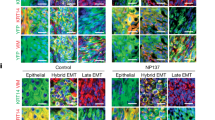
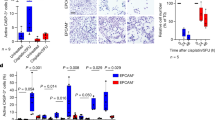
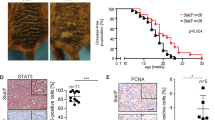
Abstract
FAT1, which encodes a protocadherin, is one of the most frequently mutated genes in human cancers1,2,3,4,5. However, the role and the molecular mechanisms by which FAT1 mutations control tumour initiation and progression are poorly understood. Here, using mouse models of skin squamous cell carcinoma and lung tumours, we found that deletion of Fat1 accelerates tumour initiation and malignant progression and promotes a hybrid epithelial-to-mesenchymal transition (EMT) phenotype. We also found this hybrid EMT state in FAT1-mutated human squamous cell carcinomas. Skin squamous cell carcinomas in which Fat1 was deleted presented increased tumour stemness and spontaneous metastasis. We performed transcriptional and chromatin profiling combined with proteomic analyses and mechanistic studies, which revealed that loss of function of FAT1 activates a CAMK2–CD44–SRC axis that promotes YAP1 nuclear translocation and ZEB1 expression that stimulates the mesenchymal state. This loss of function also inactivates EZH2, promoting SOX2 expression, which sustains the epithelial state. Our comprehensive analysis identified drug resistance and vulnerabilities in FAT1-deficient tumours, which have important implications for cancer therapy. Our studies reveal that, in mouse and human squamous cell carcinoma, loss of function of FAT1 promotes tumour initiation, progression, invasiveness, stemness and metastasis through the induction of a hybrid EMT state.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All the raw sequencing data have been deposited in the Gene Expression Omnibus with the following accession numbers: mouse RNA-seq (GSE158502), human RNA-seq (GSE158501), ATAC-seq (GSE158501), whole-exome sequencing (GSE158503), low-coverage whole-genome sequencing (GSE158505) or a global accession number (GSE158506. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD022268. All other relevant data are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Morris, L. G. et al. Recurrent somatic mutation of FAT1 in multiple human cancers leads to aberrant Wnt activation. Nat. Genet. 45, 253–261 (2013).
Dotto, G. P. & Rustgi, A. K. Squamous cell cancers: a unified perspective on biology and genetics. Cancer Cell 29, 622–637 (2016).
Sánchez-Danés, A. & Blanpain, C. Deciphering the cells of origin of squamous cell carcinomas. Nat. Rev. Cancer 18, 549–561 (2018).
The ICGC/TCGA Pan-Cancer Analysis of Whole Genomes Consortium. Pan-cancer analysis of whole genomes. Nature 578, 82–93 (2020).
Lawrence, M. S. et al. Discovery and saturation analysis of cancer genes across 21 tumour types. Nature 505, 495–501 (2014).
Li, Z. et al. Loss of the FAT1 tumor suppressor promotes resistance to CDK4/6 inhibitors via the Hippo pathway. Cancer Cell 34, 893–905.e8 (2018).
Nassar, D., Latil, M., Boeckx, B., Lambrechts, D. & Blanpain, C. Genomic landscape of carcinogen-induced and genetically induced mouse skin squamous cell carcinoma. Nat. Med. 21, 946–954 (2015).
Martin, D. et al. Assembly and activation of the Hippo signalome by FAT1 tumor suppressor. Nat. Commun. 9, 2372 (2018).
Hu, X. et al. FAT1 prevents epithelial mesenchymal transition (EMT) via MAPK/ERK signaling pathway in esophageal squamous cell cancer. Cancer Lett. 397, 83–93 (2017).
Srivastava, C. et al. FAT1 modulates EMT and stemness genes expression in hypoxic glioblastoma. Int. J. Cancer 142, 805–812 (2018).
Pastushenko, I. et al. Identification of the tumour transition states occurring during EMT. Nature 556, 463–468 (2018).
Latil, M. et al. Cell-type-specific chromatin states differentially prime squamous cell carcinoma tumor-initiating cells for epithelial to mesenchymal transition. Cell Stem Cell 20, 191–204.e5 (2017).
Pastushenko, I. & Blanpain, C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 29, 212–226 (2019).
Nieto, M. A., Huang, R. Y., Jackson, R. A. & Thiery, J. P. EMT: 2016. Cell 166, 21–45 (2016).
