Abstract
A hexanucleotide-repeat expansion in C9ORF72 is the most common genetic variant that contributes to amyotrophic lateral sclerosis and frontotemporal dementia1,2. The C9ORF72 mutation acts through gain- and loss-of-function mechanisms to induce pathways that are implicated in neural degeneration3,4,5,6,7,8,9. The expansion is transcribed into a long repetitive RNA, which negatively sequesters RNA-binding proteins5 before its non-canonical translation into neural-toxic dipeptide proteins3,4. The failure of RNA polymerase to read through the mutation also reduces the abundance of the endogenous C9ORF72 gene product, which functions in endolysosomal pathways and suppresses systemic and neural inflammation6,7,8,9. Notably, the effects of the repeat expansion act with incomplete penetrance in families with a high prevalence of amyotrophic lateral sclerosis or frontotemporal dementia, indicating that either genetic or environmental factors modify the risk of disease for each individual. Identifying disease modifiers is of considerable translational interest, as it could suggest strategies to diminish the risk of developing amyotrophic lateral sclerosis or frontotemporal dementia, or to slow progression. Here we report that an environment with reduced abundance of immune-stimulating bacteria10,11 protects C9orf72-mutant mice from premature mortality and significantly ameliorates their underlying systemic inflammation and autoimmunity. Consistent with C9orf72 functioning to prevent microbiota from inducing a pathological inflammatory response, we found that reducing the microbial burden in mutant mice with broad spectrum antibiotics—as well as transplanting gut microflora from a protective environment—attenuated inflammatory phenotypes, even after their onset. Our studies provide further evidence that the microbial composition of our gut has an important role in brain health and can interact in surprising ways with well-known genetic risk factors for disorders of the nervous system.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The 16S rDNA sequencing dataset are available through the Gene Expression Omnibus repository at GSE147325. All other data generated or analysed are included in the published Article and its Supplementary Information.
References
DeJesus-Hernandez, M. et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron 72, 245–256 (2011).
Majounie, E. et al. Frequency of the C9orf72 hexanucleotide repeat expansion in patients with amyotrophic lateral sclerosis and frontotemporal dementia: a cross-sectional study. Lancet Neurol. 11, 323–330 (2012).
Mori, K. et al. The C9orf72 GGGGCC repeat is translated into aggregating dipeptide-repeat proteins in FTLD/ALS. Science 339, 1335–1338 (2013).
Ash, P. E. A. et al. Unconventional translation of C9ORF72 GGGGCC expansion generates insoluble polypeptides specific to c9FTD/ALS. Neuron 77, 639–646 (2013).
Donnelly, C. J. et al. RNA toxicity from the ALS/FTD C9ORF72 expansion is mitigated by antisense intervention. Neuron 80, 415–428 (2013).
O’Rourke, J. G. et al. C9orf72 is required for proper macrophage and microglial function in mice. Science 351, 1324–1329 (2016).
Burberry, A. et al. Loss-of-function mutations in the C9ORF72 mouse ortholog cause fatal autoimmune disease. Sci. Transl. Med. 8, 347ra93 (2016).
Nassif, M., Woehlbier, U. & Manque, P. A. The enigmatic role of C9ORF72 in autophagy. Front. Neurosci. 11, 442 (2017).
Shi, Y. et al. Haploinsufficiency leads to neurodegeneration in C9ORF72 ALS/FTD human induced motor neurons. Nat. Med. 24, 313–325 (2018).
Whary, M. T. & Fox, J. G. Natural and experimental Helicobacter infections. Comp. Med. 54, 128–158 (2004).
Flannigan, K. L. & Denning, T. L. Segmented filamentous bacteria-induced immune responses: a balancing act between host protection and autoimmunity. Immunology 154, 537–546 (2018).
Ugolino, J. et al. Loss of C9orf72 enhances autophagic activity via deregulated mTOR and TFEB signaling. PLoS Genet. 12, e1006443 (2016).
