Abstract
Empirical and anecdotal evidence has associated stress with accelerated hair greying (formation of unpigmented hairs)1,2, but so far there has been little scientific validation of this link. Here we report that, in mice, acute stress leads to hair greying through the fast depletion of melanocyte stem cells. Using a combination of adrenalectomy, denervation, chemogenetics3,4, cell ablation and knockout of the adrenergic receptor specifically in melanocyte stem cells, we find that the stress-induced loss of melanocyte stem cells is independent of immune attack or adrenal stress hormones. Instead, hair greying results from activation of the sympathetic nerves that innervate the melanocyte stem-cell niche. Under conditions of stress, the activation of these sympathetic nerves leads to burst release of the neurotransmitter noradrenaline (also known as norepinephrine). This causes quiescent melanocyte stem cells to proliferate rapidly, and is followed by their differentiation, migration and permanent depletion from the niche. Transient suppression of the proliferation of melanocyte stem cells prevents stress-induced hair greying. Our study demonstrates that neuronal activity that is induced by acute stress can drive a rapid and permanent loss of somatic stem cells, and illustrates an example in which the maintenance of somatic stem cells is directly influenced by the overall physiological state of the organism.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The sequencing data that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) with the accession code GSE131566. Source data for all main figures and Extended Data figures are provided with the paper.
References
Ephraim, A. J. On sudden or rapid whitening of the hair. AMA Arch. Derm. 79, 228–236 (1959).
Navarini, A. A., Nobbe, S. & Trüeb, R. M. Marie Antoinette syndrome. Arch. Dermatol. 145, 656 (2009).
Alexander, G. M. et al. Remote control of neuronal activity in transgenic mice expressing evolved G protein-coupled receptors. Neuron 63, 27–39 (2009).
Zhu, H. et al. Cre-dependent DREADD (designer receptors exclusively activated by designer drugs) mice. Genesis 54, 439–446 (2016).
Müller-Röver, S. et al. A comprehensive guide for the accurate classification of murine hair follicles in distinct hair cycle stages. J. Invest. Dermatol. 117, 3–15 (2001).
Hsu, Y.-C., Li, L. & Fuchs, E. Emerging interactions between skin stem cells and their niches. Nat. Med. 20, 847–856 (2014).
Chang, C.-Y. et al. NFIB is a governor of epithelial-melanocyte stem cell behaviour in a shared niche. Nature 495, 98–102 (2013).
Rabbani, P. et al. Coordinated activation of Wnt in epithelial and melanocyte stem cells initiates pigmented hair regeneration. Cell 145, 941–955 (2011).
Nishimura, E. K. et al. Dominant role of the niche in melanocyte stem-cell fate determination. Nature 416, 854–860 (2002).
Nishimura, E. K., Granter, S. R. & Fisher, D. E. Mechanisms of hair graying: incomplete melanocyte stem cell maintenance in the niche. Science 307, 720–724 (2005).
Anthony, T. E. et al. Control of stress-induced persistent anxiety by an extra-amygdala septohypothalamic circuit. Cell 156, 522–536 (2014).
Ramirez, S. et al. Activating positive memory engrams suppresses depression-like behaviour. Nature 522, 335–339 (2015).
Heidt, T. et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 20, 754–758 (2014).
Tye, K. M. et al. Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature 493, 537–541 (2013).
Acs, G., Biro, T., Acs, P., Modarres, S. & Blumberg, P. M. Differential activation and desensitization of sensory neurons by resiniferatoxin. J. Neurosci. 17, 5622–5628 (1997).
Baral, P. et al. Nociceptor sensory neurons suppress neutrophil and γδ T cell responses in bacterial lung infections and lethal pneumonia. Nat. Med. 24, 417–426 (2018).
Ulrich-Lai, Y. M. & Herman, J. P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 10, 397–409 (2009).
Caterina, M. J. et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824 (1997).
Kondo, T. & Hearing, V. J. Update on the regulation of mammalian melanocyte function and skin pigmentation. Expert. Rev. Dermatol. 6, 97–108 (2011).
