Abstract
Influenza A virus causes millions of severe cases of disease during annual epidemics. The most abundant protein in influenza virions is matrix protein 1 (M1), which mediates virus assembly by forming an endoskeleton beneath the virus membrane1. The structure of full-length M1, and how it oligomerizes to mediate the assembly of virions, is unknown. Here we determine the complete structure of assembled M1 within intact virus particles, as well as the structure of M1 oligomers reconstituted in vitro. We find that the C-terminal domain of M1 is disordered in solution but can fold and bind in trans to the N-terminal domain of another M1 monomer, thus polymerizing M1 into linear strands that coat the interior surface of the membrane of the assembling virion. In the M1 polymer, five histidine residues—contributed by three different monomers of M1—form a cluster that can serve as the pH-sensitive disassembly switch after entry into a target cell. These structures therefore reveal mechanisms of influenza virus assembly and disassembly.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The cryo-EM and cryo-ET structures, and representative tomograms are deposited in the Electron Microscopy Data Bank (EMDB) under accession codes EMD-11075, EMD-11076, EMD-11077, EMD-11078 and EMD-11079. The associated molecular models are deposited in the PDB under accession codes 6Z5J and 6Z5L. Protein structures from published work that were used in this study are available in the PDB under accession codes: 1AA7, 1EA3, 5WCO and 5V6G. Sequences used in this study are available from UniProt.
References
Rossman, J. S. & Lamb, R. A. Influenza virus assembly and budding. Virology 411, 229–236 (2011).
Elster, C., Larsen, K., Gagnon, J., Ruigrok, R. W. & Baudin, F. Influenza virus M1 protein binds to RNA through its nuclear localization signal. J. Gen. Virol. 78, 1589–1596 (1997).
Noton, S. L. et al. Identification of the domains of the influenza A virus M1 matrix protein required for NP binding, oligomerization and incorporation into virions. J. Gen. Virol. 88, 2280–2290 (2007).
Chen, B. J., Takeda, M. & Lamb, R. A. Influenza virus hemagglutinin (H3 subtype) requires palmitoylation of its cytoplasmic tail for assembly: M1 proteins of two subtypes differ in their ability to support assembly. J. Virol. 79, 13673–13684 (2005).
Zhang, J., Pekosz, A. & Lamb, R. A. Influenza virus assembly and lipid raft microdomains: a role for the cytoplasmic tails of the spike glycoproteins. J. Virol. 74, 4634–4644 (2000).
Chu, C. M., Dawson, I. M. & Elford, W. J. Filamentous forms associated with newly isolated influenza virus. Lancet 253, 602 (1949).
Mosley, V. M. & Wyckoff, R. W. G. Electron micrography of the virus of influenza. Nature 157, 263 (1946).
Wrigley, N. G. Electron microscopy of influenza virus. Br. Med. Bull. 35, 35–38 (1979).
Ruigrok, R. W., Calder, L. J. & Wharton, S. A. Electron microscopy of the influenza virus submembranal structure. Virology 173, 311–316 (1989).
Nermut, M. V. Further investigation on the fine structure of influenza virus. J. Gen. Virol. 17, 317–331 (1972).
Fontana, J. & Steven, A. C. At low pH, influenza virus matrix protein M1 undergoes a conformational change prior to dissociating from the membrane. J. Virol. 87, 5621–5628 (2013).
Sha, B. & Luo, M. Structure of a bifunctional membrane-RNA binding protein, influenza virus matrix protein M1. Nat. Struct. Biol. 4, 239–244 (1997).
Arzt, S. et al. Combined results from solution studies on intact influenza virus M1 protein and from a new crystal form of its N-terminal domain show that M1 is an elongated monomer. Virology 279, 439–446 (2001).
Harris, A., Forouhar, F., Qiu, S., Sha, B. & Luo, M. The crystal structure of the influenza matrix protein M1 at neutral pH: M1–M1 protein interfaces can rotate in the oligomeric structures of M1. Virology 289, 34–44 (2001).