Jiang, J. et al. Gain of toxicity from ALS/FTD-linked repeat expansions in C9ORF72 is alleviated by antisense oligonucleotides targeting GGGGCC-containing RNAs. Neuron 90, 535–550 (2016).
Atanasio, A. et al. C9orf72 ablation causes immune dysregulation characterized by leukocyte expansion, autoantibody production, and glomerulonephropathy in mice. Sci. Rep. 6, 23204 (2016).
Miller, Z. A. et al. Increased prevalence of autoimmune disease within C9 and FTD/MND cohorts. Neurol. Neuroimmunol. Neuroinflamm. 3, e301 (2016).
Fredi, M. et al. C9orf72 intermediate alleles in patients with amyotrophic lateral sclerosis, systemic lupus erythematosus, and rheumatoid arthritis. Neuromolecular Med. 21, 150–159 (2019).
Stine, J. G. & Lewis, J. H. Hepatotoxicity of antibiotics: a review and update for the clinician. Clin. Liver Dis. 17, 609–642, ix (2013).
Ransohoff, R. M. How neuroinflammation contributes to neurodegeneration. Science 353, 777–783 (2016).
McCauley, M. E. & Baloh, R. H. Inflammation in ALS/FTD pathogenesis. Acta Neuropathol. 137, 715–730 (2019).
Zhao, W., Beers, D. R. & Appel, S. H. Immune-mediated mechanisms in the pathoprogression of amyotrophic lateral sclerosis. J. Neuroimmune Pharmacol. 8, 888–899 (2013).
Zondler, L. et al. Peripheral monocytes are functionally altered and invade the CNS in ALS patients. Acta Neuropathol. 132, 391–411 (2016).
Zhang, G. X., Li, J., Ventura, E. & Rostami, A. Parenchymal microglia of naïve adult C57BL/6J mice express high levels of B7.1, B7.2, and MHC class II. Exp. Mol. Pathol. 73, 35–45 (2002).
Lall, D. & Baloh, R. H. Microglia and C9orf72 in neuroinflammation and ALS and frontotemporal dementia. J. Clin. Invest. 127, 3250–3258 (2017).
Zhang, Y. et al. The C9orf72-interacting protein Smcr8 is a negative regulator of autoimmunity and lysosomal exocytosis. Genes Dev. 32, 929–943 (2018).
Li, H. et al. Different neurotropic pathogens elicit neurotoxic CCR9- or neurosupportive CXCR3-expressing microglia. J. Immunol. 177, 3644–3656 (2006).
Krasemann, S. et al. The TREM2–APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity 47, 566–581.e9 (2017).
Keren-Shaul, H. et al. A unique microglia type associated with restricting development of Alzheimer’s disease. Cell 169, 1276–1290.e17 (2017).
Nilsson, H.-O. et al. High prevalence of Helicobacter species detected in laboratory mouse strains by multiplex PCR-denaturing gradient gel electrophoresis and pyrosequencing. J. Clin. Microbiol. 42, 3781–3788 (2004).
Blacher, E. et al. Potential roles of gut microbiome and metabolites in modulating ALS in mice. Nature 572, 474–480 (2019).
Zhai, R. et al. Strain-specific anti-inflammatory properties of two Akkermansia muciniphila strains on chronic colitis in mice. Front. Cell. Infect. Microbiol. 9, 239 (2019).
Erny, D. et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat. Neurosci. 18, 965–977 (2015).
Olson, C. A. et al. The gut microbiota mediates the anti-seizure effects of the ketogenic diet. Cell 173, 1728–1741.e13 (2018).
Harach, T. et al. Reduction of Abeta amyloid pathology in APPPS1 transgenic mice in the absence of gut microbiota. Sci. Rep. 7, 41802 (2017).
Sampson, T. R. et al. Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell 167, 1469–1480.e12 (2016).
Tremlett, H., Bauer, K. C., Appel-Cresswell, S., Finlay, B. B. & Waubant, E. The gut microbiome in human neurological disease: a review. Ann. Neurol. 81, 369–382 (2017).
