Abstract
The psychedelic alkaloid ibogaine has anti-addictive properties in both humans and animals1. Unlike most medications for the treatment of substance use disorders, anecdotal reports suggest that ibogaine has the potential to treat addiction to various substances, including opiates, alcohol and psychostimulants. The effects of ibogaine—like those of other psychedelic compounds—are long-lasting2, which has been attributed to its ability to modify addiction-related neural circuitry through the activation of neurotrophic factor signalling3,4. However, several safety concerns have hindered the clinical development of ibogaine, including its toxicity, hallucinogenic potential and tendency to induce cardiac arrhythmias. Here we apply the principles of function-oriented synthesis to identify the key structural elements of the potential therapeutic pharmacophore of ibogaine, and we use this information to engineer tabernanthalog—a water-soluble, non-hallucinogenic, non-toxic analogue of ibogaine that can be prepared in a single step. In rodents, tabernanthalog was found to promote structural neural plasticity, reduce alcohol- and heroin-seeking behaviour, and produce antidepressant-like effects. This work demonstrates that, through careful chemical design, it is possible to modify a psychedelic compound to produce a safer, non-hallucinogenic variant that has therapeutic potential.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data are available at https://doi.org/10.6084/m9.figshare.11634795. Source data are provided with this paper.
Code availability
Custom-written data analysis codes are available upon request from the corresponding author.
References
Wasko, M. J., Witt-Enderby, P. A. & Surratt, C. K. DARK classics in chemical neuroscience: ibogaine. ACS Chem. Neurosci. 9, 2475–2483 (2018).
Noller, G. E., Frampton, C. M. & Yazar-Klosinski, B. Ibogaine treatment outcomes for opioid dependence from a twelve-month follow-up observational study. Am. J. Drug Alcohol Abuse 44, 37–46 (2018).
He, D. Y. et al. Glial cell line-derived neurotrophic factor mediates the desirable actions of the anti-addiction drug ibogaine against alcohol consumption. J. Neurosci. 25, 619–628 (2005).
Marton, S. et al. Ibogaine administration modifies GDNF and BDNF expression in brain regions involved in mesocorticolimbic and nigral dopaminergic circuits. Front. Pharmacol. 10, 193 (2019).
Jenks, C. W. Extraction studies of Tabernanthe iboga and Voacanga africana. Nat. Prod. Lett. 16, 71–76 (2002).
Iyer, R. N., Favela, D., Zhang, G. & Olson, D. E. The iboga enigma: the chemistry and neuropharmacology of iboga alkaloids and related analogs. Nat. Prod. Rep. https://doi.org/10.1039/D0NP00033G (2020).
Hough, L. B., Pearl, S. M. & Glick, S. D. Tissue distribution of ibogaine after intraperitoneal and subcutaneous administration. Life Sci. 58, PL119–PL122 (1996).
Koenig, X., Kovar, M., Boehm, S., Sandtner, W. & Hilber, K. Anti-addiction drug ibogaine inhibits hERG channels: a cardiac arrhythmia risk. Addict. Biol. 19, 237–239 (2014).
Thurner, P. et al. Mechanism of hERG channel block by the psychoactive indole alkaloid ibogaine. J. Pharmacol. Exp. Ther. 348, 346–358 (2014).
Alper, K. R., Stajić, M. & Gill, J. R. Fatalities temporally associated with the ingestion of ibogaine. J. Forensic Sci. 57, 398–412 (2012).
Koenig, X. & Hilber, K. The anti-addiction drug ibogaine and the heart: a delicate relation. Molecules 20, 2208–2228 (2015).
Baumann, M. H., Pablo, J. P., Ali, S. F., Rothman, R. B. & Mash, D. C. Noribogaine (12-hydroxyibogamine): a biologically active metabolite of the antiaddictive drug ibogaine. Ann. NY Acad. Sci. 914, 354–368 (2000).
Olson, D. E. Psychoplastogens: a promising class of plasticity-promoting neurotherapeutics. J. Exp. Neurosci. 12, 1179069518800508 (2018).
Ly, C. et al. Psychedelics promote structural and functional neural plasticity. Cell Rep. 23, 3170–3182 (2018).
