Abstract
Immune surveillance against pathogens and tumours in the central nervous system is thought to be limited owing to the lack of lymphatic drainage. However, the characterization of the meningeal lymphatic network has shed light on previously unappreciated ways that an immune response can be elicited to antigens that are expressed in the brain1,2,3. Despite progress in our understanding of the development and structure of the meningeal lymphatic system, the contribution of this network in evoking a protective antigen-specific immune response in the brain remains unclear. Here, using a mouse model of glioblastoma, we show that the meningeal lymphatic vasculature can be manipulated to mount better immune responses against brain tumours. The immunity that is mediated by CD8 T cells to the glioblastoma antigen is very limited when the tumour is confined to the central nervous system, resulting in uncontrolled tumour growth. However, ectopic expression of vascular endothelial growth factor C (VEGF-C) promotes enhanced priming of CD8 T cells in the draining deep cervical lymph nodes, migration of CD8 T cells into the tumour, rapid clearance of the glioblastoma and a long-lasting antitumour memory response. Furthermore, transfection of an mRNA construct that expresses VEGF-C works synergistically with checkpoint blockade therapy to eradicate existing glioblastoma. These results reveal the capacity of VEGF-C to promote immune surveillance of tumours, and suggest a new therapeutic approach to treat brain tumours.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Code availability
All of the code used for analysis is described in the methods. Detailed files will be made available from the corresponding authors on request.
Change history
26 January 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41586-021-03204-z
References
Aspelund, A. et al. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J. Exp. Med. 212, 991–999 (2015).
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Louveau, A. et al. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat. Neurosci. 21, 1380–1391 (2018).
Antila, S. et al. Development and plasticity of meningeal lymphatic vessels. J. Exp. Med. 214, 3645–3667 (2017).
Da Mesquita, S. et al. Functional aspects of meningeal lymphatics in ageing and Alzheimer’s disease. Nature 560, 185–191(2018).
Harris, M. G. et al. Immune privilege of the CNS is not the consequence of limited antigen sampling. Sci. Rep. 4, 4422 (2014).
Mathieu, E., Gupta, N., Macdonald, R. L., Ai, J. & Yücel, Y. H. In vivo imaging of lymphatic drainage of cerebrospinal fluid in mouse. Fluids Barriers CNS 10, 35 (2013).
Fankhauser, M. et al. Tumor lymphangiogenesis promotes T cell infiltration and potentiates immunotherapy in melanoma. Sci. Transl. Med. 9, eaal4712 (2017).
Skobe, M. et al. Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis. Nat. Med. 7, 192–198 (2001).
Gilbert, M. R. et al. A randomized trial of bevacizumab for newly diagnosed glioblastoma. N. Engl. J. Med. 370, 699–708 (2014).
Cloughesy, T. F. et al. Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat. Med. 25, 477–486 (2019).
Mingozzi, F. & High, K. A. Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood 122, 23–36 (2013).
Sabnis, S. et al. A novel amino lipid series for mRNA delivery: improved endosomal escape and sustained pharmacology and safety in non-human primates. Mol. Ther. 26, 1509–1519 (2018).
Joukov, V. et al. Proteolytic processing regulates receptor specificity and activity of VEGF-C. EMBO J. 16, 3898–3911 (1997).
Zincarelli, C., Soltys, S., Rengo, G. & Rabinowitz, J. E. Analysis of AAV serotypes 1–9 mediated gene expression and tropism in mice after systemic injection. Mol. Ther. 16, 1073–1080 (2008).
Zeng, J. et al. Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int. J. Radiat. Oncol. Biol. Phys. 86, 343–349 (2013).
Kim, J. E. et al. Combination therapy with anti-PD-1, anti-TIM-3, and focal radiation results in regression of murine gliomas. Clin. Cancer Res. 23, 124–136 (2017).
Filley, A. C., Henriquez, M. & Dey, M. Recurrent glioma clinical trial, CheckMate-143: the game is not over yet. Oncotarget 8, 91779–91794 (2017).
Garzon-Muvdi, T. et al. Dendritic cell activation enhances anti-PD-1 mediated immunotherapy against glioblastoma. Oncotarget 9, 20681–20697 (2018).