Boumahdi, S. et al. SOX2 controls tumour initiation and cancer stem-cell functions in squamous-cell carcinoma. Nature 511, 246–250 (2014).
Ferone, G. et al. SOX2 is the determining oncogenic switch in promoting lung squamous cell carcinoma from different cells of origin. Cancer Cell 30, 519–532 (2016).
Siegle, J. M. et al. SOX2 is a cancer-specific regulator of tumour initiating potential in cutaneous squamous cell carcinoma. Nat. Commun. 5, 4511 (2014).
Cooper, C. D. & Lampe, P. D. Casein kinase 1 regulates connexin-43 gap junction assembly. J. Biol. Chem. 277, 44962–44968 (2002).
Lewis, C. A., Townsend, P. A. & Isacke, C. M. Ca2+/calmodulin-dependent protein kinase mediates the phosphorylation of CD44 required for cell migration on hyaluronan. Biochem. J. 357, 843–850 (2001).
Huang, R. Y. et al. Identification of CaMKII phosphorylation sites in connexin43 by high-resolution mass spectrometry. J. Proteome Res. 10, 1098–1109 (2011).
Chen, C., Zhao, S., Karnad, A. & Freeman, J. W. The biology and role of CD44 in cancer progression: therapeutic implications. J. Hematol. Oncol. 11, 64 (2018).
Jolly, M. K. et al. Interconnected feedback loops among ESRP1, HAS2, and CD44 regulate epithelial–mesenchymal plasticity in cancer. APL Bioeng. 2, 031908 (2018).
Chellaiah, M. A., Biswas, R. S., Rittling, S. R., Denhardt, D. T. & Hruska, K. A. Rho-dependent Rho kinase activation increases CD44 surface expression and bone resorption in osteoclasts. J. Biol. Chem. 278, 29086–29097 (2003).
Göllner, S. et al. Loss of the histone methyltransferase EZH2 induces resistance to multiple drugs in acute myeloid leukemia. Nat. Med. 23, 69–78 (2017).
Avgustinova, A. & Benitah, S. A. Epigenetic control of adult stem cell function. Nat. Rev. Mol. Cell Biol. 17, 643–658 (2016).
Panciera, T., Azzolin, L., Cordenonsi, M. & Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 18, 758–770 (2017).
Machiels, J. P. et al. Afatinib versus methotrexate as second-line treatment in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck progressing on or after platinum-based therapy (LUX-Head & Neck 1): an open-label, randomised phase 3 trial. Lancet Oncol. 16, 583–594 (2015).
Planchard, D. et al. Dabrafenib plus trametinib in patients with previously untreated BRAFV600E-mutant metastatic non-small-cell lung cancer: an open-label, phase 2 trial. Lancet Oncol. 18, 1307–1316 (2017).
Acknowledgements
We thank the ULB animal facility and ULB genomic core facility (F. Libert and A. Lefort); and J. Allard from CMMI (supported by the European Regional Development Fund and the Walloon Region). I.P. is supported by FNRS and Foundation Against Cancer (FCC). F.M. was supported by FNRS post-doctoral fellowship and by the TELEVIE. The Department of Pathology (Erasme Hospital, ULB) acknowledges Fonds Yvonne Boel. C. Decaestecker is a senior research associate in F.R.S.-FNRS. The patient-derived xenograft project was supported by Fonds Erasme. C. Blanpain is supported by WELBIO, FNRS, Fond Erasme, Fondation Contre le Cancer, ULB Foundation, European Research Council, Worldwide Cancer Research and the Foundation Baillet Latour.
Author information
Authors and Affiliations
Contributions
I.P., F.M. and C. Blanpain designed the experiments and performed data analysis. I.P. and F.M. performed most of the biological experiments. F.H. generated Fat1-cKO mice and provided her expertise. Y. Song performed bioinformatic analysis. F.d.C. and B.S. helped to perform CRISPR experiments. B.M. and V.J. performed intratracheal AdenoCRE installation for lung cancer generation. F.I. and D.V.H. performed phosphoproteomic analysis. M.O. and M.T. performed stiffness experiments. M.V. and D.P.-M. performed electron microscopy imaging and analysis. F.I. and D.V.H. performed phosphoproteomic analysis. Y.B. and C.S. performed analysis of patient survival using the TCGA database. I.S., Y. Sokolow, S.H., A.P.-B., B.A.-M., L.R.-B., P.J., M.D.T., P.R., R.S.-G., N.D’H., J.F.M.-C. and O.S. provided human samples. C. Balsat, C. Decaestecker and Y.-R.V.E. performed staining and analysis of patient-derived xenograft samples. C. Dubois performed FACS. V.M., S.L., G.L., J.B., M.R. and S.S. performed immunostainings, western blotting, treatments and follow-up of the mice. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
C. Blanpain, I.P. and F.M. are co-inventors on a patent application on the use of SRC inhibitors for the treatment of FAT1-mutated cancers.