Zhang, W. et al. Crystal structure of an orthomyxovirus matrix protein reveals mechanisms for self-polymerization and membrane association. Proc. Natl Acad. Sci. USA 114, 8550–8555 (2017).
Sugita, Y., Noda, T., Sagara, H. & Kawaoka, Y. Ultracentrifugation deforms unfixed influenza A virions. J. Gen. Virol. 92, 2485–2493 (2011).
Vijayakrishnan, S. et al. Cryotomography of budding influenza A virus reveals filaments with diverse morphologies that mostly do not bear a genome at their distal end. PLoS Pathog. 9, e1003413 (2013).
Calder, L. J., Wasilewski, S., Berriman, J. A. & Rosenthal, P. B. Structural organization of a filamentous influenza A virus. Proc. Natl Acad. Sci. USA 107, 10685–10690 (2010).
Chlanda, P. et al. Structural analysis of the roles of influenza A virus membrane-associated proteins in assembly and morphology. J. Virol. 89, 8957–8966 (2015).
Ruigrok, R. W. H. et al. Membrane interaction of influenza virus M1 protein. Virology 267, 289–298 (2000).
Bourmakina, S. V. & García-Sastre, A. Reverse genetics studies on the filamentous morphology of influenza A virus. J. Gen. Virol. 84, 517–527 (2003).
Elleman, C. J. & Barclay, W. S. The M1 matrix protein controls the filamentous phenotype of influenza A virus. Virology 321, 144–153 (2004).
Burleigh, L. M., Calder, L. J., Skehel, J. J. & Steinhauer, D. A. Influenza A viruses with mutations in the M1 helix six domain display a wide variety of morphological phenotypes. J. Virol. 79, 1262–1270 (2005).
Shishkov, A. et al. Spatial structure peculiarities of influenza A virus matrix M1 protein in an acidic solution that simulates the internal lysosomal medium. FEBS J. 278, 4905–4916 (2011).
Chiang, M.-J. et al. Maintaining pH-dependent conformational flexibility of M1 is critical for efficient influenza A virus replication. Emerg. Microbes Infect. 6, e108 (2017).
Dahmani, I., Ludwig, K. & Chiantia, S. Influenza A matrix protein M1 induces lipid membrane deformation via protein multimerization. Biosci. Rep. 39, BSR20191024 (2019).
Zhang, X. et al. Atomic model of a nonenveloped virus reveals pH sensors for a coordinated process of cell entry. Nat. Struct. Mol. Biol. 23, 74–80 (2016).
Li, Z. & Blissard, G. W. Autographa californica multiple nucleopolyhedrovirus GP64 protein: roles of histidine residues in triggering membrane fusion and fusion pore expansion. J. Virol. 85, 12492–12504 (2011).
Calder, L. J. & Rosenthal, P. B. Cryomicroscopy provides structural snapshots of influenza virus membrane fusion. Nat. Struct. Mol. Biol. 23, 853–858 (2016).
Gui, L., Ebner, J. L., Mileant, A., Williams, J. A. & Lee, K. K. Visualization and sequencing of membrane remodeling leading to influenza virus fusion. J. Virol. 90, 6948–6962 (2016).
Martínez-Sobrido, L. & García-Sastre, A. Generation of recombinant influenza virus from plasmid DNA. J. Vis. Exp. 42, 2057 (2010).
Tobita, K., Sugiura, A., Enomote, C. & Furuyama, M. Plaque assay and primary isolation of influenza A viruses in an established line of canine kidney cells (MDCK) in the presence of trypsin. Med. Microbiol. Immunol. (Berl.) 162, 9–14 (1975).
Wan, W. et al. Structure and assembly of the Ebola virus nucleocapsid. Nature 551, 394–397 (2017).
Hagen, W. J. H., Wan, W. & Briggs, J. A. G. Implementation of a cryo-electron tomography tilt-scheme optimized for high resolution subtomogram averaging. J. Struct. Biol. 197, 191–198 (2017).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 116, 71–76 (1996).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Grant, T. & Grigorieff, N. Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. eLife 4, e06980 (2015).