Chongsathidkiet, P. et al. Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors. Nat. Med. 24, 1459–1468 (2018).
Belcaid, Z. et al. Focal radiation therapy combined with 4-1BB activation and CTLA-4 blockade yields long-term survival and a protective antigen-specific memory response in a murine glioma model. PLoS ONE 9, e101764 (2014).
Bronte, V. et al. Effective genetic vaccination with a widely shared endogenous retroviral tumor antigen requires CD40 stimulation during tumor rejection phase. J. Immunol. 171, 6396–6405 (2003).
Rooney, M. S., Shukla, S. A., Wu, C. J., Getz, G. & Hacohen, N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell 160, 48–61 (2015).
He, Y. et al. Vascular endothelial cell growth factor receptor 3-mediated activation of lymphatic endothelium is crucial for tumor cell entry and spread via lymphatic vessels. Cancer Res. 65, 4739–4746 (2005).
Hirakawa, S. et al. VEGF-C-induced lymphangiogenesis in sentinel lymph nodes promotes tumor metastasis to distant sites. Blood 109, 1010–1017 (2007).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Siddiqui, I. et al. Intratumoral Tcf1+PD-1+CD8+ T cells with stem-like properties promote tumor control in response to vaccination and checkpoint blockade immunotherapy. Immunity 50, 195–211 (2019).
Lucca, L. E. et al. TIGIT signaling restores suppressor function of Th1 Tregs. JCI Insight 4, 124427 (2019).
Huang, A. C. et al. T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 545, 60–65 (2017).
Tawbi, H. A. et al. Combined nivolumab and ipilimumab in melanoma metastatic to the brain. N. Engl. J. Med. 379, 722–730 (2018).
Taggart, D. et al. Anti-PD-1/anti-CTLA-4 efficacy in melanoma brain metastases depends on extracranial disease and augmentation of CD8+ T cell trafficking. Proc. Natl Acad. Sci. USA 115, E1540–E1549 (2018).
Medawar, P. B. Immunity to homologous grafted skin. III. The fate of skin homografts transplanted to the brain, to subcutaneous tissue, and to the anterior chamber of the eye. Br. J. Exp. Pathol. 29, 58–69 (1948).
Volovitz, I. et al. Split immunity: immune inhibition of rat gliomas by subcutaneous exposure to unmodified live tumor cells. J. Immunol. 187, 5452–5462 (2011).
Breslin, J. W. et al. Vascular endothelial growth factor-C stimulates the lymphatic pump by a VEGF receptor-3-dependent mechanism. Am. J. Physiol. Heart Circ. Physiol. 293, H709–H718 (2007).
Wang, J. et al. UV-induced somatic mutations elicit a functional T cell response in the YUMMER1.7 mouse melanoma model. Pigment Cell Melanoma Res. 30, 428–435 (2017).
Acknowledgements
We thank members of the A.I. laboratory for insightful discussions and help with protocols; M. Saltzman (Yale Biomedical Engineering) for allowing us to use mice stereotaxic equipment and for help with materials; S. Lee from the J.-L.T. laboratory for initial help in breeding mice; and the ICM Vectorology platform for production of AAV material. This study was supported by National Institutes of Health grants T32GM007205 (MSTP training grant) and F30CA239444 (to E.S.); AI054359 and AI127429 (to A.I.); R01EB016629-01 and R01 EY025979-01 (to J.-L.T.); and CA196660, CA128814 and CA121974 (to M.B.). A.I. is an investigator of the Howard Hughes Medical Institute. L.S.B.B. and J.-L.T. were supported by the Yale School of Medicine. Work in the K.A. laboratory was funded by the iCAN Digital Precision Cancer Medicine Flagship, Academy of Finland (grants 292816, 273817 and 307366), the Centre of Excellence Program 2014–2019, the Cancer Foundation in Finland, the Sigrid Juselius Foundation, the Hospital District of Helsinki, Uusimaa Research Grants, Helsinki Institute of Life Sciences (HiLIFE) and Biocenter Finland.
