Abstract
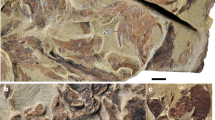
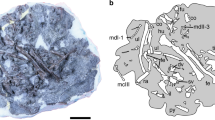
Our understanding of the earliest stages of crown bird evolution is hindered by an exceedingly sparse avian fossil record from the Mesozoic era. The most ancient phylogenetic divergences among crown birds are known to have occurred in the Cretaceous period1,2,3, but stem-lineage representatives of the deepest subclades of crown birds—Palaeognathae (ostriches and kin), Galloanserae (landfowl and waterfowl) and Neoaves (all other extant birds)—are unknown from the Mesozoic era. As a result, key questions related to the ecology4,5, biogeography3,6,7 and divergence times1,8,9,10 of ancestral crown birds remain unanswered. Here we report a new Mesozoic fossil that occupies a position close to the last common ancestor of Galloanserae and fills a key phylogenetic gap in the early evolutionary history of crown birds10,11. Asteriornis maastrichtensis, gen. et sp. nov., from the Maastrichtian age of Belgium (66.8–66.7 million years ago), is represented by a nearly complete, three-dimensionally preserved skull and associated postcranial elements. The fossil represents one of the only well-supported crown birds from the Mesozoic era12, and is the first Mesozoic crown bird with well-represented cranial remains. Asteriornis maastrichtensis exhibits a previously undocumented combination of galliform (landfowl)-like and anseriform (waterfowl)-like features, and its presence alongside a previously reported Ichthyornis-like taxon from the same locality13 provides direct evidence of the co-occurrence of crown birds and avialan stem birds. Its occurrence in the Northern Hemisphere challenges biogeographical hypotheses of a Gondwanan origin of crown birds3, and its relatively small size and possible littoral ecology may corroborate proposed ecological filters4,5,9 that influenced the persistence of crown birds through the end-Cretaceous mass extinction.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The holotype specimen of A. maastrichtensis is deposited in the permanent collection of the Natuurhistorisch Museum Maastricht under collection number NHMM 2013 008. Digital models of the A. maastrichtensis skull and postcranial elements, .tre files from phylogenetic analyses and CT scans of the A. maastrichtensis holotype are available at Zenodo (doi: 10.5281/zenodo.3610226). The Life Science Identifier for Asteriornis is urn:lsid:zoobank.org:pub:32192A46-4A43-48CE-8F17-447900FCC6DF.
References
Prum, R. O. et al. A comprehensive phylogeny of birds (Aves) using targeted next-generation DNA sequencing. Nature 526, 569–573 (2015).
Jarvis, E. D. et al. Whole-genome analyses resolve early branches in the tree of life of modern birds. Science 346, 1320–1331 (2014).
Claramunt, S. & Cracraft, J. A new time tree reveals Earth history’s imprint on the evolution of modern birds. Sci. Adv. 1, e1501005 (2015).
Field, D. J. et al. Early evolution of modern birds structured by global forest collapse at the end-Cretaceous mass extinction. Curr. Biol. 28, 1825–1831 (2018).
Larson, D. W., Brown, C. M. & Evans, D. C. Dental disparity and ecological stability in bird-like dinosaurs prior to the end-Cretaceous mass extinction. Curr. Biol. 26, 1325–1333 (2016).
Mayr, G. Avian higher level biogeography: Southern Hemispheric origins or Southern Hemispheric relicts? J. Biogeogr. 44, 956–958 (2017).
Saupe, E. E. et al. Climatic shifts drove major contractions in avian latitudinal distributions throughout the Cenozoic. Proc. Natl Acad. Sci. USA 116, 12895–12900 (2019).
Ksepka, D. T. & Phillips, M. J. Avian diversification patterns across the K–Pg boundary: influence of calibrations, datasets, and model misspecification. Ann. Mo. Bot. Gard. 100, 300–328 (2015).
Berv, J. S. & Field, D. J. Genomic signature of an avian Lilliput effect across the K–Pg extinction. Syst. Biol. 67, 1–13 (2018).