Turoňová, B., Schur, F. K. M., Wan, W. & Briggs, J. A. G. Efficient 3D-CTF correction for cryo-electron tomography using NovaCTF improves subtomogram averaging resolution to 3.4Å. J. Struct. Biol. 199, 187–195 (2017).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Förster, F., Medalia, O., Zauberman, N., Baumeister, W. & Fass, D. Retrovirus envelope protein complex structure in situ studied by cryo-electron tomography. Proc. Natl Acad. Sci. USA 102, 4729–4734 (2005).
Nickell, S. et al. TOM software toolbox: acquisition and analysis for electron tomography. J. Struct. Biol. 149, 227–234 (2005).
Castaño-Díez, D., Kudryashev, M., Arheit, M. & Stahlberg, H. Dynamo: a flexible, user-friendly development tool for subtomogram averaging of cryo-EM data in high-performance computing environments. J. Struct. Biol. 178, 139–151 (2012).
Kovtun, O. et al. Structure of the membrane-assembled retromer coat determined by cryo-electron tomography. Nature 561, 561–564 (2018).
Tan, Y. Z. et al. Addressing preferred specimen orientation in single-particle cryo-EM through tilting. Nat. Methods 14, 793–796 (2017).
Rosenthal, P. B. & Henderson, R. Optimal determination of particle orientation, absolute hand, and contrast loss in single-particle electron cryomicroscopy. J. Mol. Biol. 333, 721–745 (2003).
Pervushin, K. Impact of transverse relaxation optimized spectroscopy (TROSY) on NMR as a technique in structural biology. Q. Rev. Biophys. 33, 161–197 (2000).
Delaglio, F. et al. NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J. Biomol. NMR 6, 277–293 (1995).
Mayzel, M., Rosenlöw, J., Isaksson, L. & Orekhov, V. Y. Time-resolved multidimensional NMR with non-uniform sampling. J. Biomol. NMR 58, 129–139 (2014).
Vranken, W. F. et al. The CCPN data model for NMR spectroscopy: development of a software pipeline. Proteins 59, 687–696 (2005).
Schwarzinger, S. et al. Sequence-dependent correction of random coil NMR chemical shifts. J. Am. Chem. Soc. 123, 2970–2978 (2001).
Ferrage, F., Zoonens, M., Warschawski, D. E., Popot, J.-L. & Bodenhausen, G. Slow diffusion of macromolecular assemblies by a new pulsed field gradient NMR method. J. Am. Chem. Soc. 125, 2541–2545 (2003).
García de la Torre, J., Huertas, M. L. & Carrasco, B. Calculation of hydrodynamic properties of globular proteins from their atomic-level structure. Biophys. J. 78, 719–730 (2000).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife 7, e42166 (2018).
Desfosses, A., Ciuffa, R., Gutsche, I. & Sachse, C. SPRING – an image processing package for single-particle based helical reconstruction from electron cryomicrographs. J. Struct. Biol. 185, 15–26 (2014).
Zivanov, J., Nakane, T. & Scheres, S. H. W. A Bayesian approach to beam-induced motion correction in cryo-EM single-particle analysis. IUCrJ 6, 5–17 (2019).
He, S. & Scheres, S. H. W. Helical reconstruction in RELION. J. Struct. Biol. 198, 163–176 (2017).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Kidmose, R. T. et al. Namdinator - automatic molecular dynamics flexible fitting of structural models into cryo-EM and crystallography experimental maps. IUCrJ 6, 526–531 (2019).
Afonine, P. V. et al. Real-space refinement in PHENIX for cryo-EM and crystallography. Acta Crystallogr. D 74, 531–544 (2018).
Barad, B. A. et al. EMRinger: side chain-directed model and map validation for 3D cryo-electron microscopy. Nat. Methods 12, 943–946 (2015).