Author information
Authors and Affiliations
Contributions
E.S., J.-L.T. and A.I. planned the project. E.S. and A.I. designed, analysed and interpreted data and wrote the manuscript. E.S., L.S.B.B. and T.M. performed experiments and analysed data. H.D. bred and cared for mice. J.-L.T. provided AAV material. S.A., M.B. and K.A. provided expertise, materials and analysis of data.
Corresponding authors
Ethics declarations
Competing interests
A.I., E.S. and J.-L.T. have filed a patent related to the manuscript as inventors (application number, US 62/768,390; status of application, provisional; specific aspect of manuscript covered in patent application, manipulation of meningeal lymphatic vasculature for brain and CNS tumour therapy. K.A. is an inventor on several patents that relate to VEGF-C. He and his wife currently have no income, stock or other benefits from companies that are related to the manuscript. Helsinki University is a shareholder in companies that are related to the use of VEGF-C. M.B. is a consultant for Eli Lilly and Company.
Additional information
Peer review information Nature thanks Jonathan Kipnis, Ingo K. Mellinghoff, Michelle Monje-Deisseroth, Christine Moussion and Shannon Turley for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
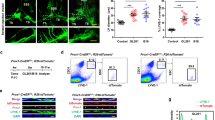
Extended Data Fig. 1 Increased meningeal lymphatic vasculature confers protection against intracranial glioblastoma challenge and provides long-term immunity without perturbance of the blood–brain barrier.
a, b, Mice inoculated with 50,000 GL261-Luc cells were imaged every 7 days and showed consistent and reliable tumour growth (n = 4). c, GL261-Luc cells result in lethality in mice in a cell-number-dependent manner (500 cells, n = 5 mice; 5,000 cells, n = 5 mice; 50,000 cells, n = 9 mice). d, Mice were injected intravenously with dextran–fluorescein (molecular weight, 70,000 kDa) and euthanized after 2 h. Brains were collected and cryosectioned (n = 4) The experiment was repeated independently with similar results. e, Mice were injected intravenously with 0.5% Evans Blue. After 2 h mice were perfused intraventricularly and Evans Blue was extracted from brain tissue using dimethylformamide (wild type, LPS, AAV-VEGF-C, VEGFC mRNA, n = 4; tumour, tumour + VEGFC mRNA, n = 5). BBB, blood–brain barrier. f, Representative images of AAV-CTRL and AAV-VEGF-C-treated mice after implantation of 5,000 cells. The experiment was repeated independently with similar results. g, Monitoring of the long-term survival of mice after AAV-VEGF-C and AAV-CTRL injections into the cisterna magna (n = 5). h, i, C57BL/6 mice received an injection of AAV-CTRL or AAV-VEGF-C through the cisterna magna. Six to eight weeks later, mice were euthanized and the dura was collected to image the lymphatic vasculature (LYVE1+) in the superior sagittal sinus (AAV-CTRL, n = 4; AAV-VEGF-C, n = 5). j, C57BL/6 mice that had been injected with CTRL-AAV or AAV-VEGF-C two months previously were implanted with 50,000 GL261-Luc cells in the striatum and monitored for survival (AAV-CTRL, n = 4; AAV-VEGF-C, n = 5). k, AAV-CTRL- or AAV-VEGF-C-treated mice were depleted of CD4 or CD8 T cells using anti-CD4 (GK1.5) or anti-CD8 (YTS169.4) antibodies starting one day before tumour inoculation (GL261) and redosed every four days afterwards (AAV-CTRL, n = 4; AAV-VEGF-C, n = 5; AAV-VEGF-C + anti-CD8, n = 4; AAV-VEGF-C + anti-CD4, n = 5). l, µMT B-cell-deficient mice were injected with AAV-CTRL or AAV-VEGF-C and challenged with 50,000 GL261-Luc cells two months afterwards (AAV-CTRL, n = 5; µMT AAV-CTRL, n = 3; µMT AAV-VEGF-C, n = 5). m, Top, schematic of the schedule of procedures for the experiments below (bottom panel) and in Fig. 1f. Mice injected with AAV-CTRL or AAV-VEGF-C that survived over 100 days after challenge with 5,000 GL261-Luc cells were rechallenged with 500,000 GL261-Luc cells in the flank. Bottom, IVIS imaging of mice ten days after flank rechallenge. Data are pooled from two independent experiments (h–m) and are mean ± s.d. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test or two-sided log-rank Mantel–Cox test).
