages of normal embryos in cultures started at E5.5. g, Representative increase in size of embryos cultured from E5.5 to the hindlimb stage (6 days of culture). Embryos dissected at the beginning and end of culture are shown. h, Immunostaining of pre-gastrulating (E5.5) embryos cultured for 6 days until the 42-somite stage. LEFTY1 and OCT4 immunostaining on a section of an E5.5 embryo (left); GATA4, MHC-II and SOX2 maximum intensity projection of an embryo at culture day 6 and stained (right). Scale bars, 50 μm (E5.5 embryos), 500 μm (all others). n, total number of embryos; x, number of experiments; all data represent mean ± s.e.m. Images are representative of a minimum of three embryos.
Extended Data Fig. 10 Ex utero culture medium supplemented with HBS supports embryo development from early/late gastrulation until the hindlimb stage (E11).
a, b, Bright-field microscopy images of mouse embryos grown ex utero from E7.5 (a) or E6.5 (b) with HCS replaced by in-house-prepared and freshly isolated adult HBS. c, Percentages of normal and defective embryos in cultures started at E7.5 and E6.5. n, total number of cultured embryos; x, number of experiments. Data represent mean ± s.e.m. Scale bars, 500 μm.
Extended Data Fig. 11 Ex utero manipulation of mouse embryonic development.
a, b, Percentages of developmentally normal (a) and GFP-expressing embryos (b) at 1–3 days after electroporation. c, Quantification of GFP+ cells in electroporated embryos at the indicated times. Dots represent individual embryos. d, e, Percentages of normally developed (d) and GFP+ embryos (e) after lentiviral transduction. Data represent mean ± s.e.m. f, Representative qPCR data showing the relative expression levels of mouse naive and primed markers in V6.5 mouse EpiS cells and formative EpiL cells, normalized to isogenic naive 2i/Lif ES cells. n = 3. g, Overlap in the transcriptional signature of differentially expressed genes measured by bulk RNA-seq in EpiS cells and ES cells used herein, compared to previously published datasets26. n = 2. h, Top, generation of mouse chimeras using isogenic naive ES cells. Bottom, GFP, SOX2 and GATA4 immunofluorescence images of chimeric embryos generated with naive ESCs. i, Whole-mount immunostaining of GFP+ cells detected in embryos injected with mouse EpiS cells or EpiL cells at E7.5, cultured ex utero for 1–4 days and stained for GFP, SOX2 and GATA4. Insets are enlargements of the dashed boxes. n ≥ 8 embryos. j, Percentages of chimeric embryos (GFP+ or tdT+) after micro-injection and ex utero culture. k, Immunostaining of +Day 1 cultured embryos injected with EpiS cells and EpiL cells in the anterior or distal epiblast. Images represent a minimum of three biological replicates. l, Representative confocal images of mouse post-implantation chimeras generated by tdT+ E7.5 in vivo epiblast orthotopic transplantation followed by ex utero culture for 1–4 days, stained for tdTomato, GATA4 or SOX9 and SOX2 or TUJ1. n ≥ 10 embryos. m, tdT+ embryos explanted at E7.5 and subjected to in toto live imaging of neural tube closure at E9.0. n = 3. n, Embryos cultured ex utero since E7.5 and exposed to vehicle or 1 mM valproic acid (VPA) from E8.5 to E9.5. n = 6. Inset shows magnification of the dashed box. Arrowheads, neural tube closure defects. Scale bars, 100 μm (m), 500 μm (all others). n, total number of embryos assessed; x, number of experiments.
Extended Data Fig. 12 Generation of human–mouse microglia interspecies chimeric embryos.
a, Protocol for differentiation of microglia progenitors from humans ES cells as previously described28. b, Flow cytometry dot plot to validate the identity of obtained microglia cells by co-expression of the microglia progenitor cell markers CD34+ and CD43+. n = 3 independent experiments. c, Merged bright-field and fluorescence images of E7.5 embryos injected with GFP+ human microglia progenitors at day 0. d, Representative immunofluorescence images of ex utero human microglia chimeric embryos 3 and 4 days after injection, labelled for GFP and TUJ1. The inset shows a magnification of the dashed box at day 4 identifying human nuclei (hNUMA), GFP and TUJ1. n = 11 embryos (day 3); n = 8 embryos (day 4). e, Quantification of GFP+ cells detected in human–mouse microglia chimeric embryos (excluding GFP+ cells found in the yolk sac). Dots represent individual embryos; n = 11 and 8 embryos for day 3 and day 4, respectively. f, Immunostaining for GFP and human TMEM119 in chimeric embryos. n = 3. g, Representative GFP immunofluorescence of a human microglia chimeric embryonic yolk sac and yolk sac vessel with circulating human GFP+ cells. n = 3. Scale bars, 50 μm (f), 500 μm (all others).
Supplementary information
Supplementary Information
This file contains Supplementary Video Legends, Supplementary Methods, Supplementary Discussion, and Supplementary References.
Supplementary Video 1
Ex utero mouse embryogenesis set-up. Part 1- Roller culture incubator with customized gas concentration and pressure regulation module for growing mouse embryos ex utero. Part 2- Media bottle exchange and transfer of an embryo into the roller culture system. Part 3- Setting up the roller culture incubator and gas regulation module.
Supplementary Video 2
Late-gastrulation to organogenesis ex utero roller culture. Representative snapshot bright field videos of E7.5 ex utero grown embryos after 1-4 days of culture.
Supplementary Video 3
Live embryo inside the culture bottle. Representative video of an embryo cultured in rotating bottles as seen by the naked eye.
Supplementary Video 4
Comparative light-sheet 3D reconstruction of ex utero and in utero embryos. E7.5 embryos grown 4 days ex utero and developmentally matched in utero embryos immunostained for Otx2/MHC-II, Sox2, and Tuj1/Sox9.
Supplementary Video 5
Robust in toto live imaging from gastrulation to somitogenesis. Representative confocal live imaging for 58 hours of an ex utero-grown tdTomato+ embryo from early gastrulation (E6.5) to somitogenesis (E8.5).
Supplementary Video 6
Early-gastrulation to organogenesis ex utero embryo culture. Representative bright field snapshot videos of E6.5 ex utero grown embryos after 1-5 days of culture.
Supplementary Video 7
Pre-gastrulation to organogenesis ex utero embryo culture. Representative bright field snapshot videos of E5.5 ex utero grown embryos after 1-6 days of culture.
Supplementary Video 8
Imaging neural tube closure in cultured embryos. Long-term in toto live imaging of neural tube closure in cultured embryos. E7.5 tdTomato+ mouse embryo cultured ex utero until E9.0 and subjected to live confocal imaging.
Rights and permissions
About this article
Cite this article
Aguilera-Castrejon, A., Oldak, B., Shani, T. et al. Ex utero mouse embryogenesis from pre-gastrulation to late organogenesis. Nature 593, 119–124 (2021). https://doi.org/10.1038/s41586-021-03416-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-03416-3
This article is cited by
-
Benchtop mesoSPIM: a next-generation open-source light-sheet microscope for cleared samples
Nature Communications (2024)
-
A 3D “sandwich” co-culture system with vascular niche supports mouse embryo development from E3.5 to E7.5 in vitro
Stem Cell Research & Therapy (2023)
-
The in vitro culture of mammalian embryos
Nature Methods (2023)
-
H3K36 methylation maintains cell identity by regulating opposing lineage programmes
Nature Cell Biology (2023)
-
Regulation, functions and transmission of bivalent chromatin during mammalian development
Nature Reviews Molecular Cell Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.