Fang, X. et al. Evaluation of the microbial diversity in amyotrophic lateral sclerosis using high-throughput sequencing. Front. Microbiol. 7, 1479 (2016).
Brenner, D. et al. The fecal microbiome of ALS patients. Neurobiol. Aging 61, 132–137 (2018).
DeSantis, T. Z. et al. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 72, 5069–5072 (2006).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 7, 335–336 (2010).
Henderson, K. S. et al. Efficacy of direct detection of pathogens in naturally infected mice by using a high-density PCR array. J. Am. Assoc. Lab. Anim. Sci. 52, 763–772 (2013).
Acknowledgements
Support to K.E. was provided by The Merkin Fund at the Broad Institute, Target ALS, NIH 5R01NS089742, Harvard Stem Cell Institute and UCB. A.B. was supported by NIH 5K99AG057808-02, M.F.W. was supported by NIH 1K99MH119327-01. We thank J. Wang for providing faeces from mice housed Johns Hopkins University.
Author information
Authors and Affiliations
Contributions
A.B., M.F.W., J.M. and K.E. conceived the study. Experiments were performed by A.B. (Figs. 1–4, Extended Data Figs. 1–9), F.L. (Fig 1, Extended Data Fig. 2), M.F.W. (Fig. 1, Extended Data Fig. 2), K.S.S. (Fig. 4, Extended Data Figs. 8, 9), A.C. (Figs. 2–4, Extended Data Figs. 3–9), J.M. (Figs. 1–3, Extended Data Figs. 2–6, 8), N.v.G. (Extended Data Fig. 2), J.-Y.W. (Fig. 4, Extended Data Fig. 8), J.K. and G.G. (Extended Data Fig. 8), and M.Q. and P.E. and C.C. (Figs. 2, 3, Extended Data Figs. 3, 4, 8). A.B., F.L., K.S.S., J.K., G.G, N.v.G., J.-Y.W., O.P., I.K., D.T.S. and K.E. interpreted results. A.B. and K.E. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
K.E. is a co-founder of Q-State Biosciences, Quralis and Enclear Therapies.
Additional information
Peer review information Nature thanks Michael Fischbach, Aaron D. Gitler and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
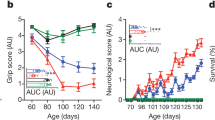
Extended Data Fig. 2 Causes of death, motor performance, levels of plasma cytokines and identification of pseudothrombocytopenia in C9orf72 LOF mice.
a, Causes of death or premature mortality of C9orf72(Harvard) mice in Fig. 1b. b, Accelerating rotarod performance of 37-week-old C9orf72(Harvard) neo-deleted mice (C9orf72(Harvard)+/+, n = 22; C9orf72(Harvard)+/−, n = 50; C9orf72(Harvard)−/−, n = 22). c, Accelerating rotarod performance of C9orf72(Broad) neo-deleted mice at 29 weeks of age (C9orf72(Broad)+/+, n = 53; C9orf72(Broad)+/−, n = 52; C9orf72(Broad)−/−, n = 48) or 42 weeks of age (C9orf72(Broad)+/+, n = 38; C9orf72(Broad)+/−, n = 48; C9orf72(Broad)−/−, n = 48). In b, c, one-way ANOVA with Dunnett’s multiple comparisons. Each point represents the average of three trials per mouse. d, Age at which mice in Fig. 1d–g were killed. One-way ANOVA with Sidak’s multiple comparisons. e, Plasma cytokines and chemokines at death from mice in Fig. 1d–g. Mean ± s.d. Two-way ANOVA with Tukey’s multiple comparisons. f, Peripheral blood smear of 18-week-old C9orf72(Harvard) neo-deleted mice. Platelets from C9orf72(Harvard)−/− mice were prone to aggregate (outlined by red dashed lines) in the presence of EDTA at 0 °C. g, Pseudothrombocytopenia could be reversed by warming the blood to room temperature. The reduced platelet count in the C9orf72(Harvard)−/− model therefore represents an indirect measure of anti-platelet auto-antibodies, rather than a reduction in platelet abundance. Two-way ANOVA with Tukey’s multiple comparisons. Each dot represents one mouse.