Extended Data Fig. 2 Correlation of VEGF-C expression profiles between human and mouse glioblastoma.
a–c, RNA-seq data of tumour tissue and healthy brain tissue from different regions (TCGA, study accession phs000178.v10.p8, and GTEX, study accession phs000424.v7.p2, respectively. a, b, Expression profiles of VEGF-A (a) and VEGF-C (b) (glioblastoma (GBM), n = 147; cortex, n = 133; amygdala, n = 81; Brodmann areas 24 and 9, n = 215; C1 segment, n = 75; caudate nucleus, n = 135; cerebellar hemisphere, n = 115; cerebellum, n = 146; hippocampus, n = 103; hypothalamus, n = 101; nucleus accumbens, n = 125; putamen, n = 103; substantia nigra, n = 72; tibial nerve, n = 329). c, RNA-seq data of mouse healthy brain and GL261 tumours were analysed (n = 3 biologically independent samples). d, ONCLNC (www.oncolnc.org) data of patients with glioblastoma stratified into two groups (VEGF-C low, lower 33%; VEGF-C high, upper 33%; n = 50). e, Kaplan–Meier survival curves of patients from d (n = 50). Data are mean ± s.d. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test, two-sided log-rank Mantel–Cox test or Pearson’s correlation).
Extended Data Fig. 3 Validation of VEGFC mRNA in vitro, and its in vivo uptake and expression tropism.
a, The complementary DNA (cDNA) sequence of VEGFC that was used for mRNA production. b, 5-Methylcytosine and pseudouridine were used to substitute all of the cytosine and uridine bases in the mRNA. c, The VEGFC mRNA construct was transfected into HEK293T cells and cell lysate and the medium was collected to detect full-length and secreted intermediate and processed peptides. d, The VEGFC mRNA construct (5 µg) was delivered in vivo into the cisterna magna of mice using a JETPEI polyplex system. Six hours later, mouse CSF was collected using a capillary tube and filtered with an amicon filter, and the wash-through was used to run a western blot (each lane is n = 10 mice pooled). e, Raw western blot images for Extended Data Fig. 4c (gel 1: column 1, control; column 2, Cy5–GFP mRNA; column 3, VEGFC mRNA; column 4, control; column 5, Cy5–GFP mRNA; column 6, VEGFC mRNA; gel 2: column 1, control; column 2, Cy5–GFP mRNA; column 3, VEGFC mRNA) and Extended Data Fig. 4d (gel 3: columns 1–3, Cy5–GFP mRNA; columns 4–6, Cy5–GFP mRNA; columns 7-9, recombinant human VEGF-C in increasing concentration). The experiments were repeated twice independently with similar results. f, VEGFC mRNA and Cy5-labelled GFP mRNA were mixed at a 1:1 ratio and delivered to mice in vivo using JETPEI. Fifteen minutes later, mice were euthanized and the whole skull cap was imaged to observe the distribution of mRNA particles. The experiments were repeated independently twice with similar results. g, h, VEGFC mRNA and Cy5-labelled GFP mRNA were mixed at a 1:1 ratio and delivered to mice in vivo using JETPEI. After 24 h, the brains, meninges and lymph nodes of treated mice were collected for flow cytometry to measure the percentage of Cy5-positive cells in each compartment (control, n = 6; Cy5-labelled mRNA, n = 9; data are pooled from two independent experiments). i, The CSF, meninges, brain and serum were collected after two months (AAV-CTRL, AAV-VEGF-C), after 24 h (GFP mRNA, VEGFC mRNA) or at days 7 and 28 after tumour inoculation, and an ELISA was performed to detect VEGF-C (CSF: AAV-VEGF-C, n = 6; other groups, n = 3; 5 mice were pooled for each sample; meninges: AAV-VEGF-C, n = 6; 7-day tumour, n = 3; other groups, n = 5; brain: AAV-CTRL, GFP mRNA, n = 6; AAV-VEGF-C, VEGFC mRNA, n = 5; 7-day tumour, n = 3; 28-day tumour, n = 7; serum: n = 3). Data are mean ± s.d. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test).