Additional information
Peer review information Nature thanks Salvador Aznar Benitah and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Fat1 LOF does not alter development and skin homeostasis.
a, Image showing Fat1-cKO mouse and its control littermate. b, Table showing the number of control mice and mice with constitutive Fat1-cKO in skin epidermis, showing the absence of deviation from Mendelian ratio. c, Haematoxylin and eosin staining in control and Fat1-cKO epidermis. Scale bar, 50 μm. d, e, Immunostaining for GFP and KRT10, KRT14, E-cadherin or ITGB4 in control (d) and Fat1-cKO (e) epidermis. Scale bar, 50 μm.
Extended Data Fig. 2 Fat1 LOF accelerates DMBA/TPA-induced tumour initiation and malignant progression.
a, Model allowing constitutive Fat1 deletion in the skin epidermis and the scheme of DMBA/TPA protocol. b, Control and Fat1-cKO littermates 24 weeks after initiation of DMBA/TPA treatment. c, d, Time elapsed from the beginning of DMBA/TPA treatment until the appearance of the tumour (log-rank Mantel–Cox test) (c) and the number of papillomas per mouse (mean ± s.e.m., two-tailed t-test) (d) in control and Fat1-cKO mice. e, Macroscopic appearance of papilloma and carcinoma. f, Number of carcinomas per mouse at 24 weeks after DMBA/TPA in control and Fat1-knockout mice. Mean ± s.e.m., two-tailed t-test. g, Acute deletion of Fat1 in DMBA/TPA-induced papillomas. h, Time elapsed from tamoxifen (tam) administration to macroscopic malignant progression from papillomas into carcinomas. log-rank Mantel–Cox test. i, Proportion of papillomas that progressed to carcinomas in control and Fat1-cKO mice. χ2 test. j, k, Immunostaining for KRT14, KRT10 and LAM5 in control (j) and Fat1-cKO papillomas (k) 0, 2, 4 and 8 weeks after tamoxifen administration. Scale bar, 50 μm. l, Haematoxylin and eosin and immunostaining for YFP, E-cadherin, ZO-1 or CLDN1 in control and Fat1-cKO papillomas. Scale bar, 50 μm. m–o, Electron microscopy images showing polarity (Scale bars, 2 μm (control papilloma), 5 μm (Fat1-cKO papilloma)) (m), cell–cell adhesion (scale bar, 0.2 μm) (n) or desmosomes (scale bar, 0.2 μm) (o) in Fat1-cKO and wild-type papillomas. Red lines indicate interface between tumour cells and stroma. Blue arrowheads, desmosomes. Red arrowheads, tight and adherens junctions. p, Width of the desmosomes measured in nm in control and Fat1-cKO papillomas. Mean ± s.e.m., two-tailed t-test. q, Electron microscopy (scale bars, 0.2 μm (control), 5 μm (Fat1-cKO)) and immunostaining for KRT14 and LAM5 (scale bar, 50 μm) of control and Fat1-cKO papillomas. Blue arrowheads, hemidesmosomes. Red arrowheads, basal lamina in control papillomas and discontinued basal lamina in Fat1-cKO papillomas. Black arrowheads show fenestration of basal lamina in Fat1-cKO papillomas. r, Number of hemidesmosomes per 1 μm. Mean ± s.e.m., two-tailed t-test).