Bogenschutz, M. P. & Johnson, M. W. Classic hallucinogens in the treatment of addictions. Prog. Neuropsychopharmacol. Biol. Psychiatry 64, 250–258 (2016).
Wender, P. A., Verma, V. A., Paxton, T. J. & Pillow, T. H. Function-oriented synthesis, step economy, and drug design. Acc. Chem. Res. 41, 40–49 (2008).
Gassaway, M. M. et al. Deconstructing the iboga alkaloid skeleton: potentiation of FGF2-induced glial cell line-derived neurotrophic factor release by a novel compound. ACS Chem. Biol. 11, 77–87 (2016).
Wager, T. T., Hou, X., Verhoest, P. R. & Villalobos, A. Central nervous system multiparameter optimization desirability: application in drug discovery. ACS Chem. Neurosci. 7, 767–775 (2016).
Glennon, R. A., Young, R., Jacyno, J. M., Slusher, M. & Rosecrans, J. A. DOM-stimulus generalization to LSD and other hallucinogenic indolealkylamines. Eur. J. Pharmacol. 86, 453–459 (1983).
Dunlap, L. E. et al. Identification of psychoplastogenic N,N-dimethylaminoisotryptamine (isoDMT) analogs through structure–activity relationship studies. J. Med. Chem. 63, 1142–1155 (2020).
Halberstadt, A. L., Chatha, M., Klein, A. K., Wallach, J. & Brandt, S. D. Correlation between the potency of hallucinogens in the mouse head-twitch response assay and their behavioral and subjective effects in other species. Neuropharmacology 167, 107933 (2020).
McCarroll, M. N. et al. Zebrafish behavioural profiling identifies GABA and serotonin receptor ligands related to sedation and paradoxical excitation. Nat. Commun. 10, 4078 (2019).
Breuer, L. et al. “Herbal seizures” – atypical symptoms after ibogaine intoxication: a case report. J. Med. Case Rep. 9, 243 (2015).
Dach, K. et al. Teratological and behavioral screening of the national toxicology program 91-compound library in zebrafish (Danio rerio). Toxicol. Sci. 167, 77–91 (2019).
Rothman, R. B. & Baumann, M. H. Serotonergic drugs and valvular heart disease. Expert Opin. Drug Saf. 8, 317–329 (2009).
Phoumthipphavong, V., Barthas, F., Hassett, S., Kwan, A. C. Longitudinal effects of ketamine on dendritic architecture in vivo in the mouse medial frontal cortex. eNeuro 3, 0133-15 (2016).
Moda-Sava, R. N. et al. Sustained rescue of prefrontal circuit dysfunction by antidepressant-induced spine formation. Science 364, eaat8078 (2019).
Cameron, L. P. & Olson, D. E. Dark classics in chemical neuroscience: N,N-dimethyltryptamine (DMT). ACS Chem. Neurosci. 9, 2344–2357 (2018).
Warnault, V., Darcq, E., Levine, A., Barak, S. & Ron, D. Chromatin remodelling — a novel strategy to control excessive alcohol drinking. Transl. Psychiatry 3, e231 (2013).
Glick, S. D. et al. Effects of iboga alkaloids on morphine and cocaine self-administration in rats: relationship to tremorigenic effects and to effects on dopamine release in nucleus accumbens and striatum. Brain Res. 657, 14–22 (1994).
Giannotti, G., Barry, S. M., Siemsen, B. M., Peters, J. & McGinty, J. F. Divergent prelimbic cortical pathways interact with BDNF to regulate cocaine-seeking. J. Neurosci. 38, 8956–8966 (2018).
Glick, S. D., Kuehne, M. E., Maisonneuve, I. M., Bandarage, U. K. & Molinari, H. H. 18-Methoxycoronaridine, a non-toxic iboga alkaloid congener: effects on morphine and cocaine self-administration and on mesolimbic dopamine release in rats. Brain Res. 719, 29–35 (1996).
Carnicella, S., He, D. Y., Yowell, Q. V., Glick, S. D. & Ron, D. Noribogaine, but not 18-MC, exhibits similar actions as ibogaine on GDNF expression and ethanol self-administration. Addict. Biol. 15, 424–433 (2010).