Das, S. C. et al. The highly conserved arginine residues at positions 76 through 78 of influenza A virus matrix protein M1 play an important role in viral replication by affecting the intracellular localization of M1. J. Virol. 86, 1522–1530 (2012).
Baudin, F., Petit, I., Weissenhorn, W. & Ruigrok, R. W. In vitro dissection of the membrane and RNP binding activities of influenza virus M1 protein. Virology 281, 102–108 (2001).
Zhang, K. et al. Two polar residues within C-terminal domain of M1 are critical for the formation of influenza A virions. Cell. Microbiol. 17, 1583–1593 (2015).
Wu, C. Y., Jeng, K. S. & Lai, M. M. C. The SUMOylation of matrix protein M1 modulates the assembly and morphogenesis of influenza A virus. J. Virol. 85, 6618–6628 (2011).
Acknowledgements
We thank V. Sonntag-Buck, V. Zila, P. Chlanda, S. Klein, D. Morado, S. Scheres, C. Yu, C.-Y. Huang and W. Hagen for technical assistance, and P. Rosenthal, S. Gamblin, J. Skehel and D. Veesler for support during the preparation of helical arrays. All NMR data were acquired at the MRS facility of the MRC-LMB. This study made use of electron microscopes at EMBL and the MRC-LMB EM Facility, as well as high-performance computing resources at EMBL and LMB and the CIID CL2 imaging facility; we thank the staff who maintain these resources. Funding was provided to J.A.G.B. by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (ERC-CoG-648432 MEMBRANEFUSION), the Medical Research Council (MC_UP_1201/16) and the European Molecular Biology Laboratory; and to J.A.G.B. and H.G.K. by the Deutsche Forschungsgemeinschaft (project number 240245660 - SFB1129).
Author information
Authors and Affiliations
Contributions
X.X., H.G.K. and J.A.G.B. conceived the project. X.X. designed and performed in vitro helical assembly of M1. L.J.C. contributed to the initial identification of helical arrays. X.X., S.E. and S.M.V.F. designed and performed NMR experiments. J.P. and J.A.G.B. designed virus and VLP experiments, which were performed by J.P. O.S. and S.K. performed preliminary sample preparation and structural characterization of M1. J.P., X.X. and K.Q. collected cryo-EM and cryo-ET data. J.P. processed cryo-ET data with assistance from W.W. and J.A.G.B. X.X. processed cryo-EM data with assistance from K.Q. and J.A.G.B. J.P., X.X., S.E. and J.A.G.B. interpreted data. J.P., X.X. and J.A.G.B. wrote the original draft, which was edited and reviewed by all authors. J.A.G.B. and H.G.K. obtained funding. J.A.G.B. supervised and managed the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks John L. Rubinstein and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 HK68 virions and VLPs have variable numbers of M1 strands and variable radius.
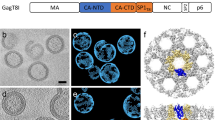
a, Slices through tomograms of two HK68 virus filaments, superimposed with a visualization of M1 subtomogram positions and orientations. Separate M1 strands are shown in different colours. Left, M1 arranged as three parallel left-handed helical strands. Right, M1 arranged as six parallel right-handed helical strands. Scale bars, 20 nm. Morphology is representative of three independent preparations of virus. b, As in a but for HK68 VLP filaments. Morphology is representative of five independent preparations of VLPs. c, Histogram showing the distribution of the number of helical strands (‘# helix starts’) for all HK68 virus (white) and VLP (grey) filaments analysed. Right-handed helices have positive start numbers, and left-handed helices have negative start numbers. d, Histogram showing the distribution of filament radii for all HK68 virus and VLP filaments analysed. Radii were determined at the position of the M1 NTD. e, A scatter plot of the number of helical strands against filament radius for each filament characterized in n = 52 virions from 1 representative preparation of virus, and n = 22 VLPs from 1 representative preparation of VLPs.