Extended Data Fig. 3 Cytokines and chemokines in lifelong-antibiotic-treated C9orf72 LOF mice and sex stratification of inflammatory phenotypes.
a, PCR analysis of Helicobacter spp. and norovirus DNA in faecal pellets. Each dot represents faeces from one cage. One-way ANOVA with Dunnett’s multiple comparisons. b, Plasma cytokines and chemokines of mice in Fig. 2. Mean ± s.d. Two-way ANOVA with Tukey’s multiple comparisons. c, Representative spleen size of mice in Fig. 2. d–f, Total blood neutrophil count (d), platelet count (e) and spleen weight (f) from mice in Fig. 2, stratified by sex. g, h, Total blood neutrophil count (g) and platelet count (h) in 30-week-old C9orf72(Harvard) neo-deleted mice stratified by sex (C9orf72(Harvard)+/+, n = 9 male and 13 female; C9orf72(Harvard)+/−, n = 25 male and 27 female; C9orf72(Harvard)−/−, n = 13 male and 9 female). i, Spleen weight in 40-week-old C9orf72(Harvard) neo-deleted mice stratified by sex (C9orf72(Harvard)+/+, n = 8 male and 11 female; C9orf72(Harvard)+/−, n = 13 male and 7 female; C9orf72(Harvard)−/−, n = 12 male and 6 female). In d–i, each dot represents one mouse. One- way ANOVA with Sidak’s multiple comparisons.
Extended Data Fig. 4 Acute antibiotic treatment improves motor function, and mitigates splenomegaly and cytokine burden, in C9orf72 LOF mice.
a, Accelerating rotarod performance of mice in Fig. 3a. Each point represents the average of three trials per mouse. Two-way ANOVA with Dunnett’s multiple comparisons. b, Plasma cytokines and chemokines of mice in Fig. 3a–d after seven weeks of treatment. c, d, Representative spleen size (c) and spleen weight (d) of mice in Fig. 3a after eight weeks of treatment. Each dot represents one mouse. One-way ANOVA with Sidak’s multiple comparisons. e, Plasma cytokines and chemokines of mice in Fig. 3e–h 10 weeks after faecal transplant. In b, e, mean ± s.d. Two-way ANOVA with Tukey’s multiple comparisons.
Extended Data Fig. 5 Bacteria and protozoa diversity across environments.
a, b, Phylum-level (a) and species-level (b) relative abundance of bacteria from 16S rDNA sequencing in Fig. 3i. Each bar represents sequencing from one pellet per cage. c, d, Relative abundance (c) and Gram-stain classification (d) of bacterial species, the abundance of which was significantly different between pro-inflammatory environments (Harvard BRI and Johns Hopkins University) and pro-survival environments (Broad Institute and Jackson Laboratory). t-test with Bonferroni multiple comparisons; 62/301 detected species had significance P < 0.0002. n = 5 faecal pellets per environment. Mean ± s.d. e, Quantitative PCR with reverse transcription analysis of T. muris 28S rDNA relative to total Eubacteria 16S rDNA in faeces. f, Simpson index of faecal α-diversity. g, Relative abundance of ε proteobacteria (Helicobacter). In e–g, each dot represents a faecal pellet from one cage. One-way ANOVA with Tukey’s multiple comparisons. h, PCR analysis of Helicobacter spp.16S rDNA and total Eubacteria 16S rDNA in faeces. i, PCR analysis of Helicobacter spp.16S rDNA and total Eubacteria 16S rDNA in faeces (six weeks after transplant) from Fig. 3e.