Field, D. J. et al. Timing the extant avian radiation: the rise of modern birds, and the importance of modeling molecular rate variation. PeerJ Preprints 7, e27521v1 (2019).
Mayr, G. Avian Evolution (Wiley, 2016).
Clarke, J. A., Tambussi, C. P., Noriega, J. I., Erickson, G. M. & Ketcham, R. A. Definitive fossil evidence for the extant avian radiation in the Cretaceous. Nature 433, 305–308 (2005).
Dyke, G. J. et al. Europe’s last Mesozoic bird. Naturwissenschaften 89, 408–411 (2002).
Xing, L., Stanley, E. L., Bai, M. & Blackburn, D. C. The earliest direct evidence of frogs in wet tropical forests from Cretaceous Burmese amber. Sci. Rep. 8, 8770 (2018).
Simões, T. R. et al. The origin of squamates revealed by a Middle Triassic lizard from the Italian Alps. Nature 557, 706–709 (2018).
Evers, S. W., Barrett, P. M. & Benson, R. B. J. Anatomy of Rhinochelys pulchriceps (Protostegidae) and marine adaptation during the early evolution of chelonioids. PeerJ 7, e6811 (2019).
Bi, S. et al. An Early Cretaceous eutherian and the placental–marsupial dichotomy. Nature 558, 390–395 (2018).
Lee, M. S. Y. & Yates, A. M. Tip-dating and homoplasy: reconciling the shallow molecular divergences of modern gharials with their long fossil record. Proc. R. Soc. Lond. B 285, 20181071 (2018).
Hope, S. in Mesozoic Birds: Above the Heads of Dinosaurs (eds Chiappe, L. M. & Witmer, L. M.) 339–388 (Univ. California Press, 2002).
Longrich, N. R., Tokaryk, T. & Field, D. J. Mass extinction of birds at the Cretaceous–Paleogene (K–Pg) boundary. Proc. Natl Acad. Sci. USA 108, 15253–15257 (2011).
Mayr, G. Paleogene Fossil Birds (Springer, 2009).
Clyde, W. C., Ramezani, J., Johnson, K. R., Bowring, S. A. & Jones, M. M. Direct high-precision U–Pb geochronology of the end-Cretaceous extinction and calibration of Paleocene astronomical timescales. Earth Planet. Sci. Lett. 452, 272–280 (2016).
Gauthier, J. A. & de Queiroz, K. in New Perspectives on the Origin and Early Evolution of Birds: Proceedings of the International Symposium in Honor of John H. Ostrom (eds Gauthier, J. & Gall, L. F.) 7–41 (Peabody Museum of Natural History, Yale University, 2001).
Keutgen, N. A bioclast-based astronomical timescale for the Maastrichtian in the type area (southeast Netherlands, northeast Belgium) and stratigraphic implications: the legacy of PJ Felder. Neth. J. Geosci. 97, 229–260 (2018).
Field, D. J., Lynner, C., Brown, C. & Darroch, S. A. F. Skeletal correlates for body mass estimation in modern and fossil flying birds. PLoS One 8, e82000 (2013).
Olson, S. L. & Feduccia, A. Presbyornis and the origin of the Anseriformes (Aves: Charadriomorphae). Smithson. Contrib. Zool. 323, 1–24 (1980).
Elzanowski, A. & Stidham, T. A. Morphology of the quadrate in the Eocene anseriform Presbyornis and extant galloanserine birds. J. Morphol. 271, 305–323 (2010).
Worthy, T. H., Degrange, F. J., Handley, W. D. & Lee, M. S. Y. The evolution of giant flightless birds and novel phylogenetic relationships for extinct fowl (Aves, Galloanseres). R. Soc. Open Sci. 4, 170975 (2017).
Tambussi, C. P., Degrange, F. J., De Mendoza, R. S., Sferco, E. & Santillana, S. A stem anseriform from the early Palaeocene of Antarctica provides new key evidence in the early evolution of waterfowl. Zool. J. Linn. Soc. 186, 673–700 (2019).