Bandarage, U. K., Kuehne, M. E. & Glick, S. D. Total syntheses of racemic albifloranine and its anti-addictive congeners, including 18-methoxycoronaridine. Tetrahedron 55, 9405–9424 (1999).
Langheinrich, U., Vacun, G. & Wagner, T. Zebrafish embryos express an orthologue of HERG and are sensitive toward a range of QT-prolonging drugs inducing severe arrhythmia. Toxicol. Appl. Pharmacol. 193, 370–382 (2003).
Sampurna, B. P., Audira, G., Juniardi, S., Lai, Y.-H. & Hsiao, C.-D. A simple ImageJ-based method to measure cardiac rhythm in zebrafish embryos. Inventions 3, 21 (2018).
Westerfield, M. The zebrafish book. A guide for the laboratory use of zebrafish (Danio rerio) 5th edn (Univ. Oregon Press, 2007).
Ahrens, M. B., Orger, M. B., Robson, D. N., Li, J. M. & Keller, P. J. Whole-brain functional imaging at cellular resolution using light-sheet microscopy. Nat. Methods 10, 413–420 (2013).
Kroeze, W. K. et al. PRESTO-Tango as an open-source resource for interrogation of the druggable human GPCRome. Nat. Struct. Mol. Biol. 22, 362–369 (2015).
Barupal, D. K. et al. A comprehensive plasma metabolomics dataset for a cohort of mouse knockouts within the international mouse phenotyping consortium. Metabolites 9, 101 (2019).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Xu, T. et al. Rapid formation and selective stabilization of synapses for enduring motor memories. Nature 462, 915–919 (2009).
Chen, C. C., Lu, J., Yang, R., Ding, J. B. & Zuo, Y. Selective activation of parvalbumin interneurons prevents stress-induced synapse loss and perceptual defects. Mol. Psychiatry 23, 1614–1625 (2018).
Vazquez, M., Frazier, J. H., Reichel, C. M. & Peters, J. Acute ovarian hormone treatment in freely cycling female rats regulates distinct aspects of heroin seeking. Learn. Mem. 27, 6–11 (2020).
Acknowledgements
This work was supported by funds from the National Institutes of Health (NIH) (R01GM128997 to D.E.O.; R37AA01684 to D.R.; R01AA022583 to D.K.; R01MH109475, R01MH104227 and R01NS104950 to Y.Z.; R01DA045836 to J.P.; and U19AG023122 to O.F.), a Hellman Fellowship (D.E.O.), UC Davis STAIR and STAIR Plus grants (D.E.O.), a Max Planck Fellowship at MPFI (Y.Z.), four NIH training grants (T32GM113770 to R.J.T., T32MH112507 to L.P.C., 5T32GM099608 to M.V.V., and 4T32GM6754714 to D.M.-T.), two UC Davis Provost’s Undergraduate Fellowships (to J.V. and A.J.P.), the Paul G. Allen Family Foundation (M.N.M. and D.K.), the Genentech Fellowship Program (D.M.-T.), and a Medical College of Wisconsin Research Affairs Counsel Pilot Grant (J.D.M.). B.M.B. was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through grant number UL1 TR001860 and linked award TL1 TR001861. Delix Therapeutics funded the large receptor screen conducted at Eurofins Discovery. We thank F. F. Wagner for help in coordinating with Eurofins Discovery. This project used the Biological Analysis Core of the UC Davis MIND Institute Intellectual and Development Disabilities Research Center (U54 HD079125). The Olympus FV1000 confocal microscope used in this study was purchased using NIH Shared Instrumentation Grant 1S10RR019266-01. We thank the MCB Light Microscopy Imaging Facility, which is a UC Davis Campus Core Research Facility, for the use of this microscope. Several of the drugs used in this study were provided by the NIDA Drug Supply Program. We thank D. R. Carty for assistance with larval zebrafish toxicity assays.