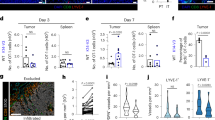
Extended Data Fig. 4 VEGF-C signals specifically in lymphatic endothelial cells in the meninges and deep cervical lymph nodes and provides survival benefits in a manner that depends on the administration time point.
a, Gating strategy for LECs and BECs. b, Concatenated FACS plots of LECs and BECs from meninges and lymph nodes, depicting AKT phosphorylation intensity. The experiment was repeated independently with similar results. c, Quantification of the AKT(pS473)-positive population and mean fluorescence intensity (MFI) within LECs and BECs in the meninges and deep cervical lymph nodes (meninges: wild type, n = 5; AAV-VEGF-C, tumour + Luc mRNA, tumour + VEGFC mRNA, n = 8; lymph nodes: wild type, n = 5; AAV-VEGF-C, n = 8; tumour + Luc mRNA, n = 7; tumour + VEGFC mRNA, n = 8). d, Fluorescence microscopy images of deep cervical lymph nodes after treatment with VEGFC mRNA in tumour-bearing mice (CD31, red; LYVE1, green; DAPI, blue). e, Fluorescence microscopy images of meninges after treatment with VEGFC mRNA in tumour-bearing mice (CD31, red; LYVE1, green; DAPI, blue). The experiment was repeated independently with similar results. f–h, Mice were treated with AAV-VEGF-C or VEGFC mRNA at different time points relative to GL261-Luc tumour inoculation (day 0). Tumour growth kinetics (g, h) and survival (f) were monitored (n = 5 for all groups, no tx refers to mice receiving no treatment). Data are mean ± s.d. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test or two-sided log-rank Mantel–Cox test).
Extended Data Fig. 5 Therapeutic delivery of VEGF-C potentiates checkpoint inhibitor therapy even at late stages of tumour development.
a, Related to Fig. 3c, d. Quantification of the number of cells per tumour-bearing brain hemisphere or lymph node using CountBright absolute counting beads and autocounter (see Methods for details) (lymph nodes: GFP mRNA, n = 6; VEGFC mRNA, n = 12; tumour: GFP mRNA, n = 3; VEGFC mRNA, n = 5). b, Mice that rejected tumours after combination therapy with VEGFC mRNA and anti-PD-1 (RMP1-14) were rechallenged in the contralateral hemisphere and observed for survival (naive, n = 5; day 100 rejected, n = 4). c, T cells from lymph nodes and spleens from mice that rejected tumours after combination therapy with VEGFC mRNA and anti-PD-1 (RMP1-14) or naive wild-type mice were isolated and transferred into naive wild-type mice intravenously. After 24 h, GL261 tumours were inoculated intracranially and the mice were observed for survival (wild type, n = 5; wild type with transfer of naive T cells, n = 5; wild type with transfer of memory T cells, n = 7). d, Mice inoculated with 50,000 GL261-Luc cells were treated with VEGFC mRNA or GFP mRNA (day 7) and with either anti-PD-1 (RMP1-14) antibodies or isotype antibodies (days 7, 9 and 11), and monitored for survival. Mice were depleted of CD4 or CD8 T cells using anti-CD4 (GK1.5) or anti-CD8 (YTS169.4) antibodies starting one day before tumour inoculation and redosed every four days afterwards (VEGFC mRNA + anti-PD-1, n = 6; GFP mRNA + anti-PD-1, n = 6; VEGFC mRNA + anti-PD-1 + anti-CD4, n = 5; VEGFC mRNA + anti-PD-1 + anti-CD8, n = 5). e, Schematic of experimental design for the results shown in f and g. f, g, Mice inoculated with 50,000 CT-2A–BFP cells (f) or CT-2A cells (g) were treated with VEGFC mRNA or GFP mRNA (day 7) and either with PBS or with anti-PD-1(RMP1-14) and/or anti-4-1BB (LOB12.3) antibodies (days 7, 9 and 11), and monitored for survival (f, VEGFC mRNA + anti-PD-1 + anti-4-1BB, n = 5; GFP mRNA + anti-PD-1 + anti-4-1BB, n = 5; VEGFC mRNA + PBS, n = 4; GFP mRNA + PBS, n = 6; g, n = 5 for all groups except VEGFC mRNA + anti-4-1BB + anti-PD-1, n = 7). h–j, Mice inoculated with 50,000 GL261 cells were treated with VEGFC mRNA or GFP mRNA (day 7) and either with PBS or with anti-PD-1 (RMP1-14) (h), anti-TIM3 (RMT3-23) (i) or anti-CTLA4 (9H10) antibodies (j) (days 7, 9 and 11), and monitored for survival (n = 5). For i and j, the same control mice were used for the GFP mRNA + PBS and VEGFC mRNA + PBS groups. k, Schematic of experimental design for the results shown in l. l, Mice inoculated with 50,000 GL261-Luc cells were treated with VEGFC mRNA or GFP mRNA (day 20) and either with PBS or with anti-PD-1 (RMP1-14) and anti-TIM3 (RMT3-23) antibodies (days 20, 22 and 24), and monitored for survival (n = 5). *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test or two-sided log-rank Mantel–Cox test).