Extended Data Fig. 3 EMT in papillomas and gating strategy for FACS analysis and cell sorting of the tumour subpopulations.
a, Percentage of EPCAM+YFP+ tumour cells in control and Fat1-cKO papillomas. Mean ± s.e.m., two-tailed t-test. b, c, mRNA (qPCR) expression of epithelial (b) and mesenchymal (c) genes in EPCAM+ and EPCAM− control and Fat1-cKO papillomas. Mean + s.e.m. d, FACS plots showing the gating strategy used to FACS-isolate or to analyse the proportion of YFP+EPCAM+ and EPCAM− tumour cells from DMBA/TPA-induced K14-cre;Fat1cKO;Rosa26YFP/+ carcinomas and papillomas, Lgr5-creER;KRasG12D;p53cKO;Fat1cKO;Rosa26YFP/+ or K14-creER;KRasG12D;p53cKO;Fat1cKO;Rosa26YFP/+ skin SCCs and KrasG12D;p53cKO;Fat1cKO;Rosa26YFP/+ lung carcinomas. e, FACS plots showing the gating strategy to define the six subpopulations of EPCAM− tumour cells: EPCAM−CD106−CD51−CD61− (triple negative), EPCAM−CD106+CD51−CD61−, EPCAM−CD106−CD51+CD61−, EPCAM−CD106+CD51+CD61−, EPCAM−CD106−CD51+CD61+ and EPCAM−CD106+CD51+CD61+ (triple positive) populations.
Extended Data Fig. 4 Fat1 LOF promotes hybrid EMT state in a genetic model of skin SCC.
a, Mouse model of skin SCC allowing YFP and KrasG12D expression as well as p53 and Fat1 deletion preferentially in the interfollicular epidermis (IFE) using Krt14-creER. b, Percentage of EPCAM+ tumour cells in control and Fat1-cKO SCCs. Mean ± s.e.m., two-tailed t-test. c, Graph showing the distribution of the different EPCAM− tumour cell subpopulations on the basis of the expression of CD106/VCAM1, CD61/ITGB3 and CD51/ITGAV in Fat1-cKO tumours. Mean ± s.e.m. d, Haematoxylin and eosin staining, showing representative control and Fat1-cKO tumours. Scale bar, 50 μm. e, f, Immunostaining for GFP, KRT14 or vimentin in representative control (e) and Fat1-cKO tumour (f). Scale bar, 50 μm. g, Mouse model of skin SCC allowing the expression of YFP and KrasG12D as well as p53 and Fat1 deletion preferentially in the hair follicle lineage using Lgr5-creER. h, Percentage of EPCAM+ tumour cells in the control and Fat1-cKO tumours. Mean ± s.e.m., two-tailed t-test. i, Graph showing the distribution of the different EPCAM− tumour cell subpopulations on the basis of the expression of CD106/VCAM1, CD61/ITGB3 and CD51/ITGAV in Fat1-cKO tumours. Mean ± s.e.m. j, Haematoxylin and eosin staining, showing a representative Fat1 wild-type and Fat1-cKO tumour. Scale bar, 50 μm. k, l, Immunostaining for KRT14 and vimentin showing the absence of keratin pearls in representative EPCAM− control SCC (k) and the presence of keratin pearls in representative EPCAM− Fat1-cKO SCC (l). White arrowheads indicate keratin pearls. Scale bar, 100 μm. m, Dot plot showing the number of keratin pearls quantified per field at magnification 20×. n = 5 fields quantified per sample, mean ± s.e.m., two-tailed t-test. n, Mouse model allowing YFP and KrasG12D expression as well as p53 and Fat1 deletion in lung epithelial cells using intratracheal instillation of Ad5CMVCre virus. o, Immunofluorescence image showing the YFP+ lung tumours 10 weeks after intratracheal instillation of Ad5CMVCre virus in Fat1 wild-type and Fat1-cKO mice. Scale bar, 1 mm.
Extended Data Fig. 5 Mutations in FAT1 promotes hybrid EMT state in human cancers.
a, Schematic representing the method of analysing the co-expression of pan-cytokeratin and vimentin using immunohistochemistry of patient-derived xenografts that present (or not) mutations in FAT1, and the definition of hybrid EMT score. b, Table summarizing the samples of patient-derived xenografts on which whole-exome sequencing was performed, and detailed information on the mutations: codon, amino acid change, the exon containing the mutation, the allelic frequency, the type of mutations and the bioinformatic prediction of the effect of the mutation on the function of the protein using three bioinformatic algorithms (SIFT, Memo and PolyPhen). c, Heat map showing the copy number variation profile of FAT1 genomic region in the patient-derived xenograft samples included in the analysis of hybrid EMT score. The colour code corresponds to the quantified copy number and the genomic coordinate (reference genome hg19) of bin set for quantification. The FAT1 gene is marked on each vertical edge. d, e, Box plot showing the distribution of the common mRNA signature (mouse skin and lung Fat1-cKO SCCs and human FAT1-knockout SCC cell line) compared to FAT1 mutation status in human lung SCC (TCGA database; for the analysis, only high-impact mutations in >20% of variant allele frequency were considered) (d) and FAT1 copy number variation status in human lung SCC (TCGA database) (e). Boundaries of the box indicate the first and third quartiles of the FAT1 RNA signature value. The bold horizontal line indicates the median and the two external horizontal lines shows the minimum and maximum values. The dots represent all data points. Differences between the two groups are assessed using a two-sided Wilcoxon rank-sum test. f, g, Venn diagram of the genes upregulated in the EPCAM+ Fat1-cKO skin SCC and upregulated in LGR5 EPCAM+ versus triple-negative hybrid EMT tumour cells (f) or in triple-negative versus EPCAM+ cells (g). Two-sided hypergeometric test.