Author information
Authors and Affiliations
Contributions
A.J.P., Z.Q.H. and G.Z. synthesized the ibogalogs. L.E.D. synthesized 5-MeO-DMT fumarate and performed the CNS MPO calculations. L.P.C. performed the dendritogenesis and spinogenesis assays. L.P.C. and J.V. performed the head-twitch response experiments. M.N.M. performed the zebrafish heart-rate and seizure experiments. J.C.T., D.M.-T. and R.J.T. performed the zebrafish behavioural experiments. R.J.T. and B.Y. performed the zebrafish toxicity assays. B.M.B. and L.P.C. performed the hERG inhibition studies. L.P.C. performed the solubility studies and conditioned place preference experiments. J.L., T.L. and L.P.C. performed the experiments assessing in vivo spine dynamics. L.J.L., E.I.A. and J.D.M. performed the receptor functional assays. J.L. and M.T. performed the forced swim test following UMS. M.V.V. performed the forced swim test study without UMS with assistance from L.E.D. Z.T.R. and L.P.C. performed the pharmacokinetic studies. J.P. performed the heroin self-administration experiments. Y.E. performed the alcohol consumption assays. L.P.C. performed the sucrose preference assay. O.F., H.W., J.D.M., P.J.L., D.K., D.R., J.P., Y.Z. and D.E.O. supervised various aspects of this project and assisted with data analysis. D.E.O. conceived the project and wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
D.E.O. is the president and chief scientific officer of Delix Therapeutics. Delix Therapeutics has licensed TBG-related technology from the University of California, Davis.
Additional information
Peer review information Nature thanks Amy Newman, Yavin Shaham and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
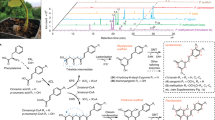
Extended Data Fig. 1 Synthesis of ibogalogs.
a, Ibogalogs lacking the tetrahydroazepine of ibogaine were synthesized in only a few steps. In brief, acylation of pyridine 1 under reductive conditions yielded the carboxybenzyl (Cbz)-protected dihydropyridine 2, which was immediately subjected to a Diels–Alder reaction with methyl vinyl ketone (3) followed by an in situ epimerization with NaOMe to afford an inseparable 1:1 mixture of exo (4a) and endo (4b) isomers (73% over 3 steps). Reaction of 4a and 4b with tosylhydrazide yielded the hydrazones 5a and 5b, which were separable via a combination of selective crystallization and chromatography (total yield of the two isomers, 75%). Caglioti reduction of the tosylhydrazones yielded 6a or 6b, which were readily converted to a variety of analogues via reaction sequences involving hydrogenolysis of the Cbz group, hydrogenation of the olefin, and C–N bond formation (Supplementary Information). b, Ibogalogs lacking the isoquinuclidine of ibogaine were synthesized in a single step through Fischer indole cyclization. See Supplementary Information for details.
Extended Data Fig. 2 The effects of ibogalogs on dendritogenesis.
a, Representative images of rat embryonic cortical neurons (DIV6) treated with the indicated compounds. Scale bar, 10 μm. b, Maximum numbers of crossings (Nmax) of the Sholl plots demonstrate that tetrahydroazepine-containing ibogalogs are more effective at increasing dendritic arbor complexity than are isoquinuclicine-containing ibogalogs. c, Sholl analysis (circle radii, 1.34-μm increments) demonstrates that cultured cortical neurons treated with several ibogalogs have more complex dendritic arbors compared to vehicle control (n = 52–83 neurons per treatment). The shaded area surrounding each line represents 95% confidence intervals. Control compounds, isoquinuclidines and tetrahydroazepines are shown in blue, purple and red, respectively. Exact n values for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility, and exact P values are reported in Methods and Supplementary Table 1.