Steingrímsson, E., Copeland, N. G. & Jenkins, N. A. Melanocyte stem cell maintenance and hair graying. Cell 121, 9–12 (2005).
Liao, C.-P., Booker, R. C., Morrison, S. J. & Le, L. Q. Identification of hair shaft progenitors that create a niche for hair pigmentation. Genes Dev. 31, 744–756 (2017).
Inomata, K. et al. Genotoxic stress abrogates renewal of melanocyte stem cells by triggering their differentiation. Cell 137, 1088–1099 (2009).
Harris, M. L. et al. A direct link between MITF, innate immunity, and hair graying. PLoS Biol. 16, e2003648 (2018).
Bosenberg, M. et al. Characterization of melanocyte-specific inducible Cre recombinase transgenic mice. Genesis 44, 262–267 (2006).
Köhler, C. et al. Mouse cutaneous melanoma induced by mutant BRaf arises from expansion and dedifferentiation of mature pigmented melanocytes. Cell Stem Cell 21, 679–693 (2017).
Moon, H. et al. Melanocyte stem cell activation and translocation initiate cutaneous melanoma in response to UV exposure. Cell Stem Cell 21, 665–678 (2017).
Sheng, M. & Greenberg, M. E. The regulation and function of c-fos and other immediate early genes in the nervous system. Neuron 4, 477–485 (1990).
Kostrzewa, R. M. & Jacobowitz, D. M. Pharmacological actions of 6-hydroxydopamine. Pharmacol. Rev. 26, 199–288 (1974).
Boullin, D. J., Costa, E. & Brodie, B. B. Discharge of tritium-labeled guanethidine by sympathetic nerve stimulation as evidence that guanethidine is a false transmitter. Life Sci. 5, 803–808 (1966).
Acar, M. et al. Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature 526, 126–130 (2015).
Lay, K., Kume, T. & Fuchs, E. FOXC1 maintains the hair follicle stem cell niche and governs stem cell quiescence to preserve long-term tissue-regenerating potential. Proc. Natl Acad. Sci. USA 113, E1506–E1515 (2016).
Wang, L., Siegenthaler, J. A., Dowell, R. D. & Yi, R. Foxc1 reinforces quiescence in self-renewing hair follicle stem cells. Science 351, 613–617 (2016).
Cho, I. J. et al. Mechanisms, hallmarks, and implications of stem cell quiescence. Stem Cell Reports 12, 1190–1200 (2019).
Nishimura, E. K. et al. Key roles for transforming growth factor β in melanocyte stem cell maintenance. Cell Stem Cell 6, 130–140 (2010).
Takeo, M. et al. EdnrB governs regenerative response of melanocyte stem cells by crosstalk with Wnt signaling. Cell Rep. 15, 1291–1302 (2016).
Tirosh, I. et al. Dissecting the multicellular ecosystem of metastatic melanoma by single-cell RNA-seq. Science 352, 189–196 (2016).
Peters, E. M. J., Tobin, D. J., Botchkareva, N., Maurer, M. & Paus, R. Migration of melanoblasts into the developing murine hair follicle is accompanied by transient c-Kit expression. J. Histochem. Cytochem. 50, 751–766 (2002).
Chou, W. C. et al. Direct migration of follicular melanocyte stem cells to the epidermis after wounding or UVB irradiation is dependent on Mc1r signaling. Nat. Med. 19, 924–929 (2013).
Losiewicz, M. D., Carlson, B. A., Kaur, G., Sausville, E. A. & Worland, P. J. Potent inhibition of CDC2 kinase activity by the flavonoid L86-8275. Biochem. Biophys. Res. Commun. 201, 589–595 (1994).
Wyatt, P. G. et al. Identification of N-(4-piperidinyl)-4-(2,6-dichlorobenzoylamino)-1H-pyrazole-3-carboxamide (AT7519), a novel cyclin dependent kinase inhibitor using fragment-based X-ray crystallography and structure based drug design. J. Med. Chem. 51, 4986–4999 (2008).