Mayr, G., De Pietri, V. L., Love, L., Mannering, A. & Scofield, R. P. Oldest, smallest and phylogenetically most basal pelagornithid, from the early Paleocene of New Zealand, sheds light on the evolutionary history of the largest flying birds. Pap. Palaeontol. https://doi.org/10.1002/spp2.1284 (2019).
Budd, G. E. & Mann, R. P. The dynamics of stem and crown groups. Sci. Adv. 6, eaaz1626 (2020).
Ksepka, D. T. & Clarke, J. Phylogenetically vetted and stratigraphically constrained fossil calibrations within Aves. Palaeontologia Electronica 18, 18.1.3FC (2015).
Mayr, G., De Pietri, V. L., Scofield, R. P. & Worthy, T. H. On the taxonomic composition and phylogenetic affinities of the recently proposed clade Vegaviidae Agnolín et al., 2017 – neornithine birds from the Upper Cretaceous of the Southern Hemisphere. Cretaceous Research 86, 178–185 (2018).
Clarke, J. A. et al. Fossil evidence of the avian vocal organ from the Mesozoic. Nature 538, 502–505 (2016).
Agnolín, F. L., Egli, F. B., Chatterjee, S., Marsà, J. A. G. & Novas, F. E. Vegaviidae, a new clade of southern diving birds that survived the K/T boundary. Naturwissenschaften 104, 87 (2017).
O’Connor, J. K., Chiappe, L. M. & Bell, A. in Living Dinosaurs: The Evolutionary History of Modern Birds (eds Dyke, G. & Kaiser, G.) 39–114 (Wiley-Blackwell, 2011).
Cracraft, J. in The Phylogeny and Classification of the Tetrapods Vol. 1 (ed. Benton, M. J.) 339–361 (Oxford Univ. Press, 1988).
Livezey, B. C. A phylogenetic analysis of basal Anseriformes, the fossil Presbyornis, and the interordinal relationships of waterfowl. Zool. J. Linn. Soc. 121, 361–428 (1997).
Cracraft, J. & Clarke, J. The basal clades of modern birds. In New Perspectives on the Origin and Early Evolution of Birds: Proceedings of the International Symposium in Honor of John H. Ostrom (eds Gauthier, J. & Gall, L. F.) 143–156 (Peabody Museum of Natural History, Yale University, 2001).
Felice, R. N. & Goswami, A. Developmental origins of mosaic evolution in the avian cranium. Proc. Natl Acad. Sci. USA 115, 555–560 (2018).
Field, D. J. Endless skulls most beautiful. Proc. Natl Acad. Sci. USA 115, 448–450 (2018).
Huxley, T. H. On the classification of birds; and on the taxonomic value of the modifications of certain of the cranial bones observable in that class. Proc. Zool. Soc. Lond. 1867, 415–472 (1867).
Ericson, P. G. P. Systematic relationships of the Palaeogene family Presbyornithidae (Aves: Anseriformes). Zool. J. Linn. Soc. 121, 429–483 (1997).
Cooney, C. R. et al. Mega-evolutionary dynamics of the adaptive radiation of birds. Nature 542, 344–347 (2017).
Bright, J. A., Marugán-Lobón, J., Rayfield, E. J. & Cobb, S. N. The multifactorial nature of beak and skull shape evolution in parrots and cockatoos (Psittaciformes). BMC Evol. Biol. 19, 104 (2019).
Field, D. J. & Hsiang, A. Y. A North American stem turaco, and the complex biogeographic history of modern birds. BMC Evol. Biol. 18, 102 (2018).
Mourer-Chauviré, C. Les oiseaux fossiles des phosphorites du Quercy (Éocène supérieur a Oligocène supérieur): implications paléobiogéographiques. Geobios 15, 413–426 (1982).
Mayr, G. Two-phase extinction of “Southern Hemispheric” birds in the Cenozoic of Europe and the origin of the Neotropic avifauna. Palaeobiodivers. Palaeoenviron. 91, 325–333 (2011).
O’Connor, J. K. & Zhou, Z. The evolution of the modern avian digestive system: insights from paravian fossils from the Yanliao and Jehol biotas. Palaeontology 63, 13–27 (2020).