Extended Data Fig. 3 TBG is safer than ibogaine.
a, Unlike ibogaine, IBG and TBG do not induce bradycardia in larval zebrafish. Sertindole (SI) was used as a positive control. b, Heat maps are shown representing aggregate larval zebrafish locomotor activity per well compared to vehicle controls (pseudo-Z-score). Red and blue indicate higher and lower activity than the mean of vehicle controls, respectively, while white indicates activity within ±1 s.d. of the control. Stimuli applied over time are indicated under the heat maps. Colours indicate bright LED light of respective colours, black traces represent the waveforms of acoustic stimuli, and grey vertical lines indicate physical tapping as secondary acoustic stimuli. c, Confusion matrix for classification of compounds (200 μM) plus vehicle and lethal controls. d, Concentration–response curves are shown for treated zebrafish subjected to the series of stimuli depicted in b. Lower percentages indicate treatments that were more often classified as vehicle (blue) or lethal (red). The solid line denotes the median and the shading denotes a 95th percentile confidence interval calculated by bootstrap. n = 8 wells per condition (64 zebrafish per condition). Blue lines indicate that all compounds produce behavioural phenotypes more distinct from vehicle at higher concentrations. Red lines indicate that known toxins (for example, PTZ, picrotoxin, endosulfan), known hERG inhibitors (sertindole, haloperidol, terfenadine) and iboga alkaloids (IBO, NOR) produce behavioural phenotypes more closely resembling a lethal phenotype as their concentrations are increased. Increasing concentrations of IBG or TBG do not produce lethal-like behavioural phenotypes. e, Transgenic larval zebrafish expressing GCaMP5G were immobilized in agarose, treated with compounds, and imaged over time. The known seizure-inducing compound PTZ was used as a positive control. Ibogaine and TBG were treated at 50 μM (n = 2 per condition). f, Proportion of viable and non-viable (malformed + dead) zebrafish following treatment with vehicle and TBG (66 μM) for 5 dpf (Fisher’s exact test: P = 0.3864). Representative images of zebrafish treated with vehicle and TBG (66 μM) for 2 and 5 dpf are shown. Scale bar, 2 mm. Exact n values for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility and exact P values are reported in the Methods and Supplementary Table 1.
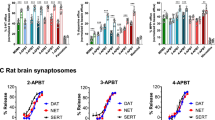
Extended Data Fig. 4 Concentration–response curves demonstrating the abilities of ibogalogs and related compounds to activate 5-HT and opioid receptors.
All compounds were assayed in parallel using the same drug dilutions. Graphs reflect representative concentration–response curves plotting mean and s.e.m. of data points performed in duplicate or triplicate. Assay details are described in Methods. Exact n values for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility and exact P values are reported in Methods and in Supplementary Table 1.
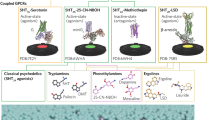
Extended Data Fig. 5 Pharmacological profiles of ibogalogs and related compounds.
EC50 and Emax estimates from at least two independent concentration-response curves performed in duplicate or triplicate. log(Emax/EC50) activity relative to the system Emax. Inactive, inactive in agonist mode; N.D., not determined; blue boxes indicate that the compound exhibits antagonist activity; dark grey boxes indicate that the compound is inactive in agonist mode but not tested in antagonist mode; orange boxes indicate that the compound is an inverse agonist. Ibogalogs are more selective 5-HT2A agonists than is 5-MeO-DMT. Exact n values for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility and exact P values are reported in Methods and in Supplementary Table 1.
Extended Data Fig. 6 High doses of TBG do not produce a conditioned place preference.
a, Schematic of the design of the conditioned place preference experiments. On day 1, the amount of time the mice spent in each distinct side of a two-chamber apparatus was recorded. Next, vehicle and TBG were administered to mice on alternating days while they were confined to chamber A (white box) or chamber B (grey parallel lines), respectively. Conditioning lasted for a total of 6 days. On day 8, preference for each distinct side of the two-chamber apparatus was assessed. b, A low dose of TBG (1 mg kg−1) did not produce conditioned place preference or conditioned place aversion. Higher doses (10 and 50 mg kg−1) produce a modest conditioned place aversion. c, TBG does not produce any long-lasting (>24 h) effects on locomotion. There is no statistical difference in locomotion between any pre- or post-conditioning groups (P = 0.9985, one-way ANOVA). White bars indicate groups before receiving TBG (that is, pre-conditioning), and blue bars indicate groups 24 h after the last TBG administration (that is, post-conditioning). Exact n values for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility and exact P values are reported in Methods and in Supplementary Table 1.