Extended Data Fig. 6 Validation of endogenous retrovirus EMV2 as a tumour antigen for GL261.
a, RNA-seq analysis of mouse endogenous retrovirus elements in publicly available datasets on C57BL/6J mice brains and GL261 cell lines from various sources. b, Quantification of endogenous retrovirus elements in healthy brain and tumour tissue, from RNA-seq (brain, n = 9; tumour, n = 6). c, Gating strategies for tetramer staining. d, Mice were injected with 500,000 GL261 cells or PBS in the flank. Seven days after tumour inoculation, draining inguinal lymph nodes were collected and emv2-env (Kb-restricted peptides aa 604–611 of p15E protein (KSPWFTTL)) tetramers were used to validate tumour-specific T cell proliferation. The experiments were repeated twice independently with similar results. Data are mean ± s.e.m.
Extended Data Fig. 7 VEGF-C-dependent anti-PD-1 potentiation is specific to VEGF-C among proteins of the VEGF family, and is not caused by a direct effect on tumour or immune cells.
a, b, C57BL/6 mice received an injection of AAV-CTRL or AAV-s(oluble)VEGFR-3 intracisternally through the cisterna magna. After four weeks, mice were euthanized and the dura mater was collected to image the lymphatic vasculature (LYVE1) in the confluence of sinuses (a). The relative area of lymphatic vasculature in the confluence of sinuses was quantified (b) (n = 5). c, Mice were pretreated with AAV-sVEGFR-3 four to six weeks before tumour inoculation. Seven days after tumour inoculation, mice were treated with VEGFC mRNA and anti-PD-1 (RMP1-14) antibodies (days 7, 9 and 11) (n = 5). d–f, Mice were treated with 5 µg of recombinant protein (VEGF-A, VEGF-B, VEGF-Cs or VEGF-D) in combination with anti-PD-1 (RMP1-14) antibodies (days 7, 9 and 11) and monitored for survival and tumour growth (n = 5). g–k Mice were injected with CT-2A–BFP tumours and were treated with VEGFC mRNA at day 7. On day 8, brains and lymph nodes from all mice were collected and analysed using flow cytometry. The experiment was repeated independently with similar results. g, Sample flow cytometry plots of experiments. h–k, Quantification of experiments (n = 5). l, Flow cytometry was used to evaluate the expression of VEGFR-3 in GL261 cells. A VEGFR3–GFP plasmid was transfected into HEK293T cells as a positive control. The experiment was repeated independently with similar results. m, MTT assay to measure the proliferation of GL261 cancer cells in the presence of VEGF-C after 48 h (n = 8 per group). n, Flow cytometry was used to evaluate the expression of VEGFR-3 in leukocyte compartments in the tumour. The experiment was repeated independently with similar results. o, Bone-marrow-derived dendritic cells were cultured with VEGF-C and evaluated for the expression of costimulatory molecules in the naive state (top row) or with LPS stimulation (bottom row). p, Isolated T cells were activated in vitro with CD3 or CD28 and IL-2 in the presence of VEGF-C. Data are mean ± s.d. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test or two-sided log-rank Mantel–Cox test).