Extended Data Fig. 6 EPCAM+ Fat1-cKO tumour cells are epigenetically primed to undergo EMT, whereas EPCAM− Fat1-cKO sustain the expression of epithelial program.
a, ATAC-seq profiles of the chromatin regulatory regions of mesenchymal genes closed in control EPCAM+ tumour cells and opened in EPCAM+ Fat1-cKO tumour cells, showing epigenetic priming of EPCAM+ Fat1-cKO tumour cells to undergo EMT. b, ATAC-seq profiles of the chromatin regulatory regions of mesenchymal genes with open chromatin regions only in EMT EPCAM− tumour cells. c, Transcription factor motifs enriched in the ATAC-seq peaks upregulated between the EPCAM+ Fat1-cKO and EPCAM+ control tumour cells as determined by Homer. Cumulative hypergeometric distributions. White boxes show core transcription factors; boxes highlighted in green show epithelial transcription factors; and boxes highlighted in orange show EMT transcription factors. d, Transcription factor motifs enriched in the ATAC-seq peaks that are upregulated between the EPCAM− Fat1-cKO and EPCAM− control tumour cells as determined by Homer analysis. Cumulative hypergeometric distributions. White boxes show core transcription factors; boxes highlighted in green show epithelial transcription factors; boxes highlighted in orange show EMT transcription factors; and boxes highlighted in grey show other transcription factors. e, ATAC-seq of the chromatin regulatory regions of epithelial genes with open chromatin regions in EPCAM− Fat1-cKO tumour cells as compared to EPCAM− tumour cells from LGR5-derived SCCs, showing the sustained opening of epithelial enhancers in EPCAM− Fat1-cKO tumour cells. f, ATAC-seq of the chromatin regulatory regions of epithelial genes that are closed upon EMT, irrespective of Fat1 deletion. g, Immunostaining for GFP and AP2G, KLF4, SOX2, p63, GRHL2 or ZEB1 in EPCAM+ and EPCAM− control and Fat1-cKO DMBA/TPA skin SCCs. Scale bar, 50 μm.
Extended Data Fig. 7 Loss of cell adhesion is not sufficient to induce the hybrid EMT phenotype.
a, Images showing spheroids formed 7 d after plating 4,000 FAT1 wild-type or FAT1 wild-type, CDH1-knockout human A388 skin SCC cells on an ultra-low adherent plate. b, Bar chart showing the quantification by FACS of the number of cells in FAT1 wild-type, and FAT1 wild-type and CDH1-knockout, spheroids. Mean + s.e.m., two-tailed t-test. c, Immunostaining for E-cadherin, YAP1, ZEB1 and SOX2 in FAT1 wild-type, and FAT1 wild-type and CDH1-knockout, tumour cells. Scale bar, 50 μm. d, Images showing spheroids formed 7 d after plating 4,000 FAT1-knockout or FAT1-knockout and CDH1-overexpressing human A388 skin SCC cells on an ultra-low attachment plate. e, Bar chart showing the quantification by FACS of the number of cells in FAT1-knockout or FAT1-knockout and CDH1-overexpressing spheroids. Mean + s.e.m., two-tailed t-test. f, Immunostaining for E-cadherin, YAP1, ZEB1 and SOX2 in FAT1-knockout or FAT1-knockout and CDH1-overexpressing tumour cells. Scale bar, 50 μm. g–i, Immunostaining of K14 and vimentin after subcutaneous transplantation of Fat1-cKO (g), Fat1 and Sox2 double-knockout (h) or Fat1, Yap1 and Taz triple-knockout (i) mouse skin SCC cells. Mean ± s.e.m. Scale bars, 50 μm. j, k, Venn diagram of the genes upregulated in EPCAM− Fat1-cKO skin SCC upon Sox2 deletion (j) or upon Yap1 and Taz deletion (k), and upregulated genes in hybrid EMT triple-negative cells versus late EMT triple-positive cells (early hybrid EMT signature) and in triple-positive versus EPCAM+ cells (late EMT signature). Two-sided hypergeometric test.