Extended Data Fig. 6 Environment-enriched bacteria engraft faecal-transplant recipients.
a–f, Analysis of bacteria in faeces at 10 weeks after transplant (from mice in Fig. 3e) by 16S rDNA sequencing. Each bar represents a faecal sample from an individual cage. a, b, Phylum-level (a) and species-level (b) relative abundance. c, Relative abundance of bacterial species grouped as those only observed in cages from Harvard BRI (Harvard-only), those only observed in cages from the Broad Institute (Broad-only), those observed in cages from Harvard BRI and the Broad Institute (Harvard/Broad-shared) or those not observed in Harvard BRI or Broad Institute cages but detectable in transplant recipient cages (emergent). d, Bray–Curtis dissimilarity matrix of faeces β-diversity. e, Relative abundance of ε proteobacteria (Helicobacter). f, Putative pro-inflammatory species (n = 27) enriched in pro-inflammatory environments (Harvard BRI and Johns Hopkins University) that were also enriched in Harvard-to-Harvard recipients, and putative pro-survival species (n = 12) enriched in pro-survival environments (Broad Institute and Jackson Laboratory) and enriched in Broad-to-Harvard recipients.
Extended Data Fig. 7 C9orf72 restricts myeloid cytokine release in response to foreign stimuli.
a–d, Analysis of cytokines and chemokines in supernatant 24 h after stimulation of bone-marrow-derived macrophages (BMDM) with activators of Toll-like receptor (Tlr) or NOD-like receptor (Nlr) agonists (a–c) or filtered Eubacteria-normalized faecal preparations (d). c, The abundance of cytokine and chemokine in the supernatant was normalized and colour-coded (blue, low; red, high) relative to the average level of each molecule in unstimulated C9orf72+/+ bone-marrow-derived macrophage wells. Levels of each analyte were measured by Luminex in multiplex. d, The abundance of total Eubacteria in each faecal sample was measured by qPCR for 16S rDNA and this value was used to normalize faecal Eubacteria bacteria concentration before generation of the dilution curve. Each dot represents one well. Panels are representative of n = 2 replicate experiments (a); n = 5 replicate experiments (b); one representative experiment with average of n = 3 technical replicates per condition (c); n = 2 replicate experiments (d). In a, two-way ANOVA with Sidak’s multiple comparison. In b, two-way ANOVA with Dunnett’s multiple comparisons. In c, two-way ANOVA with Sidak’s multiple comparison for each analyte tested. In d, one-way ANOVA with Sidak’s multiple comparisons.
Extended Data Fig. 8 Neutrophils and T cells infiltrate spinal cord of C9orf72 LOF mice.
a–f, Mass cytometry investigation of single-cell-dissociated forebrain or spinal cord from 36-week-old C9orf72(Harvard) neo-deleted male and female mice (C9orf72(Harvard)+/+, n = 7; C9orf72(Harvard)+/−, n = 7; C9orf72(Harvard)−/−, n = 8). One C9orf72 +/+ forebrain sample failed, and was excluded from analysis. Representative gating scheme can be found in Supplementary Information. Populations were defined as CD45mid CX3CR1+CD39+ microglia (a), CD45highLy6C+Ly6Ghigh neutrophils (b), CD45highLy6C+Ly6Glow monocytes (c), CD45highCD3e+CD4+ T cells (d), CD45highCD3e+CD4− T cells (e) and CD45highCD19+ B cells (f). Quantification of total cells per tissue was obtained by multiplying the percentage of each gated population by the total cells recovered from the tissue of that mouse. Each dot represents one mouse. Two-way ANOVA with Dunnett’s multiple comparisons. g, Quantitative PCR with reverse transcription of Ly6c expression in total cortex tissue of 47-week-old C9orf72(Harvard) neo-deleted mice (C9orf72(Harvard)+/+, n = 8; C9orf72(Harvard)−/−, n = 9) or C9orf72(Broad) neo-deleted mice (C9orf72(Broad)+/+, n = 10; C9orf72(Broad)−/−, n = 9). Each dot represents one mouse. One-way ANOVA with Sidak’s multiple comparisons. h, Orthogonal projection of confocal imaging of CD11b and mouse immunoglobulin IgG in lumbar spinal cord of a 43-week-old C9orf72(Harvard) mouse.