Extended Data Fig. 8 Flow cytometry analysis of myeloid cell populations after treatment with VEGF-C.
Mice bearing 7-day tumours were treated with Luc mRNA or VEGFC mRNA and evaluated for changes in the populations of myeloid cells. a, Gating strategy for different myeloid cells. b–d, Cell counts of different cell types were measured at different time points after VEGFC mRNA treatment. cDC, classical dendritic cell; DC, dendritic cell; pDC, plasmacytoid dendritic cell. e–g, MHC II and CD80 MFI levels were quantified and showed no significant alteration after treatment with VEGFC mRNA. b and e are leukocytes from brain tissue; c and f are leukocytes from draining cervical lymph nodes; d and g are leukocytes from meninges (n = 3, 3 mice were pooled for each replicate). Data are mean ± s.e.m *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test).
Extended Data Fig. 9 VEGF-C treatment changes T cell phenotypes and functionality.
Mice bearing 7-day-tumours were treated with Luc mRNA or VEGFC mRNA and evaluated for changes. a, Mouse brains were collected two days after treatment with VEGFC mRNA, cryosectioned and analysed by immunofluorescence microscopy. b, Gating strategy for flow cytometry analysis of T cells. c, Example of TCF7 staining in CD3+CD8+CD44+ cell populations after VEGFC mRNA treatment. The experiment was repeated twice independently with similar results. d, Percentage of TCF7+ T cells in the CD3+CD8+CD44+ population in the brain (Luc mRNA, n = 14; VEGFC mRNA, n = 9; data pooled from 3 independent experiments). e, Number of CD3+CD8+CD44+ cells producing IFNγ, TNF, IL-2 and GZMB in the brain (n = 3; 3 mice were pooled for each sample). Violin plots display quartiles (dotted horizontal lines), median (dashed line) and minimum and maximum values (solid lines). f, Quantification of cell counts in different compartments after treatment with VEGFC mRNA. Percentage of cells expressing specific transcription factors or immune checkpoint inhibitors after treatment with VEGFC mRNA. g, Gating strategy for cytokine production in T cells. h, i, Quantification of T cells expressing multiple cytokines (n = 3, 3 mice were pooled for each replicate). Data are mean ± s.e.m. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-tailed unpaired Student’s t-test).
Extended Data Fig. 10 Therapeutic application of VEGF-C mediates protection against intracranial melanoma cell lines when combined with various checkpoint inhibitors.
a, Schematic of experimental design for Fig. 4a–c and b–e. b, Growth kinetics of flank tumours from Fig. 4a–c were measured using a caliper (n = 12 for all groups except the ligation group, n = 7). c–e, Mice were given either only B16 intracranial tumours (IC) (c) or both a B16 intracranial tumour and a B16 flank tumour (FT) (d) and treated with GFP mRNA or VEGFC mRNA on day 7, and anti-PD-1 (RMP1-14), anti-CTLA4 (9H10) and anti-TIM3 (RMT3-23) on days 7, 9 and 11. f, Flow cytometry gating strategy for Fig. 4e, f. g, Schematic of VEGF-C-induced tumour rejection. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001 (two-sided log-rank Mantel–Cox test).
Supplementary information
Source data
Rights and permissions
About this article
Cite this article
Song, E., Mao, T., Dong, H. et al. VEGF-C-driven lymphatic drainage enables immunosurveillance of brain tumours. Nature 577, 689–694 (2020). https://doi.org/10.1038/s41586-019-1912-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1912-x
This article is cited by
-
Blood pressure lowering enhances cerebrospinal fluid efflux to the systemic circulation primarily via the lymphatic vasculature
Fluids and Barriers of the CNS (2024)
-
The cytoskeleton adaptor protein Sorbs1 controls the development of lymphatic and venous vessels in zebrafish
BMC Biology (2024)
-
Enhanced meningeal lymphatic drainage ameliorates lipopolysaccharide-induced brain injury in aged mice
Journal of Neuroinflammation (2024)
-
Piezo1 regulates meningeal lymphatic vessel drainage and alleviates excessive CSF accumulation
Nature Neuroscience (2024)
-
Nasopharyngeal lymphatic plexus is a hub for cerebrospinal fluid drainage
Nature (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.