Borden, P., Houtz, J., Leach, S. D. & Kuruvilla, R. Sympathetic innervation during development is necessary for pancreatic islet architecture and functional maturation. Cell Rep. 4, 287–301 (2013).
Zeng, X. et al. Innervation of thermogenic adipose tissue via a calsyntenin 3β-S100b axis. Nature 569, 229–235 (2019).
Katayama, Y. et al. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 124, 407–421 (2006).
Reed, C. M. The ultrastructure and innervation of muscles controlling chromatophore expansion in the squid, Loligo vulgaris. Cell Tissue Res. 282, 503–512 (1995).
Fan, S. M.-Y. et al. External light activates hair follicle stem cells through eyes via an ipRGC–SCN-sympathetic neural pathway. Proc. Natl Acad. Sci. USA 115, E6880–E6889 (2018).
Lerner, A. B. Gray hair and sympathectomy. Report of a case. Arch. Dermatol. 93, 235–236 (1966).
Ortonne, J. P., Thivolet, J. & Guillet, R. Graying of hair with age and sympathectomy. Arch. Dermatol. 118, 876–877 (1982).
Hinoi, E. et al. The sympathetic tone mediates leptin’s inhibition of insulin secretion by modulating osteocalcin bioactivity. J. Cell Biol. 183, 1235–1242 (2008).
Abraira, V. E. et al. The cellular and synaptic architecture of the mechanosensory dorsal horn. Cell 168, 295–310 (2017).
Pruitt, S. C., Freeland, A., Rusiniak, M. E., Kunnev, D. & Cady, G. K. Cdkn1b overexpression in adult mice alters the balance between genome and tissue ageing. Nat. Commun. 4, 2626 (2013).
Szallasi, A. & Blumberg, P. M. Resiniferatoxin, a phorbol-related diterpene, acts as an ultrapotent analog of capsaicin, the irritant constituent in red pepper. Neuroscience 30, 515–520 (1989).
Riol-Blanco, L. et al. Nociceptive sensory neurons drive interleukin-23-mediated psoriasiform skin inflammation. Nature 510, 157–161 (2014).
Kashem, S. W. et al. Nociceptive sensory fibers drive interleukin-23 production from CD301b+ dermal dendritic cells and drive protective cutaneous immunity. Immunity 43, 515–526 (2015).
Marshall, I. C. B. et al. Activation of vanilloid receptor 1 by resiniferatoxin mobilizes calcium from inositol 1,4,5-trisphosphate-sensitive stores. Br. J. Pharmacol. 138, 172–176 (2003).
Neubert, J. K. et al. Peripherally induced resiniferatoxin analgesia. Pain 104, 219–228 (2003).
Watanabe, T., Sakurada, N. & Kobata, K. Capsaicin-, resiniferatoxin-, and olvanil-induced adrenaline secretions in rats via the vanilloid receptor. Biosci. Biotechnol. Biochem. 65, 2443–2447 (2001).
Zhang, B., He, M. & Hsu, Y.-C. FACS isolation of melanocyte stem cells from mouse skin. Protoc. Exch. https://doi.org/10.21203/rs.2.17987/v1 (2019).
Gilchrest, B. A., Vrabel, M. A., Flynn, E. & Szabo, G. Selective cultivation of human melanocytes from newborn and adult epidermis. J. Invest. Dermatol. 83, 370–376 (1984).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Huang, W., Sherman, B. T. & Lempicki, R. A. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 37, 1–13 (2009).
Huang, W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protocols 4, 44–57 (2009).