Feduccia, A. Explosive evolution in tertiary birds and mammals. Science 267, 637–638 (1995).
Clarke, J. A. Morphology, phylogenetic taxonomy, and systematics of Ichthyornis and Apatornis (Avialae: Ornithurae). Bull. Am. Mus. Nat. Hist. 286, 1–179 (2004).
Field, D. J. et al. Complete Ichthyornis skull illuminates mosaic assembly of the avian head. Nature 557, 96–100 (2018).
Mayr, G. & Weidig, I. The early Eocene bird Gallinuloides wyomingensis – a stem group representative of Galliformes. Acta Palaeontol. Pol. 49, 211–217 (2004).
Ksepka, D. T. Broken gears in the avian molecular clock: new phylogenetic analyses support stem galliform status for Gallinuloides wyomingensis and rallid affinities for Amitabha urbsinterdictensis. Cladistics 25, 173–197 (2009).
Mayr, G. & Rubilar-Rogers, D. Osteology of a new giant bony-toothed bird from the Miocene of Chile, with a revision of the taxonomy of Neogene Pelagornithidae. J. Vertebr. Paleontol. 30, 1313–1330 (2010).
Bourdon, E. Osteological evidence for sister group relationship between pseudo-toothed birds (Aves: Odontopterygiformes) and waterfowls (Anseriformes). Naturwissenschaften 92, 586–591 (2005).
Mayr, G. Cenozoic mystery birds - on the phylogenetic affinities of bony-toothed birds (Pelagornithidae). Zool. Scr. 40, 448–467 (2011).
Goloboff, P. A. & Catalano, S. A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32, 221–238 (2016).
Ronquist, F. et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Miller, M. A., Pfeiffer, W. & Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Gateway Computing Environments Workshop (GCE 2010) 45–53 (IEEE, 2010).
Lewis, P. O. A likelihood approach to estimating phylogeny from discrete morphological character data. Syst. Biol. 50, 913–925 (2001).
Heath, T. A., Huelsenbeck, J. P. & Stadler, T. The fossilized birth–death process for coherent calibration of divergence-time estimates. Proc. Natl Acad. Sci. USA 111, E2957–E2966 (2014).
Zhang, C., Stadler, T., Klopfstein, S., Heath, T. A. & Ronquist, F. Total-evidence dating under the fossilized birth–death process. Syst. Biol. 65, 228–249 (2016).
Kealy, S. & Beck, R. Total evidence phylogeny and evolutionary timescale for Australian faunivorous marsupials (Dasyuromorphia). BMC Evol. Biol. 17, 240 (2017).
Vinther, J., Parry, L., Briggs, D. E. & Van Roy, P. Ancestral morphology of crown-group molluscs revealed by a new Ordovician stem aculiferan. Nature 542, 471–474 (2017).
Gill, F., Donsker, D & Rasmussen, P. (eds) IOC World Bird List (v.10.1) https://www.worldbirdnames.org/ioc-lists/crossref/ (2020).
Field, D. J., LeBlanc, A., Gau, A. & Behlke, A. D. B. Pelagic neonatal fossils support viviparity and precocial life history of Cretaceous mosasaurs. Palaeontology 58, 401–407 (2015).
Ericson, P. G. P. et al. Diversification of Neoaves: integration of molecular sequence data and fossils. Biol. Lett. 2, 543–547 (2006).
Jetz, W., Thomas, G. H., Joy, J. B., Hartmann, K. & Mooers, A. O. The global diversity of birds in space and time. Nature 491, 444–448 (2012).
Phillips, M. J. Geomolecular dating and the origin of placental mammals. Syst. Biol. 65, 546–557 (2016).
He, H. Y. et al. Timing of the Jiufotang Formation (Jehol Group) in Liaoning, northeastern China, and its implications. Geophys. Res. Lett. 31, (2004).
Wang, X. et al. The earliest evidence for a supraorbital salt gland in dinosaurs in new Early Cretaceous ornithurines. Sci. Rep. 8, 3969 (2018).
Musser, G., Ksepka, D. T. & Field, D. J. New material of Paleocene-Eocene Pellornis (Aves: Gruiformes) clarifies the pattern and timing of the extant Gruiform radiation. Diversity 11, 102 (2019).