Extended Data Fig. 2 Comparison of HK68 virus and VLP M1 structures, and resolution measurements.
a, Representative orthoslice through a HK68 virus tomogram. Scale bar, 50 nm. Morphology is representative of three independent preparations of virus. b, Projection through 1.8 nm of the M1 subtomogram average obtained from HK68 virus data. c, Global FSC curve for the structure of M1 from HK68 virus (black), and analysis of anisotropy by FSC curves in x, y and z directions (blue, green and red, respectively). d–f, As in a–c, for VLP data. Morphology is representative of five independent preparations of VLPs. g, The structure of M1 from HK68 virus derived from subtomogram averaging, shown as a pink isosurface and fitted with the M1 NTD crystal structure. Cylinders represent the secondary structure elements of the M1 CTD. h, As in g, but for the subtomogram averaging structure of M1 from VLPs. We did not observe density that might correspond to the cytoplasmic tail of HA. i, FSC calculated between the virus M1 and VLP M1 structures, indicating that the structures are the same up to a resolution of 9 Å.
Extended Data Fig. 3 Analysis of the M1 structure determined within virions and VLPs.
a, Overlay of the model of M1 subunits as they are arranged in virions and VLPs with two neighbouring M1 NTD domains (pink) within the crystal packing in PDB 1EA313. Inset highlights the positions of the interhelix loops (L). b, Model of M1 subunits as they are arranged in virions and VLPs, shown in three different orientations (as in Fig. 2b). c, Surface charge representation for the NTD in equivalent orientations to those shown in a, coloured from negative (red) to positive (blue). Charged residues that form interstrand interfaces, and their respective helix numbers, are marked in the top and bottom panels. Basic residues shown in Fig. 2c and involved in membrane interaction are marked in the middle panel. Mutation of residues 77 and 78 to alanine reduces M1 incorporation into virions63. d, Overlay of the M1 model with two neighbouring infectious salmon anaemia virus matrix proteins (magenta) from the crystal packing in PDB 5WCO15. The difference in orientation of the CTD could reflect differences in influenza A virus or infectious salmon anaemia virus virion morphology, but we consider it more likely that it reflects the sequence divergence between infectious salmon anaemia virus and influenza A virus.
Extended Data Fig. 4 NMR analysis of M1.
a, 1H–15N HSQC spectrum of full-length M1 at pH 5. The spectrum is representative of three independent sample preparations. b, Representative diffusion curves acquired at pH 5 and 7, and fitted diffusion coefficients and calculated radii of hydration at pH values 5, 6, 7, 8 and 10. Error bars indicate the uncertainties of the intensities of the picked peak using Bruker Dynamics Center 2.6.1. HYDROPRO53 calculated radii of hydration are listed below for comparison. c, 1H–15N HSQC spectra of full-length M1 at different pH values. Increasing pH in absence of DNA and membrane does not induce folding of the CTD. At high pH (>8), only resonances from the first part of the NTD are visible. The protein remains largely monomeric throughout the pH titration. d, Secondary chemical shifts analysis. When compared to random coil, positive Cα and negative Cβ chemical shifts designate α-helical secondary structure. α-Helical segments from the single-particle structure are depicted above for comparison. Missing assignments are indicated by blue dots.
Extended Data Fig. 5 Electron microscopy of in vitro-reconstituted M1 helical tubes.
a, Negative-stain image of aggregated M1 tubes found within the pellet after sucrose cushion centrifugation. b, Negative-stain image of an in vitro-assembled M1 tube in the presence of nucleic acid. Images in a, b are representative of at least five independent preparations. c, A typical cryo-EM image of in vitro assembled M1 tubes at 2.7 μm underfocus, representative of three independent preparations. d, Selected class averages with tube diameter between 327–329 Å as determined by segclassexam. The lower left class average has minimal out-of-plane tilt and was used to test possible helical parameters by segclassreconstruct. e, Cryo-EM density of an M1 monomer is shown, the surface is coloured by local resolution of the map as determined by RELION. Boxes indicate regions magnified in g–i. f, Global FSC curve of the final in vitro-assembled M1 tube helical reconstruction (Fig. 2d). g–i, Magnified regions of cryo-EM density as indicated in e and their fitted molecular models.