Acknowledgements
We thank G. Karsenty for Adrb2fl/fl mice, D. Ginty for THcreERT2 mice and many colleagues who donated mice to the Jackson Laboratory; A. Wagers, W. Anderson, C.-Y. Chang, Y. Fong, Q. Ma, M. Nahrendorf, A. Sahay and members of the Y.-C.H. laboratory, in particular M. Gonzalez-Celeiro, for discussions and comments on the manuscript; S. Kim, Y.-L. Kang and O. Chung for technical assistance; and HCBI, the HSCRB FACS core and histology core, FAS Small Molecule Mass Spectrometry Facility, Office of Animal Resources and the Bauer Core Facility at Harvard University for technical support. This work was supported in part by the Harvard Stem Cell Institute; Harvard NeuroDiscovery Center; Harvard Medical School Dean’s Innovation Grant; Smith Family Foundation Odyssey Award; American Cancer Society (RSG-18-152-01-DDC); NIH (R01-AR070825 to Y.-C.H., R01 AR043369-23, R01CA222871, R01AR072304 and P01 CA163222 to D.E.F., R01CA103846 and P01CA163222 to L.I.Z. and DP2AT009499 and R01AI130019 to I.M.C.); and grants from the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation to D.E.F. and from Klarman Cell Observatory to A.R. Y.-C.H. is a Pew Scholar and a NYSCF – Robertson Investigator; A.R. and L.I.Z. are HHMI investigators; B.Z. is a recipient of the Charles A. King Trust Postdoctoral Research Fellowship; M.H. is a recipient of the NSF Graduate Research Fellowships Program (DGE1745303) and was supported by the Joint Program in Molecules, Cells, and Organisms (5T32GM007598-40); Y. Shwartz is a Helen Hay Whitney postdoctoral fellow and a recipient of the Woman in Science Weizmann Institute of Science Award; T.M.C. had a CAPES/HARVARD fellowship for visiting professor from CAPES (Process no. 88881.162285/2017-01) and is supported by a FAPESP grant (2013/08216-2); E.M.F. was supported by a Leukemia & Lymphoma Society Scholar grant (5372-15); and J.D.B. and S.M. acknowledge support from the Broad Institute Fellows Program.
Author information
Authors and Affiliations
Contributions
B.Z. and Y.-C.H. conceived the project. B.Z. performed most of the experiments. S.M. performed bioinformatic analysis. I.R. and Y. Su performed the human melanocyte experiments. M.H. performed the immunostaining of sympathetic ganglia and quantifications of sympathetic nerve density. P.B. and I.M.C. made the initial observations of hair greying in RTX-injected mice. S.C. performed experiments related to chronic unpredictable stress and corticosterone feeding. Y. Shwartz performed the sympathetic nerve ablation experiments. W.A.G. and T.M.C. performed the guanethidine experiments. E.M.F. performed the radiation experiments. Y.-C.H., D.E.F., I.M.C., T.M.C., J.D.B., A.R. and L.I.Z. provided intellectual input and helped shape the research. B.Z. and Y-C.H. wrote the manuscript, with discussions and feedback from all co-authors.
Corresponding author
Ethics declarations
Competing interests
L.I.Z. is a founder and stockholder of Fate Therapeutics, Scholar Rock and CAMP4 Therapeutics. D.E.F. has a financial interest in Soltego, a company developing salt-inducible kinase inhibitors for topical skin-darkening treatments that might be used for a broad set of human applications. The interests of D.E.F. were reviewed and are managed by Massachusetts General Hospital and Partners HealthCare in accordance with their conflict of interest policies. A.R. is a member of the Scientific Advisory Board (SAB) of Thermo Fisher Scientific, Neogene Therapeutics, Asimov and Syros Pharmaceuticals; an equity holder of Immunitas; and a founder and an equity holder of Celsius Therapeutics. I.M.C. is an SAB member of GSK Pharmaceuticals and Kintai Therapeutics. A provisional patent application has been filed based on this work (applicants: President and Fellows of Harvard College and The General Hospital Corporation; inventors: Y.-C.H., B.Z., D.E.F. and I.R.; application number: 62/903,517; status: pending/provisional; aspect covered: methods and compositions for controlling hair greying). All of the other authors declare no competing interests.
Additional information
Peer review information Nature thanks Salvador Aznar Benitah, Christopher Deppmann, William Lowry and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Effects of stress on the hair pigmentation.