Ksepka, D. T., Stidham, T. A. & Williamson, T. E. Early Paleocene landbird supports rapid phylogenetic and morphological diversification of crown birds after the K–Pg mass extinction. Proc. Natl Acad. Sci. USA 114, 8047–8052 (2017).
Parham, J. F. et al. Best practices for justifying fossil calibrations. Syst. Biol. 61, 346–359 (2012).
Püschel, H. P., O'Reilly, J. E., Pisani, D. & Donoghue, P. C. J. The impact of fossil stratigraphic ranges on tip-calibration, and the accuracy and precision of divergence time estimates. Palaeontology 63, 67–83 (2020).
Worthy, T. H. et al. Osteology supports a stem-galliform affinity for the giant extinct flightless bird Sylviornis neocaledoniae (Sylviornithidae, Galloanseres). PLoS One 11, e0150871 (2016).
Benton, M. J. & Donoghue, P. C. J. Paleontological evidence to date the tree of life. Mol. Biol. Evol. 24, 26–53 (2007).
Reddy, S. et al. Why do phylogenomic data sets yield conflicting trees? Data type influences the avian tree of life more than taxon sampling. Syst. Biol. 66, 857–879 (2017).
Hackett, S. J. et al. A phylogenomic study of birds reveals their evolutionary history. Science 320, 1763–1768 (2008).
Kimball, R. T. et al. A phylogenomic supertree of birds. Diversity 11, 109 (2019).
Dunning, J. B. CRC Handbook of Avian Body Masses 2nd edn (CRC Press, 2007).
Acknowledgements
We thank M. van Dinther for collecting and donating the specimen; J. Vellekoop for advice on geochronology; K. Smithson, T. Thompson and V. Fernandez for scanning support; M. Brooke, M. Lowe, M. Clementz, L. Vietti, J. Cooper, J. White, C. Levitt, R. Irmis, K. MacKenzie and J. Sertich for collections assistance; B. Creisler for etymological information; and T. Worthy and J. Watanabe for anatomical advice. We are grateful to L. Witmer and F. Degrange for sharing three-dimensional models of Presbyornis and Conflicto, respectively, and to P. Krzeminski for his artwork. D.J.F. acknowledges support from the UK Research and Innovation Future Leaders Fellowship MR/S032177/1, the Royal Society Research Grant RGS/R2/192390, a Systematics Association Research Grant and the Isaac Newton Trust; J.B. acknowledges the Hesse Award from the American Ornithological Society and grants from the Jurassic Foundation, Geological Association and Paleontological Society; and D.T.K. acknowledges support from the NSF award DEB 1655736.
Author information
Authors and Affiliations
Contributions
J.W.M.J. provided the material and stratigraphic data; D.J.F. prepared the specimens; D.J.F. and J.B. acquired CT scans and discovered the skull; D.J.F., J.B. and A.C. performed digital segmentation of the material and created figures; D.J.F., J.B., A.C. and D.T.K. performed anatomical comparisons; J.B. and A.C. performed the phylogenetic analyses; and D.J.F. wrote the paper, with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Jingmai O’Connor and Kevin Padian for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Detailed cranial and mandibular anatomy of A. maastrichtensis (NHMM 2013 008).
The views of the cranium are similar to those in Fig. 1, but with the jaws removed to illustrate the ventral portion of the skull.
Extended Data Fig. 2 Higher-resolution cranial and mandibular anatomy of A. maastrichtensis (NHMM 2013 008).
Labels have been removed and images enlarged to show details.
Extended Data Fig. 3 Morphology of individually segmented skull elements from A. maastrichtensis (NHMM 2013 008).
Dorsal, ventral and rostral views of the frontals show the nasals separated from their in situ position to illustrate the morphology of the nasofrontal contact. Scale bars, 1 cm.
Extended Data Fig. 4 Detailed comparisons of galloanseran quadrate morphology.