Extended Data Fig. 7 TBG produces antidepressant effects in mice.
a, Schematic illustrating the stressors used as part of the 7-day UMS protocol. White and grey boxes represent the light and dark phases of the light cycle, respectively. b, TBG rescues the effects of UMS on immobility. c, TBG (50 mg kg−1) reaches high brain concentrations and is rapidly eliminated from the body. Mice were administered 3 different doses of TBG via i.p. injection and euthanized either 15 min or 3 h later. Whole brains and livers were collected, dried, homogenized and extracted with MTBE. Quantification was accomplished using LC–MS and concentrations of TBG in the two organs were calculated. Several samples for the 10 and 1 mg kg−1 doses at the 3 h time point had TBG at levels below the limit of quantification (around 5 nmol g−1). In those cases, the values were recorded as 0. Exact n numbers for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility and exact P values are reported in Methods and in Supplementary Table 1.
Extended Data Fig. 8 Effects of TBG on locomotion and sucrose-seeking behaviour in rats.
a, Acute administration of TBG does not impair locomotion in the open field. Rats were subjected to novelty-induced locomotion (baseline) for 30 min. At that time, cocaine was administered and psychostimulant-induced locomotion (+cocaine) was assessed for 60 min. There were no differences between the vehicle- and TBG-treated groups with respect to total distance travelled or average velocity. Furthermore, there was no difference in thigmotaxis measured during the baseline period (that is, the percentage of time in the centre of the open field). b–e, A sucrose self-administration experiment was conducted in a similar manner to the heroin self-administration experiment in Fig. 4. Doses in mg kg−1 are shown in parentheses. b, Sucrose seeking over time is shown. Coloured arrows indicate when each group received TBG. VEH was administered at all other time points to each group. c, TBG acutely reduces sucrose-seeking behaviour in a dose-dependent manner when administered during self-administration. d, TBG acutely reduces sucrose seeking when administered immediately before the first extinction session. The CUE (injection 1 = vehicle; injection 2, vehicle) and EXT (injection 1, vehicle; injection 2, TBG) groups were compared, as they were matched for the number of withdrawal days between the last self-administration and first extinction session. e, TBG does not have long-lasting effects on sucrose-seeking behaviour, as it does not reduce active lever pressing during the cued reinstatement when administered 12–14 days previously during self-administration (SA) or immediately before extinction (EXT). Exact n values for each experimental condition are reported in Supplementary Table 1. Specific statistical tests, information on reproducibility and exact P values are reported in Methods and in Supplementary Table 1.
Supplementary information
Supplementary Information
Supplemental methods describing the synthesis of key compounds. Spectral data are provided.
Supplementary Table
Supplementary Table 1: The number of replicates for each experimental condition, statistical parameters for each experiment, and information on reproducibility are provided.
Video 1
: Ibogaine induces bradycardia. Larval zebrafish were immobilized in agarose and treated with either vehicle (VEH), ibogaine (IBO), ibogainalog (IBG), or tabernanthalog (TBG). Sertindole (SI) was used as a positive control.
Video 2
: PTZ induces excessive neural activity. Zebrafish larvae expressing GCaMP5G were immobilized in agarose and treated with either vehicle (VEH), ibogaine (IBO), or tabernanthalog (TBG). The seizure-inducing compound pentylenetetrazol (PTZ) was used as a positive control.
Rights and permissions
About this article
Cite this article
Cameron, L.P., Tombari, R.J., Lu, J. et al. A non-hallucinogenic psychedelic analogue with therapeutic potential. Nature 589, 474–479 (2021). https://doi.org/10.1038/s41586-020-3008-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-3008-z
This article is cited by
-
Therapeutic mechanisms of psychedelics and entactogens
Neuropsychopharmacology (2024)
-
Psychedelics: preclinical insights provide directions for future research
Neuropsychopharmacology (2024)
-
The role of neurotrophic factors in novel, rapid psychiatric treatments
Neuropsychopharmacology (2024)
-
Magnesium–ibogaine therapy in veterans with traumatic brain injuries
Nature Medicine (2024)
-
Psychedelics for acquired brain injury: a review of molecular mechanisms and therapeutic potential
Molecular Psychiatry (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.