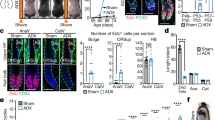
a, Schematic of MeSC behaviour during the hair cycle. b, Hair greying after mice were subjected to chronic unpredictable stress. Quantifications were done by plucking around 100 hairs from different regions across the skin and counting the number of white hairs (n = 9 plucked regions from 3 mice for each condition, two-tailed unpaired t-test). c, Hair greying after mice were subjected to restraint stress. Quantifications as described in b. d, LC–MS/MS quantification of corticosterone and noradrenaline after restraint stress (n = 5 mice for control and n = 6 mice for restraint, two-tailed unpaired t-test). e, Left, immunofluorescent staining of hair bulbs for melanocyte-inducing transcription factor (MITF, red) from mice five days after treatment with saline or RTX. Right, quantification of MITF+ cells (n = 30 hair follicles from 3 mice for each condition, two-tailed unpaired t-test). f, Fontana-Masson staining of hair bulbs for melanin from mice five days after treatment with saline or RTX (n = 6 mice for each condition). g, Coat colour in mice five days after RTX injection in anagen. RTX was injected in full anagen and the mice were examined five days later, at late anagen. The coat colour remained black (n = 6 mice for each condition). h, Fontana-Masson staining of hair follicles for melanin from mice that were treated with saline or RTX at first anagen and examined at second anagen (see Fig. 1e for corresponding fluorescent images) (n = 6 mice for each condition). i, Quantification of the number of MeSCs in the skin of mice injected with saline or RTX. For the RTX-injected mice, the numbers of MeSCs in regions with predominantly black hairs and regions with many white hairs are quantified separately. Orange and green dashed boxes denote representative black and white hair regions, respectively, in RTX-injected mice. Enlarged boxes show representative immunofluorescent images of hair follicles from each region. White arrowheads indicate regions where MeSCs reside (n = 30 hair follicles from 3 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). j, Quantification of the body area covered by white hairs in female versus male mice (n = 5 mice for each sex, two-tailed unpaired t-test). All data are mean ± s.d.
Extended Data Fig. 2 Loss of MeSCs in three different models of stress.
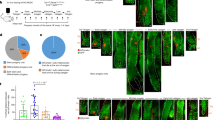
a, Top, schematic of experimental design for RTX injection in first telogen (red arrows indicate collection of skin samples). Bottom left, representative mouse images 5 days and 16 days after RTX injection in first telogen. Bottom right, quantification of the body area covered by white hairs 16 days after RTX injection (n = 4 mice for each condition, two-tailed unpaired t-test). b, Immunofluorescent staining for TRP2 from mice injected with saline or RTX in first telogen (n = 30 hair follicles from 4 mice for each condition, two-tailed unpaired t-test). Yellow boxes denote the upper region of the hair follicle, where MeSCs reside. Enlarged views of the yellow-boxed regions are shown to the right. White arrowheads indicate MeSCs. Quantifications of MeSCs are also shown (bottom). c, Immunofluorescent staining for TRP2 (red) (left) and quantification of MeSCs (right) from mice subjected to chronic unpredictable stress or restraint stress (n = 30 hair follicles from 5 mice for each condition, two-tailed unpaired t-test). d, Hair coat colour was monitored in RTX-injected mice for multiple rounds of hair-follicle regeneration (waxing was used to initiate new rounds of anagen) (n = 3 mice for each condition). Schematic denotes the experimental design. Scale bars, 50 μm. All data are mean ± s.d.