Skulls and quadrates of extant Galliformes and total-group Anseriformes. The skull of Presbyornis USNM 299846 is shown. Scale bars, 5 mm (quadrates); 1 cm (skulls). Skulls are in left lateral view except Presbyornis, which is in reflected right lateral view. FPB, foramen pneumaticum basiorbitale; FPR, foramen pneumaticum rostromediale.
Extended Data Fig. 5 Detailed comparisons of galloanseran retroarticular morphology.
Retroarticular regions of left mandibles in lateral (left), medial (middle) and dorsal (right) views. Both the left and right mandibles of Asteriornis are shown in dorsal view, as the retroarticular process is only preserved on the left mandible and the medial process is only preserved on the right mandible. Images of Anatalavis are mirrored. Scale bars, 1 cm.
Extended Data Fig. 6 Postcranial morphology of A. maastrichtensis (NHMM 2013 008).
The left distal femur of Presbyornis pervetus (UW 27596) is shown for comparison.
Extended Data Fig. 7 Internal composition of NHMM 2013 008 blocks.
a, Block containing the left femur, left tibiotarsus and the main portion of the skull, viewed from the side with the femur exposed. b, Same block as in a, viewed from the side containing the tibiotarsus. c, Block containing the right femur and tarsometatarsus. d, Block containing the right distal radius, several unidentified bone fragments and a portion of the cranial roof near the frontoparietal suture. e, Block containing the right tibiotarsus. Numerous fragments of fossil echinoderms and molluscs are visible within the blocks. Scale bars, 1 cm.
Extended Data Fig. 8 Relative body size of A. maastrichtensis.
Estimate of the mean body size of the Asteriornis holotype25 compared with extant Galloanserae82, ranked on the x axis from smallest to largest. The mean body-size estimate for Asteriornis (394 g) is closest to that of male Anas crecca (392 g; 7.8th percentile among Anseriformes) and female Perdix perdix (393 g; 33rd percentile among Galliformes).
Extended Data Fig. 9 Expanded phylogenetic results.
a, Results of the parsimony analysis. Asteriornis (pink) resolves as the sister taxon to crown Galloanserae. b, Results of the tip-dated Bayesian analysis with a soft-maximum neornithine root age of 86.5 million years. An estimated timescale is shown on the x axis, although see caveats relating to divergence times in the Supplementary Information. Asteriornis (pink) resolves as the stemward-most member of Pangalliformes. Colours match those in Fig. 3 and extinct taxa are denoted with daggers. See Supplementary Information for full details of phylogenetic analyses, character information, synapomorphies of key clades, support values and tree files.
Supplementary information
Supplementary Information
This file contains: Museum Abbreviations, Scan Parameters, Supplementary Video Descriptions, Supplementary Methods (Phylogenetic Analyses), and Supplementary Notes (Provenance Data for New Fossil Material, Phylogenetic Definitions of Clade Names, Additional Anatomical Observations, Synapomorphies Diagnosing Key Clades, Morphological Character Descriptions, Supplementary References) – see Contents page for details.
Supplementary Data
Character-taxon matrix: Nexus file used for the phylogenetic analyses.
Video 1
NHMM 2013 008, Skull of Asteriornis maastrichtensis holotype, yaw video.
Video 2
NHMM 2013 008, Skull of Asteriornis maastrichtensis holotype, roll video.
Rights and permissions
About this article
Cite this article
Field, D.J., Benito, J., Chen, A. et al. Late Cretaceous neornithine from Europe illuminates the origins of crown birds. Nature 579, 397–401 (2020). https://doi.org/10.1038/s41586-020-2096-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2096-0
This article is cited by
-
A juvenile bird with possible crown-group affinities from a dinosaur-rich Cretaceous ecosystem in North America
BMC Ecology and Evolution (2024)
-
Cretaceous ornithurine supports a neognathous crown bird ancestor
Nature (2022)
-
The Transversal Heritage of Maastricht Stone, a Potential Global Heritage Stone Resource from Belgium and the Netherlands
Geoheritage (2022)
-
Evolution and development of the bird chondrocranium
Frontiers in Zoology (2021)
-
‘Dinosaur-bird’ macroevolution, locomotor modules and the origins of flight
Journal of Iberian Geology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.