Extended Data Fig. 8 Phosphoproteomic analysis reveals signalling cascades downstream of FAT1 LOF.
a, Table showing kinases that are significantly more phosphorylated in FAT1 wild-type cells as compared to FAT1-knockout cells. t-test, FDR = 0.05, S0 = 1. b, Bar chart showing the kinases that are predicted to phosphorylate phosphosites significantly enriched in FAT1 wild-type tumour cells. c, Table showing kinases that are significantly more phosphorylated in FAT1-knockout cells as compared to FAT1 wild-type cells. t-test, FDR = 0.05, S0 = 1. d, Bar chart showing the kinases that are predicted to phosphorylate phosphosites that are significantly enriched in FAT1-knockout tumour cells. e, Table showing the sites in FAT1-knockout cells predicted to be phosphorylated by CAMK2. f, g, Western blot showing pEGFR and EGFR (f) or pMEK and MEK (g) in Fat1 wild-type and Fat1-knockout Lgr5-creER;KrasG12D;p53cKO;Fat1fl/fl;RYFP mouse skin SCC cells. h, Western blot showing the expression levels of pSRC, total SRC and YES on the input of wild-type and FAT1-knockout cells, and upon immunoprecipitation of SRC and YES (n = 4). The apparent molecular weight reference in kDa is indicated close to f–h.
Extended Data Fig. 9 Full scan of western blot membranes.
a–k, Images showing full scan of western blot membranes displayed in Fig. 4. Each panel indicates the figure and the panel to which the full membrane image belongs. Molecular weight size standards are indicated on each membrane. In all the experiments the controls (β-actin) were run on the same gel as the samples.
Extended Data Fig. 10 Increase in YAP1 and SOX2 signalling downstream of FAT1 LOF is independent of the stiffness of the substrate.
a, Immunostaining for YAP1 in FAT1 wild-type and FAT1-knockout human SCC cells upon increasing stiffness conditions. Scale bar, 50 μm. b, Quantification of YAP1 expression on the basis of fluorescence intensity in FAT1 wild-type and FAT1-knockout cells in different stiffness conditions. Mean + s.e.m. c, Quantification of YAP1 nuclear/cytoplasmic ratio on the basis of fluorescence intensity in FAT1 wild-type and FAT1-knockout cells in different stiffness conditions. Mean + s.e.m. d, Immunostaining for SOX2 in FAT1 wild-type and FAT1-knockout human SCC cells in increasing stiffness conditions. Scale bar, 50 μm e, Model of the signalling pathways that are activated or repressed in FAT1-knockout cells to induce a hybrid EMT state and to predict a differential effect on the response to therapy.
Supplementary information
Supplementary Information
This file contains Supplementary Methods.
Supplementary Figure
This file contains full scan gels for Extended Data figure 9.
Rights and permissions
About this article
Cite this article
Pastushenko, I., Mauri, F., Song, Y. et al. Fat1 deletion promotes hybrid EMT state, tumour stemness and metastasis. Nature 589, 448–455 (2021). https://doi.org/10.1038/s41586-020-03046-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-03046-1
This article is cited by
-
Polycomb repressive complex 2 and its core component EZH2: potential targeted therapeutic strategies for head and neck squamous cell carcinoma
Clinical Epigenetics (2024)
-
STAT3–mediated up-regulation of DAB2 via SRC-YAP1 signaling axis promotes Helicobacter pylori-driven gastric tumorigenesis
Biomarker Research (2024)
-
LINC00460-FUS-MYC feedback loop drives breast cancer metastasis and doxorubicin resistance
Oncogene (2024)
-
Genetic and prognostic analysis of blastoid and pleomorphic mantle cell lymphoma: a multicenter analysis in China
Annals of Hematology (2024)
-
Visualization of breast cancer-related protein synthesis from the perspective of bibliometric analysis
European Journal of Medical Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.