Extended Data Fig. 3 Stress-induced hair greying is not mediated by corticosterone or immune attack.
a, Left, formation of white hairs after RTX injection in Rag1 mutant mice that are devoid of T and B cells (Rag1 KO) (n = 6 for each condition, two-tailed unpaired t-test). Right, immunofluorescent staining for the T cell marker CD3 (green) in control and Rag1 KO skin (n = 6 mice for each condition, two-tailed unpaired t-test). b, Left, hair greying occurs when RTX is injected into CD11b-DTR mice that were treated with diphtheria toxin to deplete myeloid cells (n = 6 mice for each condition). Right, immunofluorescent staining for CD11B (green) in skin from control and CD11b-DTR mice that were treated with diphtheria toxin (n = 6 mice for each condition). c, Expression of adrenergic receptors and glucocorticoid receptor (GR) in MeSCs (n = 2 biologically independent samples). d, Formation of white hairs after RTX injection into TyrcreERT2;GR fl/fl mice (MeSC-GR cKO) (n = 6 mice for each condition, two-tailed unpaired t-test). e, Left, enzyme-linked immunosorbent assay (ELISA) measurement of the level of corticosterone in the blood three days after supplying corticosterone in the drinking water (n = 4 mice for each condition). Middle, immunofluorescent staining of hair follicles for TRP2 (red) from mice five days after treatment with corticosterone (n = 30 hair follicles from 3 mice for each condition, two-tailed unpaired t-test). Right, coat colour after hair follicles in corticosterone-treated mice enter another round of anagen to regenerate new hairs. Scale bars, 50 μm. All data are mean ± s.d.
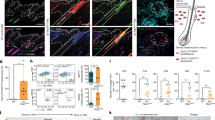
Extended Data Fig. 4 Perturbations of the noradrenaline–ADRB2 pathway.
a, Left, immunofluorescent staining of hair follicles for phosphorylated CREB (pCREB; green) and TRP2 (red) 12 h after RTX injection. Right, quantification of the percentage of pCREB+ MeSCs (n = 30 hair follicles from 3 mice for each condition, two-tailed unpaired t-test). White arrowheads indicate pCREB+ MeSCs in upper hair follicles after RTX injection. b, Formation of white hairs after RTX injection into K15crePGR;Adrb2fl/fl mice (HFSC-Adrb2 cKO) (n = 3 mice for each condition, two-tailed unpaired t-test). c, Top left, coat colour in unstressed TyrcreERT2;Adrb2fl/fl mice (MeSC-Adrb2 cKO) in the second telogen after treatment with tamoxifen (seven times) at the first telogen. Bottom left, immunofluorescent staining of hair bulbs for MITF (red) in TyrcreERT2;Adrb2fl/fl mice in anagen. Right, Fontana-Masson melanin staining of anagen hair follicles from TyrcreERT2;Adrb2fl/fl mice (n = 3 mice for each condition). d, Top left, schematic of experimental design for mosaic labelling in unstressed control and Adrb2 knockout mice (red arrows indicate collection of skin samples). Bottom left, immunofluorescent staining for GFP (green) and TRP2 (red) from TyrcreERT2;RosamT/mG mice (MeSC-mT/mG) and TyrcreERT2;Adrb2fl/fl;RosamT/mG mice (MeSC-Adrb2 cKO-mT/mG) after treatment with tamoxifen (three times) at first telogen. Right, immunofluorescent staining of hair follicles for GFP (green) and TRP2 (red) after the mice enter anagen (n = 3 mice for each condition). e, Quantification of the percentage of white hairs after intradermal injection of saline or noradrenaline (n = 10 injected sites from 6–8 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). f, Left, immunofluorescent staining of hair follicles for TRP2 (red) from the skins of mice that were intradermally injected with noradrenaline (n = 30 hair follicles from 10 injection sites for each condition, one-way ANOVA with Tukey’s multiple comparisons test). Right, quantification of MeSCs. g, White hairs are formed after intradermal injection of noradrenaline in K15crePGR;Adrb2fl/fl mice (HF-Adrb2 cKO) (n = 3 injection sites for each condition, two-tailed unpaired t-test). Yellow dashed circles denote intradermal injection sites. Scale bars, 50 μm. All data are mean ± s.d.