Extended Data Fig. 9 Elevated lysosomal proteins and microgliosis in spinal cords of C9orf72 LOF mice.
a, b, e–g, Orthogonal projection and quantification of confocal imaging of LAMP1 (a), cathepsin B (b), CCR9 (e), dectin 1 (CLEC7A) (f) and LPL (g) in IBA1+ microglia in spinal cord of a 55-week-old C9orf72(Harvard) mouse. One-way ANOVA with Sidak’s multiple comparisons. Each dot represents the average mean fluorescent intensity (MFI) of the antigen within microglia on a given spinal cord section. Over 100 microglia were surveyed per section. Sections from n = 3 C9orf72+/+ and n = 3 C9orf72−/− mice were surveyed. c, d, Flow cytometry quantification of LAMP1 (c) or cathepsin B (d) in CD45midCD11b+CD39+ microglia from spinal cord of C9orf72(Harvard) neo-deleted mice in Fig. 2. One-way ANOVA with Sidak’s multiple comparisons. h, Graphical illustration of C9orf72 functioning within the haematopoietic system to restrict the development of inflammation, autoimmunity, peripheral immune infiltration into the central nervous system (CNS) and microgliosis in response to hyper-stimulatory communities of gut microflora. The microglia image was modified from Servier Medical Art (https://smart.servier.com/smart_image/microglia-2/) under a CC BY 3.0 licence.
Supplementary information
Supplementary Table
SI Table 1 | Difference in microbiota between vivaria and SI Mass cytometry gating | Forebrain and spinal cord population gating.
Supplementary Figures
SI Un-cropped gel images | Un-cropped gel images for Extended Data Fig 5h-i.
Video 1
SI Video. 1 | CD45 (Green) and mouse IgG (Red) staining of C9orf72 +/+ spinal cord (mp4).
Video 2
SI Video. 2 | CD45 (Green) and mouse IgG (Red) staining of C9orf72 -/- spinal cord (mp4).
Video 3
SI Video. 3 | Iba1 (Green) and Lamp1 (Magenta) staining of C9orf72 +/+ spinal cord (mp4).
Video 4
SI Video. 4 | Iba1 (Green) and Lamp1 (Magenta) staining of C9orf72 -/- spinal cord (mp4).
Video 5
SI Video. 5 | CD11b (Green) and Cathepsin B (Magenta) staining of C9orf72 +/+ spinal cord (mp4).
Video 6
SI Video. 6 | CD11b (Green) and Cathepsin B (Magenta) staining of C9orf72 -/- spinal cord (mp4).
Video 7
SI Video. 7 | Iba1 (Green) and Dectin1 (Magenta) staining of C9orf72 +/+ spinal cord (mp4).
Video 8
SI Video. 8 | Iba1 (Green) and Dectin1 (Magenta) staining of C9orf72 -/- spinal cord (mp4).
Source data
Rights and permissions
About this article
Cite this article
Burberry, A., Wells, M.F., Limone, F. et al. C9orf72 suppresses systemic and neural inflammation induced by gut bacteria. Nature 582, 89–94 (2020). https://doi.org/10.1038/s41586-020-2288-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2288-7
This article is cited by
-
Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases
Signal Transduction and Targeted Therapy (2024)
-
Molecular hallmarks of ageing in amyotrophic lateral sclerosis
Cellular and Molecular Life Sciences (2024)
-
Gut-Brain Axis Deregulation and Its Possible Contribution to Neurodegenerative Disorders
Neurotoxicity Research (2024)
-
Neuroprotective Effect of a Multistrain Probiotic Mixture in SOD1G93A Mice by Reducing SOD1 Aggregation and Targeting the Microbiota-Gut-Brain Axis
Molecular Neurobiology (2024)
-
Humanized APOE genotypes influence lifespan independently of tau aggregation in the P301S mouse model of tauopathy
Acta Neuropathologica Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.