Extended Data Fig. 6 Analysis of the in vitro M1 structure.
a, A view of M1 monomers extracted from the helical reconstruction to show the sites of interaction with the two nucleic acid strands (Fig. 2). One nucleic acid strand binds at the NTD–NTD interface (yellow), the other binds to a groove formed at the CTD–CTD interface (pink). Residues interacting with nucleic acid are all positively charged and are shown as sticks. Residues that form part of the nuclear localization signal that was previously shown to bind the viral ribonucleoprotein64 are underlined. b, Alignment of in situ (blue) and in vitro (green) M1 dimers extracted from their respective linear polymers. The differences are limited to small movements at the interfaces, perturbations in the orientation of α-helix 9 that accommodate the different curvatures, and a small change in the orientation of α-helix 12. It is possible that these differences reflect differences between spherical PR8 virions and filamentous HK68 virions, but we think it is more likely that they reflect the different curvature and the presence of nucleic acid in the in vitro sample. Inset highlights selected residues in the CTD. The mutations Ser183Ala and Thr185Ala cause spherical influenza A virus WSN to make more filamentous particles65. We speculate that these mutations may modulate folding of the CTD. Residue 204 is Glu in filamentous Udorn and HK68, but is Asp in spherical WSN, and the mutation Glu204Asp reduces the number of long filaments21. This residue is close to the C-terminal end of the neighbouring CTD and we speculate that this difference may influence this interaction. Residue 242 can be sumoylated66. This residue faces the inside of the virion where sumoylation could be accommodated without altering M1 packing. c, Alignment of the full-length M1 structure determined by helical reconstruction (green) to crystal structures of M1 NTD: PDB 1AA7 (blue)12, PDB 1EA3 (yellow)13 and PDB 5V6G (cyan)14. The structures are the same except for small differences in the H4–H5 loop.
Extended Data Fig. 7 Alignment of M1 protein sequences.
M1 sequences of the following viruses: influenza A M1 PR8 (A/Puerto Rico/8/1934 (H1N1)), influenza A M1 HK68 (A/Hong Kong/1/1968 (H3N2)), influenza A M1 (A/chicken/Fujian/25/2000 (H9N2)), bat influenza M1 H17N10 (A/little yellow-shouldered bat/Guatemala/164/2009 (H17N10)), bat influenza M1 H18N11 (A/flat-faced bat/Peru/033/2010 (H18N11)), influenza B M1 (B/Lee/1940) were downloaded from UniProt and aligned using mafft (https://mafft.cbrc.jp/alignment/software/). Locations of α-helices 1–12 are marked above the amino acid sequences. Conserved histidines are shaded blue, and conserved charged residues are shaded cyan. Substituted histidine locations and compensatory histidine substitutions are shaded red.
Supplementary information
Rights and permissions
About this article
Cite this article
Peukes, J., Xiong, X., Erlendsson, S. et al. The native structure of the assembled matrix protein 1 of influenza A virus. Nature 587, 495–498 (2020). https://doi.org/10.1038/s41586-020-2696-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2696-8
This article is cited by
-
Bridging structural and cell biology with cryo-electron microscopy
Nature (2024)
-
Influenza viral matrix 1 protein aggravates viral pathogenicity by inducing TLR4-mediated reactive oxygen species production and apoptotic cell death
Cell Death & Disease (2023)
-
Peptide Models of the Cytoplasmic Tail of Influenza A/H1N1 Virus Hemagglutinin Expand Understanding its pH-Dependent Modes of Interaction with Matrix Protein M1
The Protein Journal (2023)
-
Atomic model of vesicular stomatitis virus and mechanism of assembly
Nature Communications (2022)
-
An overview of influenza A virus genes, protein functions, and replication cycle highlighting important updates
Virus Genes (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.