Extended Data Fig. 5 Activation of the sympathetic nervous system by nociception-induced stress or sympathetic-nerve-specific inducible Gq-DREADD.
a, LC–MS/MS quantification of stress hormones in sham-operated and adrenalectomized mice (n = 3 mice for each condition, two-way ANOVA with Benjamini–Hochberg correction). b, Top, immunofluorescent staining of sympathetic nerves in the skin regions with predominantly black hairs (orange box) and with mostly white hairs (green box) (n = 3 mice for each condition). Bottom, 3D surfaces of tyrosine hydroxylase (TH) staining, created using Imaris software, and quantification of the sympathetic nerve volume from regions with different numbers of unpigmented hairs (n = 20 hair follicles for each region from 3 mice, two-tailed unpaired t-test). c, Left, immunofluorescent staining of sympathetic ganglia for tyrosine hydroxylase (green) and FOS (red) from mice injected with RTX. Cells were collected at different time points between 0 and 24 h. Right, quantification of FOS+ cells (n = 6 sympathetic ganglia from 3 mice for each time point). d, Quantification of the efficiency of chemical sympathectomy (n = 6 mice for each condition, two-tailed unpaired t-test), and percentage of white hairs in RTX-injected mice treated with vehicle (ctrl) or 6-OHDA (SN abla) (n = 6 mice for each condition, two-tailed unpaired t-test). e, Guanethidine (gua) injection blocks the formation of white hairs that is induced by RTX injection (quantification for percentage of white hairs: n = 14 mice for each condition, two-tailed unpaired t-test; quantification for numbers of MeSCs: n = 30 hair follicles from 6 mice for each condition, two-tailed unpaired t-test). f, Left, immunofluorescent staining of sympathetic ganglia for tyrosine hydroxylase (green) and FOS (red) from THcreERT2;CAG-LSL-Gq-DREADD mice that were injected with CNO; cells were collected 6 h later. Right, quantification of FOS+ cells (n = 6 sympathetic ganglia from 2 mice for each condition, two-tailed unpaired t-test). g, Formation of white hairs after intradermal injection of CNO into THcreERT2;CAG-LSL-Gq-DREADD mice (n = 6 injection sites from 5 mice for each condition, two-tailed unpaired t-test). Yellow dashed circles denote intradermal CNO injection sites. h, Quantification of the percentage of white hairs at CNO injection sites in mosaically induced THcreERT2;CAG-LSL-Gq-DREADD;RosamT/mG mice (n = 5 injection sites from 4 mice for each condition, two-tailed unpaired t-test). Scale bars, 50 μm. All data are mean ± s.d.
Extended Data Fig. 6 Analysis of apoptosis and proliferation of MeSCs and the effect of RTX or noradrenaline on mature melanocytes.
a, Left, immunofluorescent staining of active caspase-3 (aCAS3; green) and TRP2 (red) from mice one day after injection with RTX or noradrenaline. Right, quantification of aCAS3+ MeSCs (n = 30 hair follicles from 6 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). b, TUNEL assay of hair follicles from mice one day after treatment with RTX or noradrenaline. Catagen hair follicles were used as positive controls for TUNEL. White arrowhead points to apoptotic hair follicle cells (n = 30 hair follicles from 6 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). c, Formation of white hairs in Ripk3 mutant mice (RIPK3 KO) injected with RTX (n = 5 mice for each condition, two-tailed unpaired t-test). d, Left, immunofluorescent staining of hair follicles for the DNA damage marker γ-H2AX (green) and TRP2 (red) from mice one day after treatment with RTX or noradrenaline. Hair follicles from irradiated mice were used as positive controls. White arrowhead indicates the MeSCs with DNA damage. Right, quantification of γ-H2AX+ MeSCs (n = 30 hair follicles from 6 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). e, Left, immunofluorescent staining for phosphorylated histone H3 (pHH3; green) and TRP2 (red) of control hair follicles at different stages of the hair cycle. Right, quantification of pHH3+ MeSCs (n = 25 hair follicles from 3 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). f, Left, immunofluorescent staining of hair bulbs for aCAS3 (green) and TRP2 (red) from mice one day after injection with RTX or noradrenaline. Right, quantification of aCAS3+ mature melanocytes (MCs) (n = 30 hair follicles from 3 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). g, Left, immunofluorescent staining of hair bulbs for pHH3 (green) and TRP2 (red) from mice one day after injection with RTX or noradrenaline. Right, quantification of pHH3+ mature melanocytes (n = 30 hair follicles from 3 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). h, Left, schematic of the strategy for isolation of MeSCs. Right, FACS analysis of the numbers of MeSCs one day after RTX injection (n = 5 mice for each condition, two-tailed unpaired t-test). i, Fontana-Masson melanin staining of anagen or telogen samples five days after injection of saline or RTX (n = 6 mice for each condition, two-tailed unpaired t-test). Blue arrowheads indicate ectopic pigments. Scale bars, 50 μm. All data are mean ± s.d.
Extended Data Fig. 7 Differential gene expression in normal and stressed MeSCs.
a, FACS strategy for the purification of MeSCs. MeSCs were selected on the basis of their expression of CD117, from a population that is negative for CD140A, CD45, SCA1 and CD34 and that shows modest expression for integrin α6. b, Sample clustering based on Pearson's correlation of transcriptomes between control and stressed MeSCs (n = 2 biologically independent samples for each condition). c, Heat map of all differentially expressed genes (n = 2 biologically independent samples for each condition; P values were calculated using the Wald test implemented in DESeq2, and adjusted using the Benjamini–Hochberg method. Differentially expressed genes were those that had the absolute value of log2(gene expression in stressed versus control MeSCs) ≥ 0.58 and adjusted P value < 0.05. d, Expression levels of marker genes for different cell types in the skin, confirming the purity of MeSCs that were used for RNA-seq (n = 4 biologically independent samples). TPM, transcripts per million. e, Heat maps showing expression of signature genes that are related to the differentiation of MeSCs. f, Heat maps showing expression of cell-cycle signature genes. Hn1 is also known as Jpt1. g, qRT–PCR validation of selected differentially expressed genes in FACS-purified mouse MeSCs from skins of control and RTX-injected mice (n = 4 biological replicates for each condition, two-way ANOVA with Benjamini–Hochberg correction). All data are mean ± s.d.
Extended Data Fig. 8 Proliferation analysis of RTX-injected mice that were treated with CDK inhibitors chemically or genetically.
a, b, Left, immunofluorescent staining of upper hair follicles (a) and hair bulbs (b) for pHH3 (green) and TRP2 (red) from mice one day after RTX injection, RTX injection with topical application of CDK inhibitors (AT7519 or flavopiridol) or RTX injection with MeSC-specific overexpression of P27 (MeSC-P27 OE). Right, quantification of pHH3+ cells (n = 30 hair follicles from 3 mice for each condition, one-way ANOVA with Tukey’s multiple comparisons test). Scale bars, 50 μm. All data are mean ± s.d.
Supplementary information
Supplementary Information
This document contains further discussions of the mechanisms of action of RTX, the sympathetic nervous system, DREADDs as a chemogenetic tool to manipulate nerve activity, mechanisms of MeSC depletion under stress, and ectopic pigmentation under stress.
Source data
Rights and permissions
About this article
Cite this article
Zhang, B., Ma, S., Rachmin, I. et al. Hyperactivation of sympathetic nerves drives depletion of melanocyte stem cells. Nature 577, 676–681 (2020). https://doi.org/10.1038/s41586-020-1935-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-1935-3
This article is cited by
-
Sexual dimorphism in melanocyte stem cell behavior reveals combinational therapeutic strategies for cutaneous repigmentation
Nature Communications (2024)
-
Deciphering the molecular mechanisms of stem cell dynamics in hair follicle regeneration
Experimental & Molecular Medicine (2024)
-
Local and systemic mechanisms that control the hair follicle stem cell niche
Nature Reviews Molecular Cell Biology (2024)
-
Reproducible strategy for excisional skin-wound-healing studies in mice
Nature Protocols (2024)
-
Skeletal interoception in osteoarthritis
Bone Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.